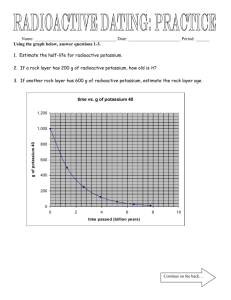
1. Which equation represents the radioactive decay 5. In
advertisement

1. Which equation represents the radioactive decay of 226 88 Ra? 222 4 1) 226 88 Ra - 86 Rn + 2He 226 226 0 2) 88 Ra - 89 Ac + –1e 226 0 3) 226 88 Ra - 87 Fr + +1e 225 1 4) 226 88 Ra - 88 Ra + 0n 2. The chart below shows the spontaneous nuclear decay of U-238 to Th-234 to Pa-234 to U-234. 5. In the equation: 234 91 Pa → 234 92 U +X The X represents a 1) helium nucleus 2) beta particle 3) proton 4) neutron 6. According to the equation: 4 X → 208 82 Pb + 2He The nucleus correctly represented by X is 1) 204 80 Hg 2) 212 84 Po 3) 204 80 Bi 4) 212 84 Pb 7. In the equation: 234 90 Th What is the correct order of nuclear decay modes for the change from U-238 to U-234? 1) î‚ decay, decay, î‚‚ decay 2) î‚ decay, î‚ decay, í decay 3) í decay, í decay, î‚ decay 4) í decay, î‚ decay, î‚ decay 3. Which product of nuclear decay has mass but no charge? 1) alpha particles 3) gamma rays 2) neutrons 4) beta positrons 4. In the equation: 239 40 Th 234 → 91 Pa + X The symbol X represents 1) 0+1e 0 2) –1 e 1 3) 0n 4) 11H 234 → 91 Pa + X Which particle is represented by X? 0 1) –1 e 4 2) 2He 3) 11H 4) 0+1e 8. In the equation: 226 88 Ra → 222 86 Rn +X X represents 1) a neutron 2) a proton 3) a beta particle 4) an alpha particle 9. Given the reaction: 234 91 Pa → X + 0 –1e When the equation is correctly balanced the nucleus represented by X is 1) 234 92 U 2) 235 92 U 3) 230 90 Th 4) 232 90 Th 10. Given the equation: 18 9F 18 → 8O + X Which symbol, when substituted for X, correctly balances the equation? 1) 11H 2) 10n 0 3) –1 e 0 4) +1e 11. In the equation: 4 X → 226 88Ra + 2He The symbol X represents 1) 222 86Th 230 2) 90Th 3) 222 86Rn 230 4) 90Rn 12. A neutron has approximately the same mass as 1) an alpha particle 3) an electron 2) a beta particle 4) a proton 13. What is the half-life of sodium-25 if 1.00 gram of a 16.00-gram sample of sodium-25 remains unchanged after 237 seconds? 1) 47.4 s 3) 79.0 s 2) 59.3 s 4) 118 s 14. Based on Reference Table N, what is the fraction of a sample of potassium-42 that will remain unchanged after 62.0 hours? 15. What is the half-life and decay mode of Rn-222? 1) 1.91 days and alpha decay 2) 1.91 days and beta decay 3) 3.82 days and alpha decay 4) 3.82 days and beta decay 16. Based on Reference Table N, what fraction of a radioactive 90Sr sample would remain unchanged after 56.2 years? 1) 12 2) 14 3) 18 1 4) 116 17. How many days are required for 200. grams of radon-222 to decay to 50.0 grams? 1) 1.91 days 3) 7.64 days 2) 3.82 days 4) 11.5 days 18. Which radioisotope undergoes beta decay and has a half-life of less than 1 minute? 1) Fr-220 3) N-16 2) K-42 4) P-32 19. Based on Reference Table N, what fraction of a sample of gold-198 remains radioactive after 2.69 days? 1) 14 2) 12 3) 34 4) 78 20. After 32 days, 5 milligrams of an 80-milligram sample of a radioactive isotope remains unchanged. What is the half-life of this element? 1) 8 days 3) 16 days 2) 2 days 4) 4 days 21. According to Reference Table N, which radioactive isotope will retain only one-eighth (18 ) its original radioactive atoms after approximately 43 days? 1) gold-198 3) phosphorus-32 2) iodine-131 4) radon-222 22. According to Table N, which radioactive isotope is best for determining the actual age of Earth? 1) 238U 2) 90Sr 3) 60Co 4) 14C Base your answers to questions 23 through 26 on on the graph below. The graph represents the decay of radioactive material X into a stable decay product. 24. Each of the objects below has different amounts remaining of the original radioactive material X. Which object is most likely the oldest? 