Phase Equilibria and Phase Diagrams
advertisement

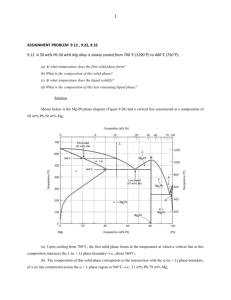
Chapter 7 Phase Equilibria and Phase Diagrams • The one-component phase diagram – Gibbs Phase Rule • Phase equilibria in a two-component system – The isomorphous diagram – The lever rule – Equilibrium solidification and microstructure of isomorphous alloys • Liquidius and solidus boundaries • Deviations from ideal behavior Chapter 7 Phase Equilibria and Phase Diagrams, Continued • Phase equilibria in a two-component system – – – – – The eutectic phase diagram The peritectic phase diagram The monotectic phase diagram Complex diagrams Phase equilibria involving solid-to-solid reactions 1 Why important ? Some properties that might be difficult to predict using a “common sense” without the knowledge of the phase diagrams • example 1: Melting temperature of a mixture AB (solution) of two components A and B could be either lower or higher than the melting point of each component (!). This could be a failure mechanism in electronic or mechanical components. But could also be used to your advantage. • example 2: Upon cooling to a lower temperature a phase transformation of a material could cause expansion, which could cause internal stresses and failure (e.g. tin food cans will crumble at low T) Example: Chip-Solder-Joint-Failure • example 3: No abrupt liquid-to-solid transformation when two components are present (solid + liquid in a temperature range) Why important ? Some properties that might be difficult to predict using a “common sense” without the knowledge of the phase diagrams • example 4: Tmelt (Sn) = 232 C, Tmelt (Pb) = 327 C…… but Tmelt(Sn0.62Pb0.38) = 183 C, so this is a common soldering alloy • example 5: Tmelt (Au) = 1064 C, Tmelt (Si) = 2550 C…… but Tmelt(Au0.97Si0.03) = 363 C, so thin layer of gold is used to attach Si chip to a ceramic substrate (shock protection) • example 6: Mechanical properties (hardness and tensile strength) of an alloy could be substantially higher than that of the individual components (e.g. hardness (AgCu) about twice the harness of Ag or Cu) 2 One-Component Phase Diagrams # of state variables (e.g. two: P and T) F=C–P+2 C- Components P- Number of phases F- Degrees of freedom Gibbs Phase rule: F=2 F=1 F=0 Two-Component Phase Diagrams Isomorphous system ( complete solubility over the composition range) F=1 T F=C–P+1 If pressure is fixed (1 atm) F=2 In a two-phase field need to specify either the temperature or the composition of one of the phases. Hume-Rothery Rules for substitutional solution: • • Xs Xl • The size < ~15%. The electronegativities and valance – similar The crystal structures of the two species must be the same to form a continuous series of solid solutions. 3 Two-Component Phase Diagrams fl + f s = 1 X o = X l fl + X s f s fl = 1 − f s Temperature X o = X l (1 − f s ) + X s f s X o = X l − X l fs + X s fs X o − X l = fs ( X s − X l ) ( Xo − Xl ) ( Xs − Xl ) ( Xs − Xo ) fl = ( Xs − Xl ) fs = Composition, XB The Lever Rule in a Two-Component System 4 Two-Component Phase Diagrams Temperature, C Two-Component Phase Diagrams Time Time Composition, XB 5 Two-Component Phase Diagrams Congruent melting maximum EAB > 0.5 (EAA + EBB) Deviation from ideal behavior Two-Component Phase Diagrams EAB > 0.5 (EAA + EBB) Deviation from ideal behavior Congruent melting minimum 6 Eutectic Phase Diagrams TA F=2 T Temperature F=1 T Xs Xl F = 2, must specify temperature and composition F=1 F=2 F=0 Xα Xβ F=2 Solvus F=1 T X α A TB Xl Xs Xβ X1 XE Composition, XB X2 B F = 1, must specify temperature or the composition of one of the phases F = 0, temperature and compositions of the phase are fixed. Cooling Curves and Phase Boundaries Temperature Alloy 1 Time Composition, XB 7 Cooling Curves and Phase Boundaries Temperature Alloy 2 Time Composition, XB Cooling Curves and Phase Boundaries Temperature Alloy 3 Time Composition, XB 8 Various physical properties and their relationship to a eutectic phase diagram Eutectic Phase Diagrams Temperature 1. For the alloy composition of 0.27 % B calculate the fraction of solid and the fraction of liquid that forms under equilibrium cooling at the eutectic T Composition, in % B 2. Calculate the amount of β and α that will form from the liquid just below the eutectic isotherm 3. Calculate the amount of α in the alloy at temperature just below the eutectic T 9 Eutectic Phase Diagrams Temperature Just above the eutectic temperature the fraction of liquid and solid are: 0.37 − 0.27 0.27 − 0.20 fα ≈ fl ≈ 0.37 − 0.20 0.37 − 0.20 f ≈ 0.59 fl ≈ 0.41 α The first solid that forms is called primary α Composition, in % B This liquid becomes the eutectic mixture of α and β when the temperature drops