bromothymol blue - National Biochemicals Corp.
advertisement

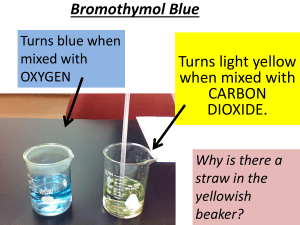
National Biochemicals, LLC. 1780 Enterprise Parkway • P.O. Box 1986, Twinsburg, OH 44087, USA Phone: 330. 425. 2522 • Fax: 330. 425. 2583 www.nationalbiochem.com BROMOTHYMOL BLUE Product No.: MB0128 CAS No.: 76-59-5 Storage Temperature: Ambient Synonyms: BTB; 3’,3”-Dibromothymolsulfonphthalein PROPERTIES HO Bromothymol Blue acts as a weak acid in solution. It can thus be in protonated or deprotonated form, appearing yellow or blue, respectively. It is bluish green in neutral solution. The deprotonation of the neutral form results in a highly conjugated structure, accounting for the difference in color. Br OH Bromothymol Blue is sparingly soluble in water, but soluble in alcohol, ether and in aqueous solutions of alkalis. It is less soluble in non-polar solvents such as benzene, toluene, and xylene, and practically insoluble in petroleum ether.1 Br O PREPARATION S O O To prepare a solution for use as a pH indicator, dissolve 0.10 g in 8.0 cm3 N/50 NaOH and dilute with water to 250 cm3. To prepare a solution for use as indicator in volumetric work, dissolve 0.1 g in 100 cm3 of 50% (v/v)ethanol.1 PRODUCT SPECIFICATIONS Molecular Formula: C27H28Br2O5S Molecular Weight: 624.38 Appearance: Dark Red Crystalline Powder Clarity of Solution: To Pass Visual Transition (pH 6.0 to 7.6): Yellow to Blue PRODUCT DESCRIPTION Bromothymol Blue is a chemical indicator for weak acids and bases. It is mostly used in applications that require measuring substances that would have a relatively neutral pH (near 7), such as managing the pH of pools and fish tanks. A common use is for measuring the presence of carbonic acid in a liquid. USES Bromothymol Blue may be used for observing photosynthetic activities, or as a respiratory indicator (turns yellow as CO2 is 2,3 added). A common demonstration of BTB’s pH indicator properties involves exhaling through a tube into a neutral solution of BTB. As carbon dioxide is absorbed from the breath into the solution, forming carbonic acid, the solution changes color from green to yellow. Thus, BTB is commonly used in middle school science classes to demonstrate that the more that muscles are used, the greater CO2 output. It may also be used in the laboratory as a biological slide stain. At this point, it is already blue and a drop or two is used on a water slide. The cover slip is placed on top of the water droplet and the specimen in it, with the blue coloring mixed in. It is sometimes used to define cell walls or nuclei under the microscope. page 1 Bromothymol Blue is used in obstetrics for detecting premature rupture of membranes. Amniotic fluid typically has a pH > 7.2, Bromothymol Blue will therefore turn blue when brought in contact with fluid leaking from the amnion. As vaginal pH normally is acidic, the blue color indicates the presence of amniotic fluid. The test may be false-positive in the presence of other alkaline substances such as blood, semen, or in the presence of bacterial vaginosis. PRECAUTIONS & DISCLAIMER This product is solely for Research and Development use. It is not for drug, household, or any other uses. Please review the Safety Data Sheet for information regarding risks and safe handling practices. REFERENCES 1. The Merck Index, 2. Sabnis, R.W., 3. Sabnis, R.W., Handbook of Biological Synthesis and Industrial Applications, Handbook of Entry #1445, (2006). Acid-Base Indicators, (2007). Dyes and Stains: 1st ed., (2010). NBC warrants that its products will conform with the information provided in this information sheet, though the Purchaser must determine the suitability of the products for their particular usage needs. Additional terms and conditions may apply. page 2 MB0128 • 1/29/13 • Rev. 1