chemistry
Slide
1 of 37
11.1
Describing Chemical Reactions
On May 6, 1937, the huge airship
Hindenburg erupted into a fireball.
Within a short time, 210,000 cubic
meters of hydrogen had burned and
the airship was destroyed. The
chemical reaction that occurred is
“hydrogen combines with oxygen to
produce water.” You will learn to
represent this chemical reaction by a
chemical equation.
Slide
2 of 37
© Copyright Pearson Prentice Hall
1
11.1
Describing Chemical
Reactions
>
Writing Chemical Equations
Writing Chemical Equations
How do you write a word equation?
Slide
3 of 37
© Copyright Pearson Prentice Hall
11.1
Describing Chemical
Reactions
>
Writing Chemical Equations
Word Equations
To write a word equation, write the
names of the reactants to the left of the
arrow separated by plus signs; write the
names of the products to the right of the
arrow, also separated by plus signs.
Reactant + Reactant Product + Product
Slide
4 of 37
© Copyright Pearson Prentice Hall
2
11.1
Describing Chemical
Reactions
>
Writing Chemical Equations
Methane + Oxygen Carbon dioxide + Water
Slide
5 of 37
© Copyright Pearson Prentice Hall
11.1
Describing Chemical
Reactions
>
Writing Chemical Equations
iron + oxygen → iron(III) oxide
Slide
6 of 37
© Copyright Pearson Prentice Hall
3
11.1
Describing Chemical
Reactions
>
Writing Chemical Equations
Hydrogen Peroxide Water and Oxygen
Slide
7 of 37
© Copyright Pearson Prentice Hall
11.1
Describing Chemical
Reactions
>
Writing Chemical Equations
Chemical Equations
A chemical equation is a representation of a
chemical reaction; the formulas of the reactants
(on the left) are connected by an arrow with the
formulas of the products (on the right).
Slide
8 of 37
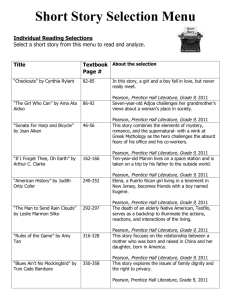
© Copyright Pearson Prentice Hall
4
11.1
Describing Chemical
Reactions
>
Writing Chemical Equations
How do you write a skeleton equation?
Slide
9 of 37
© Copyright Pearson Prentice Hall
11.1
Describing Chemical
Reactions
>
Writing Chemical Equations
Write the formulas of the reactants to
the left of the yields sign (arrow) and
the formulas of the products to the
right.
Slide
10 of 37
© Copyright Pearson Prentice Hall
5
11.1
Describing Chemical
Reactions
>
Writing Chemical Equations
A skeleton equation is a chemical equation
that does not indicate the relative amounts
of the reactants and products.
Here is the equation for rusting:
Fe + O2 Fe2O3
Slide
11 of 37
© Copyright Pearson Prentice Hall
11.1
Describing Chemical
Reactions
>
Writing Chemical Equations
A catalyst is a substance that speeds up the
reaction but is not used up in the reaction.
Without Catalyst
With Catalyst
Slide
12 of 37
© Copyright Pearson Prentice Hall
6
11.1
Describing Chemical
Reactions
>
Writing Chemical Equations
Slide
13 of 37
© Copyright Pearson Prentice Hall
Describing Chemical
Reactions
>
Writing Chemical Equations
Animation 12
Relate chemical symbols and formulas to the
information they communicate.
Slide
14 of 37
© Copyright Pearson Prentice Hall
7
Slide
15 of 37
© Copyright Pearson Prentice Hall
Slide
16 of 37
© Copyright Pearson Prentice Hall
8
Slide
17 of 37
© Copyright Pearson Prentice Hall
Practice Problems for Conceptual Problem 11.1
Problem Solving 11.2 Solve Problem
2 with the help of an interactive guided
tutorial.
Slide
18 of 37
© Copyright Pearson Prentice Hall
9
11.1
Describing Chemical
Reactions
>
Balancing Chemical Equations
Balancing Chemical Equations
What are the steps in writing a balanced
chemical equation?
Slide
19 of 37
© Copyright Pearson Prentice Hall
11.1
Describing Chemical
Reactions
>
Balancing Chemical Equations
To write a balanced chemical equation,
first write the skeleton equation. Then
use coefficients to balance the equation
so that it obeys the law of conservation
of mass.
Slide
20 of 37
© Copyright Pearson Prentice Hall
10
11.1
Describing Chemical
Reactions
>
Balancing Chemical Equations
This is a balanced equation for making a bicycle.
The numbers are called coefficients—small
whole numbers that are placed in front of the
formulas in an equation in order to balance it.
Slide
21 of 37
© Copyright Pearson Prentice Hall
11.1
Describing Chemical
Reactions
>
Balancing Chemical Equations
A chemical reaction is also described by a
balanced equation in which each side of the
equation has the same number of atoms of each
element and mass is conserved.
Slide
22 of 37
© Copyright Pearson Prentice Hall
11
Describing Chemical
Reactions
>
Balancing Chemical Equations
Simulation 11
Sharpen your skills by balancing chemical
equations.
Slide
23 of 37
© Copyright Pearson Prentice Hall
Slide
24 of 37
© Copyright Pearson Prentice Hall
12
Slide
25 of 37
© Copyright Pearson Prentice Hall
Slide
26 of 37
© Copyright Pearson Prentice Hall
13
Slide
27 of 37
© Copyright Pearson Prentice Hall
Practice Problems for Conceptual Problem 11.2
Problem Solving 11.4 Solve Problem
4 with the help of an interactive guided
tutorial.
Slide
28 of 37
© Copyright Pearson Prentice Hall
14
Slide
29 of 37
© Copyright Pearson Prentice Hall
Slide
30 of 37
© Copyright Pearson Prentice Hall
15
Slide
31 of 37
© Copyright Pearson Prentice Hall
Slide
32 of 37
© Copyright Pearson Prentice Hall
16