158
Cyclin D1 serves as a cell cycle regulatory switch in
actively proliferating cells
Dennis W Stacey
Much of our current understanding of the cell cycle involves
analyses of its induction in quiescent cells. To better
understand the control of cell cycle propagation and
termination, studies have been performed in actively cycling
cultures using time-lapse photography and quantitative image
analysis. These studies reveal a highly ordered sequence of
events required for promotion of continued proliferation. The
decision to continue cell cycle progression takes place in G2
phase, when cellular Ras induces the elevation of cyclin D1
levels. These levels are maintained through G1 phase and are
required for the initiation of S phase, at which time cyclin D1
levels are automatically reduced to low levels. The reduction of
cyclin D1 to low levels during S phase is required for DNA
synthesis, and forces the cell to induce high cyclin D1 levels
once again when it enters G2 phase. In this way, cyclin D1 is
proposed to serve as an active switch in the regulation of
continued cell cycle progression.
Addresses
Department of Molecular Biology, The Lerner Research Institute,
The Cleveland Clinic Foundation, 9500 Euclid Avenue, Cleveland,
OH 44195, USA
e-mail: staceyd@ccf.org
Current Opinion in Cell Biology 2003, 15:158–163
This review comes from a themed issue on
Cell regulation
Edited by Pier Paolo di Fiore and Pier Giuseppe Pelicci
0955-0674/03/$ – see front matter
ß 2003 Elsevier Science Ltd. All rights reserved.
DOI 10.1016/S0955-0674(03)00008-5
Abbreviations
BrdU bromodeoxyuridine
CDK
cyclin-dependent kinase
DAPI 40 ,6-diamidino-2-phenylindole
Rb
retinoblastoma protein
Introduction
Cell cycle studies are complicated by the fact that growing cell populations are normally randomized with respect
to cell cycle position. While techniques for separation of
cells according to cell cycle phase are available, most
biochemical studies involve synchronization [1]. A particularly attractive method to induce cell cycle synchrony
involves removal of serum growth factors to render a
culture quiescent. Upon re-addition of serum growth
factors, the culture begins to cycle synchronously for
several hours. With this approach, the molecular interactions that control the cell cycle re-entry have been studied
Current Opinion in Cell Biology 2003, 15:158–163
actively. Growth factor stimulation induces an increase in
Ras activity that in turn induces cyclin D1 levels. Cyclin
D1, in association with cyclin-dependent kinase (CDK)
phosphorylates the retinoblastoma protein (RB), blocking
its growth inhibitory activity and promoting the release
of bound E2F transcription factor [2,3]. These events
facilitate the activation of cyclin E–CDK2 and cyclin
A–CDK2, molecules required for the entry into and
completion of S phase [4].
Despite the wealth of information concerning the molecular interactions involved in release from quiescence,
little is known about the control of cell cycle progression
after the cells have started cycling. While quiescent cells
enter the cell cycle synchronously, each of them has its
own temporal pattern of response to growth stimuli, such
that within a few hours synchrony is lost. Without synchrony, the detailed biochemical studies upon which the
above conclusions are based cannot be performed. Consequently, we have little information on the steps
involved in propagation and ultimately the termination
of cell cycle progression from studies of serum-deprived
cultures. Moreover, even the information that is obtained
following serum stimulation cannot always be directly
applied to actively cycling cells. For example, while Rb is
phosphorylated in the late G1 phase of stimulated cells,
when actively cycling cultures were analyzed the protein
was phosphorylated early in G1 phase [5], or in all cell
cycle phases [6,7]. It appears that the phosphorylation of
Rb is more a consequence of growth state than cell cycle
position [8,9]. In addition, careful studies of cyclin E
expression using time-lapse analysis of actively cycling
cultures suggests that it is expressed as a consequence of
passage through the restriction point, rather than a
requirement for the restriction point as suggested from
studies in serum-stimulated cultures [10].
It was with these limitations in mind that studies of the
control of proliferation in asynchronous cultures were
carried out. Instead of attempting to drive all cells into
a single cell cycle phase to establish synchrony, various
means were used to determine the cell cycle position of
each individual cell within a rapidly proliferating culture.
