soln set 10
advertisement

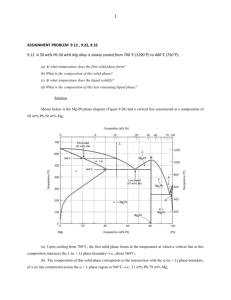
E344 SOLUTION SET 10 2012 FALL 1. Callister 9.1 Consider the sugar–water phase diagram of Figure 9.1. (a) How much sugar will dissolve in 1500 g water at 90°C (194°F)? (b) If the saturated liquid solution in part (a) is cooled to 20°C (68°F), some of the sugar will precipitate out as a solid. What will be the composition of the saturated liquid solution (in wt% sugar) at 20°C? (c) How much of the solid sugar will come out of solution upon cooling to 20°C? Solution (a) We are asked to determine how much sugar will dissolve in 1000 g of water at 90°C. From the solubility limit curve in Figure 9.1, at 90°C the maximum concentration of sugar in the syrup is about 77 wt%. It is now possible to calculate the mass of sugar using Equation 4.3 as Solving for msugar yields msugar = 5022 g (b) Again using this same plot, at 20°C the solubility limit (or the concentration of the saturated solution) is about 64 wt% sugar. (c) The mass of sugar in this saturated solution at 20°C may also be calculated using Equation 4.3 as follows: which yields a value for of 2667 g. Subtracting the latter from the former of these sugar concentrations yields the amount of sugar that precipitated out of the solution upon cooling ; that is 2. Based on the data given in Callister 9.7 sketch well-labeled cooling curves (Temperature vs. time) describing the solidification of pure Si, pure Ge, and a Si-50 wt% Ge alloy. Solution 3. Callister 9.18 A 30 wt% Sn-70 wt% Pb alloy is heated to a temperature within the α + liquid phase region. If the mass fraction of each phase is 0.5, estimate: (a) The temperature of the alloy (b) The compositions of the two phases Solution (a) We are given that the mass fractions of α and liquid phases are both 0.5 for a 30 wt% Sn-70 wt% Pb alloy and asked to estimate the temperature of the alloy. Using the appropriate phase diagram, Figure 9.8, by trial and error with a ruler, a tie line within the α + L phase region that is divided in half for an alloy of this composition exists at about 230°C. (b) We are now asked to determine the compositions of the two phases. This is accomplished by noting the intersections of this tie line with both the solidus and liquidus lines. From these intersections, Cα = 15 wt% Sn, and CL = 43 wt% Sn. 4. Core 10.2 A) A Cu rod 1 cm in diameter and 20 cm long rod is joined at one end to an identical rod of Ni to form a rod 40 cm long with a perfectly sharp interface in the middle. Suppose this couple is heated to 1000 oC and held there for several hours. Sketch profiles of Cu concentration across the interface for t=0 and for three subsequent times. No calculations are needed, simply a well labeled sketch. B) Suppose an identical couple is made from rods of pure lead and pure tin and heated to 175 oC. Draw a similar set of composition profiles. You should refer to the Cu-Ni and Pb-Sn phase diagrams. Solution A) At 1000 C the material is in the single phase region. o B) It is not possible to have a single-phase Sn-Pb ally with a composition in the range of 17-97.8 wt% Sn at 175 oC 5. Callister 9.28 Briefly explain why, upon solidification, an alloy of eutectic composition forms a microstructure consisting of alternating layers of the two solid phases. Solution Upon solidification, an alloy of eutectic composition forms a microstructure consisting of alternating layers of the two solid phases because during the solidification atomic diffusion must occur, and with this layered configuration the diffusion path length for the atoms is a minimum. 6. Callister 9.29 What is the difference between a phase and a microconstituent? Solution A “phase” is a homogeneous portion of the system having uniform physical and chemical characteristics, whereas a “microconstituent” is an identifiable element of the microstructure that may consist of more than one phase) 7. From spring 2007 final exam: The Pb-Sn equilibrium binary phase diagram is given on the following page. Identify: A. the melting temperature of pure Pb. B. the melting temperature of a Pb-Sn alloy with the eutectic composition. C. the maximum solid solubility of Pb dissolved in (Sn) For Sn-40 at% Pb, determine at T1=250 oC, T2 = 200 oC, and T3 = 150 oC: , the phase(s) present; the composition of each phase; the amount of each phase present; and sketch a possible microstructure Solution: (A) 327 oC; (B) 183 oC; (C) 1.45% at% Pb At T1 = 250 oC: Phase: single-phase liquid, cL = Sn - 40% at Pb, Amount: 100% L ------------------------------------------------------------------------------------------------------------At T2 = 200 oC: Phases: (Sn) + (Pb) fL = Composition: cL = Sn – 32 at% Pb ; c(Pb) = Sn - 73% at Pb. 73 − 40 = 0.80 73 − 32 f(Pb ) = 40 − 32 = 0.20 73 − 32 _____ 1.0 + --------------------------------------------------------------------------------------------------------------At T3 = 150 oC: Phases: L + (Pb) € Composition: c(Sn) = Sn – 1 at% Pb ; c(Pb) = Sn - 83% at Pb. f(Sn ) = 83 − 40 = 0.52 83 −1 f(Pb ) = 40 −1 = 0.48 83 −1 + € _____ 1.0 8. Refer to the Al-Li phase diagram from the AlLi_supplement.pdf file. A) Suppose an alloy with a composition of Al – 28.2 at %Li is held at a temperature of 601.5 o C. The alloy is a single-phase liquid. If this alloy is now cooled to T=600.0 oC it will solidify. Suppose the alloy is instantly quenched to room temperature at the moment solidification is complete, so no diffusion takes place. Determine: A) i) the phase or phases present; ii) the composition of each phase; iii) the fraction of each phase present; and iv) draw a well-labeled sketch of a likely microstructure. B) Suppose, instead of immediately quenching, after solidification the alloy is held at 600.0 o C for one hour and then quenched instantly to room temperature. Repeat questions Ai – Aiv. Solution A) i) two phases: α and β fα = 45 − 28.2 = 0.73 45 − 22 fβ = 28.2 − 22 = 0.27 45 − 22 iii) ii) cα = Al - 22 at% Li cβ = Al - 45 at% Li + iv) € B) i, ii, and iii will be exactly the same as above. The difference will be in the microstructure. The β lamellae will spherodize in order to reduce the overall surface energy (by reducing the total amount of α/β interface). _____ 1.0