1) 2) 23. If radioactive material X were heated, the length of its half-life period would 1) decrease 3) remain the same 2) increase 3) 4) 25. Which graph best represents the relative percentages of radioactive material X and its stable decay product after 15,000 years?(The shaded region represents radioactive material while the non-shaded region represents stable decay products.) 1) 3) 2) 4) 26. What is the approximate half-life of radioactive material X? 1) 5,000 yr 3) 50,000 yr 2) 10,000 yr 4) 100,000 yr 27. Exactly how much time must elapse before 16 grams of potassium-42 decays, leaving 2 grams of the original isotope? 1) 8 × 12.4 hours 3) 3 × 12.4 hours 2) 2 × 12.4 hours 4) 4 × 12.4 hours 28. As a sample of the radioactive isotope 131I decays, its half-life 1) decreases 3) remains the same 2) increases 29. The half-life of a radioactive substance is 2.5 minutes. What fraction of the original radioactive substance remains after 10 minutes? 1) 1 2 2) 1 4 3) 1 8 4) 1 16 30. Approximately what fraction of an original Co–60 sample remains after 21 years? 1) 1 2 2) 1 4 3) 1 8 4) 1 16 31. The half-life of 131I is 8.07 days. What fraction of a sample of 131I remains after 24.21 days? 1) 1 2 2) 1 4 3) 1 8 4) 1 16 32. What was the original mass of a radioactive sample that decayed to 25 grams in four half-life periods? 1) 50 g 3) 200 g 2) 100 g 4) 400 g 33. What is the total number of grams of a 32-gram sample of 32P remaining after 71.5 days of decay? 1) 1.0 g 3) 8.0 g 2) 2.0 g 4) 4.0 g 34. In how many days will a 12-gram sample of 131 53 I decay, leaving a total of 1.5 grams of the original isotope? 1) 8.0 3) 20. 2) 16 4) 24 35. What mass of a 60.0-gram sample of 16N will remain unchanged after 28.8 seconds? 1) 3.75 g 3) 15.0 g 2) 7.50 g 4) 30.0 g 36. The half-life of 14 6 C is 5,730 years. What fraction of a 1 gram sample of 14 6 C would remain after 17,190 years? 1) 1 2 2) 1 4 3) 1 8 4) 1 16 42. If 8.0 grams of a sample of 60Co existed in 1990, in which year will the remaining amount of 60Co in the sample be 0.50 gram? 1) 1995 3) 2006 2) 2000 4) 2011 37. What is the number of half-life periods required for a sample of a radioactive material to decay to one-sixteenth its original mass? 1) 8 3) 3 2) 16 4) 4 44. As a sample of a radioactive element decays, its half-life 1) decreases 3) remains the same 2) increases 38. A sample of 131I decays to 1.0 gram in 40. days. What was the mass of the original sample? 1) 8.0 g 3) 32 g 2) 16 g 4) 4.0 g 45. If 80 milligrams of a radioactive element decays to 10 milligrams in 30 minutes, what is the element's half-life in minutes? 1) 10 3) 30 2) 20 4) 40 39. Compared to 37K, the isotope 42K has a 1) shorter half-life and the same decay mode 2) shorter half-life and a different decay mode 3) longer half-life and the same decay mode 4) longer half-life and a different decay mode 46. An 80 milligram sample of a radioactive isotope decays to 5 milligrams in 32 days. What is the half-life of this element? 1) 8 days 3) 16 days 2) 2 days 4) 4 days 40. What total mass of a 16 gram sample of 60Co will remain unchanged after 15.9 years? 1) 1.0 g 3) 8.0 g 2) 2.0 g 4) 4.0 g 41. A 40.0 milligram sample of 33P decays to 10.0 milligrams in 50.0 days. What is the half-life of 33 P? 1) 12.5 days 3) 37.5 days 2) 25.0 days 4) 75.0 days 43. If 3.0 grams of 90Sr in a rock sample remained in 1989, approximately how many grams of 90Sr were present in the original rock sample in 1933? 1) 9.0 g 3) 3.0 g 2) 6.0 g 4) 12. g 47. If a radioactive substance has a half-life of 9 days what fraction of its original mass would remain after 27 days? 1) 1 8 2) 1 4 3) 1 2 4) 3 4 48. The graph below represents the decay of a radioactive isotope. 52. The graph below represents the decay curve of a radioactive isotope. The half-life of this isotope is 1) 8 years 2) 30 years Based on Reference Table H, which radioisotope is best represented by the graph? 