In this way, all cell cycle phases could be studied simultaneously within the same culture without the necessity
of interrupting the very process being studied, cell cycle
progression. In this review, I present a summary of these
studies. The evidence presented will demonstrate the
central role played by G2 phase in the control of cyclin D1
expression, and in the control of cell cycle progression in
general. A model based upon these findings suggests that
www.current-opinion.com
Cyclin D1 in actively proliferating cells Stacey 159
cyclin D1 performs a critical switch function in the control
of continued cell cycle progression.
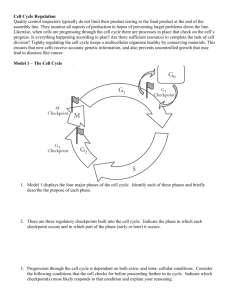
Figure 1
(Ras)
(CyclinD1)
Ras and cyclin D1 activity in cycling cells
Time-lapse analyses, quantitative fluorescence microscopy, and a combination of the two, have been used
to study the cell cycle in asynchronous cultures. In timelapse analyses, it is possible to determine the age of a cell,
or the time since it had passed through mitosis [11,12].
This information then yields a rough indication of the cell
cycle position of each living cell within the culture. The
first goal was to use this approach to determine at which
cell cycle point cellular Ras and cyclin D1 activities were
required for continued proliferation. The experiment
involved two time-lapse analyses separated by the microinjection of antibodies able to neutralize either cellular
Ras or cyclin D1. From the first film, it was possible to
predict the cell cycle position of the cells at the time of
their microinjection. From the second film, it was possible
to determine the fate of each injected cell. The results
indicated that the activity of cyclin D1 was required
throughout G1 phase. Surprisingly, however, cells
injected with anti-Ras antibody all divided exactly once
following the injection, and then terminated cell cycle
progression [13]. This latter result was totally unexpected, based on studies in quiescent cells, and indicates
that cellular Ras activity is required only during G2 phase,
but that its effects do not become apparent until after the
next mitosis (for an explanation, see Figure 1).
To understand the results following anti-Ras injections,
quantitative fluorescence microscopy was used. In this
approach, asynchronously proliferating cultures were
fixed and stained with fluorescent stains against selected
targets. Experiments were performed to ensure that the
fluorescent intensity following the stain was proportional
to the actual concentration of the target molecule within
the cell. When the fluorescence of DNA stained with
DAPI was quantified, the cell cycle position of each cell
could be determined (unless the cells were highly aneuploid). Staining with BrdU identified cells in S phase.
This information was then related to the level of cyclin
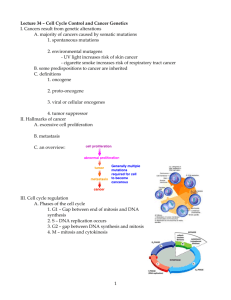
within each cell. For example, the profile of DNA versus
cyclin A is presented (Figure 2a). It is clear that BrdUunlabeled cells with lowest DNA-associated fluorescence
(the cells in G1 phase) had essentially background levels
of cyclin A. As cells entered S phase (as indicated by
increasing DNA content and labeling with BrdU), the
cyclin A content increased. In G2-phase cells containing
the highest DNA content that fail to incorporate BrdU,
the levels of cyclin A were maximal. The power of this
analysis is evident from the easy identification of tetraploid cells remaining in G1 (Figure 2c; DW Stacey,
unpublished data).
This approach was used to analyse the cell cycle expression profile of cyclin D1. Cyclin D1 levels were consiswww.current-opinion.com
M
G1
S
G2
Decision
M
G1
Execution
∗
∗
∗
Anti-Ras injection (∗)
Cell cycle
termination
Current Opinion in Cell Biology
The cell cycle requirement of cellular Ras activity. Regardless of the cell
cycle period in which anti-Ras antibody was injected, it blocked cell
cycle progression only following passage through one mitosis (M). To
explain this result, we assume that Ras activity is required during G2
phase, but its inhibitory effects are not manifested until after mitosis.