just below the eutectic temperature which is composed of: 0.37 − 0.20 0.73 − 0.37 fβ ≈ fα ≈ 0.73 − 0.20 0.73 − 0.20 f β ≈ 0.32 fα ≈ 0.68 Just below the eutectic temperature the microstructure is composed of primary α that formed above the eutectic temperature and α from the eutectic mixture fαtotal = fα primary + fαeutectic fαtotal = 0.59 + (0.41)(0.68) = 0.87 or fα = 0.73 − 0.27 f = 0.87 α 0.73 − 0.20 Microstructure Above and Below the Eutectic Temperature for an Off-Eutectic Alloy Just above TE Just below TE 10 Microstructure Above and Below the Eutectic Temperature for Off-Eutectic Alloys Just below TE Increasing primary α Decreasing eutectic Decreasing primary α Increasing eutectic Temperature Deviation from Hume-Rothery’s Rules Increasing deviation leads to decrease in the maximum solid solubility of B in α. A Composition, XB 11 Temperature Eutectic Phase Diagram, No Solid Solubility Composition, XB Eutectic Phase Diagrams Al-Si System 12 Methods for Determining a Phase Diagram Microstructure of an Aluminum-Silicon Alloy Primary α-aluminum α−aluminum / silicon eutectic 13 Phase Diagrams Containing Two Eutectics Line compound • Possible to have several solid solution regions: e.g. 2 eutectic reactions and 3 solid solutions (α, β, and γ) • Note that upon cooling from T max at the alloy composition X there is a phase change but no composition change (CONGRUENT melting) Peritectic Phase Diagrams • if both the L and S phases have a tendency to cluster, the liquidus temperature increase and the solidus temperature decreases • In addition, a miscibility gap (region of non-mixing) appears • A progressive increase in the clustering tendency leads to the PERITECTIC phase diagram l +α = β 14 The Use of Cooling Curves for Determining a Peritectic Phase Diagram l +α = β Temperature TA TL TP X2 XP X2 Composition, XB Time Analysis of a Peritectic Phase Diagram Alloy 1 Alloy 2 Alloy 3 Temperature Alloy 3 at T2 0.88 − 0.60 0.88 − 0.3 f l = 0.48 fl = Alloy 3 at T5 0.90 − 0.60 0.90 − 0.34 fα = 0.54 fα = Composition 15 Monotectic Phase Diagrams • A region of immiscibility (nonmixing) develops in the L phase L2 L1 • example: oil and water • Liquid1 = Liquid2 + α (solid) L2 L2 XM Review of Invariant Binary Reactions Eutectic Type Eutectic l α+β α Eutectoid α γ α+β Monotectic l1 α + l2 Monotectoid α2 α1 + β α α1 l γ l1 α2 β Al-Si, Fe-C β Fe-C l2 Cu-Pb β Al-Zn, Ti-V On cooling one phase going to two phases 16 Review of Invariant Binary Reactions Peritectic Type Peritectic l+β α Peritectoid α+β γ l α α β Fe-C β Cu-Al γ On cooling two phases going to one phase HW Questions 1. When a solid melts congruently, the liquid and solid have different / the same composition(s). 2. At constant temperature the fraction of the phases in a two-phase field changes / remains the same when the overall composition of the alloy is changed, but remains in the two-phase field. 3. Why would alloys close to the eutectic composition be suitable for castings rather than alloy compositions far from the eutectic composition? 4. On cooling when a two-phase liquid plus solid transforms to a solid phase the transformation is eutectic / peritectic in nature. 5. On cooling the peritectoid reaction written symbolically has one phase going to two / two phases going to one. 17 HW Questions • At what T an alloy containing 88% B will start melting? • At what T it will completely transform into liquid? • What is the composition of α phase for this alloy @ T8? • What is the maximum solid solubility of B in a and A in b? Whose rules apply here? • For an alloy containing 88% B, calculate the fraction of the liquid and solid phases and their compositions at temperature T3, T4, and T5 • At a temperature just below the eutectic temperature, how much β is primary β, what is the total fraction of β, and what is the fraction β in the eutectic. (Alloy composition is 88% B) Labeling Complex Phase Diagrams Temperature TB 1. Label all phase fields. 2. Identify all invariant reactions. TA A Composition, XB B 18 Labeling Complex Phase Diagrams Peritectic Eutectic Eutectic l =α +β Peritectic l +γ = β Eutectic l =γ +δ Eutectic Labeling Complex Phase Diagrams 19 Summary • One-component phase diagrams with temperature and pressure as the experimental variables that affect equilibrium. • Introduction to the Gibbs Phase Rule and its application to one-component systems. • Two-component systems and the rules that govern the composition of the phases, the number of phases and the amount of each phase at equilibrium. • The applications of these rules to complex, two-component systems illustrated that regardless of how complex the phase diagram appeared, the rules that were developed could be easily applied. 20