1) 32P 2) 131I 3) 198Au 4) 222Rn 49. Which sample will decay least over a period of 30 days? 1) 10 g of Au-198 3) 10 g of P-32 2) 10 g of I-131 4) 10 g of Rn-222 50. As the temperature of a sample of a radioactive element decreases, the half-life of the element 1) decreases 3) remains the same 2) increases 51. Which radioactive sample would contain the greatest remaining mass of the radioactive isotope after 10 years? 1) 2.0 grams of 198Au 2) 2.0 grams of 42K 3) 4.0 grams of 32P 4) 4.0 grams of 60Co 3) 45 years 4) 60 years 53. After 30 days, 5.0 grams of a radioactive isotope remains from an original 40.-gram sample. What is the half-life of this element? 1) 5 days 3) 15 days 2) 10 days 4) 20 days 54. Approximately how many grams of a 40-gram sample of 131 53 I will remain unchanged after 24 days? 1) 5 3) 13 2) 10 4) 20 55. The half-life of a radioactive isotope is 20.0 minutes. What is the total amount of a 1.00gram sample of this isotope remaining after 1.00 hour? 1) 0.500 g 3) 0.250 g 2) 0.333 g 4) 0.125 g 56. Which of the following radioisotopes has the shortest half-life? 1) 14C 2) 3H 3) 37K 4) 32P 57. A radioactive element has a half-life of 2 days. Which fraction represents the amount of an original sample of this element remaining after 6 days? 1) 1 8 2) 1 2 3) 1 3 4) 1 4 58. Which of the following 10-gram samples of radioisotope will decay to the greatest extent in 28 days? 1) 32 15P 2) 85 36Kr 3) 220 87 Fr 4) 131 53 I 59. An original sample of a radioisotope had a mass of 10 grams. After 2 days, 5 grams of the radioisotope remains unchanged. What is the half-life of this radioisotope? 1) 1 day 3) 5 days 2) 2 days 4) 4 days 62. Which isotopic ratio needs to be determined when the age of ancient wooden objects is investigated? 1) uranium-235 to uranium-238 2) hydrogen-2 to hydrogen-3 3) nitrogen-16 to nitrogen-14 4) carbon-14 to carbon-12 63. Which procedure is based on the half-life of a radioisotope? 1) accelerating to increase kinetic energy 2) radiating to kill cancer cells 3) counting to determine a level of radioactivity 4) dating to determine age 64. A radioactive-dating procedure to determine the age of a mineral compares the mineral's remaining amounts of isotope 238U and isotope 1) 206Pb 2) 206Bi 3) 214Pb 4) 214Bi 65. The course of a chemical reaction can be traced by using a 1) polar molecule 3) stable isotope 2) diatomic molecule 4) radioisotope 60. The decay of which radioisotope can be used to estimate the age of the fossilized remains of an insect? 1) Rn-222 3) Co-60 2) I-131 4) C-14 66. The radioisotope I-131 is used to 1) control nuclear reactors 2) determine the age of fossils 3) diagnose thyroid disorders 4) trigger fussion reactors 61. Which isotope is most commonly used in the radioactive dating of the remains of organic materials? 1) 14C 2) 16N 3) 32P 4) 37K 67. Radioisotopes used for medical diagnosis must have 1) long half-lives and be quickly eliminated by the body 2) long half-lives and be slowly eliminated by the body 3) short half-lives and be quickly eliminated by the body 4) short half-lives and be slowly eliminated by the body 68. A radioisotope which is sometimes used by doctors to pinpoint a brain tumor is 1) carbon-12 3) technetium-99 2) lead-206 4) uranium-238 Reference Tables Reference Tables Reference Tables Reference Tables Answer Key 1. 1 25. 3 2. 4 26. 1 3. 2 27. 3 4. 2 28. 3 5. 2 29. 4 6. 2 30. 4 7. 1 31. 3 8. 4 32. 4 9. 1 33. 1 10. 4 34. 4 11. 2 35. 1 12. 4 36. 3 13. 2 37. 4 38. 3 14. 11 32 or 0.3125 15. 3 39. 4 16. 2 40. 2 17. 3 41. 2 18. 3 42. 4 19. 2 43. 4 20. 1 44. 3 21. 3 45. 1 22. 1 46. 1 23. 3 47. 1 24. 1 48. 2 Answer Key 49. 3 50. 3 51. 4 52. 2 53. 2 54. 1 55. 4 56. 3 57. 1 58. 3 59. 2 60. 4 61. 1 62. 4 63. 4 64. 1 65. 4 66. 3 67. 3 68. 3