Thus, if a cell were injected during G1 phase it would proceed to G2
phase, where the decision to stop proliferating would be made. This
decision would not interfere with the upcoming mitosis, but would be
become apparent immediately thereafter. This same result would be
observed for cells injected during each cell cycle phase. Even if a cell
were injected during G2 phase, the inhibitory effects would not be
observed until after mitosis. On the basis of further experimentation, it is
clear that Ras induces cyclin D1 during G2 phase. It is the induction of
cyclin D1 that requires Ras activity, yet cyclin D1 itself is not required
until the next G1 phase. In this way, Ras is required during G2 phase,
but its effect is not felt until after mitosis.
tently found to be low in S phase, and high in G1 and G2
phases (Figure 2b,d) [14]. This profile has been observed
in each monolayer cell type analysed, including several
tumour cell lines. This result is best explained by assuming that cyclin D1 increases in G2 phase, and is then
maintained through mitosis and G1 phase, before its
suppression during S phase (Figure 3). Importantly, when
anti-Ras antibody was injected into cells and this analysis
was repeated at varying times thereafter, it was apparent
that cyclin D1 expression levels were highly dependent
upon cellular Ras activity. Interestingly, following the
injection of anti-Ras antibody the levels of cyclin D1 fell
first in G2 phase cells, and only thereafter did the levels
fall in G1 phase [14]. This information, together with the
time-lapse results described above, form the basis for our
model of the control of proliferation in continuously
cycling cells. It is proposed [14] that Ras is required
during G2 phase to stimulate cyclin D1 levels. Once
stimulated, these levels remain high through mitosis
and into G1 phase, even in the absence of continued
Ras activity. High levels of cyclin D1 in G1 phase
promote entry into S phase. In this way, Ras is needed
only during G2 phase, but its effect is not felt until the
G1/S phase transition (Figure 3) [15]. This explains why
anti-Ras-injected cells were able to pass through exactly
one mitosis following anti-Ras injection, and why cyclin
Current Opinion in Cell Biology 2003, 15:158–163
160 Cell regulation
Figure 2
Cyclin A
(a)
Cyclin D1
(b)
2500
BrdU (–)
+ BrdU (+)
3000
BrdU (–)
+ BrdU (+)
Cyclin D1 level
Cyclin A level
2000
1500
1000
500
2500
2000
1500
1000
500
0
0
G2
G1
G1
DNA level
(c)
(d)
2500
BrdU (–)
+ BrdU (+)
G2 phase
S phase
1500
1000
500
0
Polyploid
G1 phase
G1 phase
G2
G1
DNA level
Cyclin D1 level
Cyclin A level
2000
3000
BrdU (–)
+ BrdU (+)
2500
G1 phase
G2
DNA level
G2 phase
2000
1500
1000
500
S phase
0
G2
G1
DNA level
Current Opinion in Cell Biology
Quantitative analysis of cyclin A and cyclin D1 expression through the cell cycle. Human diploid fibroblasts (MRC5 cells) were pulsed with BrdU for 30
min before fixation and staining with fluorescent antibodies against cyclin A ([a], [c]) or cyclin D1 ([b], [d]). DNA was stained with DAPI. The
fluorescence associated with each of these stains was quantified on a cell by cell basis, and plotted with DNA fluorescent level versus cyclin
fluorescent level. Each symbol represents the properties of an individual cell, with the BrdU-positive and -negative cells noted. The profiles with cyclin
A (a) or cyclin D1 (b) were then marked to indicate the position of cells in each cell cycle phase ([c] and [d], respectively).
D1 levels during G2 phase were totally dependent on Ras
activity. In support of the central role played by G2 phase
in the control of cyclin D1 expression, oncogenic Ras was
injected into actively cycling cells and the levels of cyclin
D1 determined at various times thereafter. Despite the
fact that oncogenic Ras would be expected to be active in
all cell cycle phases, it was able to induce increased cyclin
D1 expression only during G2 phase [16].
The critical role of G2 phase in cell cycle
control
It is clear from many previous studies and from the data
presented above that the induction of cyclin D1 levels by
Ras activity is critical in the control of cell growth [3].
From the above studies, it is also clear that in cycling cells
this induction, and therefore the critical decision to continue proliferation, takes place during G2 phase. Thus,
the cell cycle phase that had in some cases been considered only a time for the cell to assess the completion of
Current Opinion in Cell Biology 2003, 15:158–163
DNA synthesis and to prepare for mitosis, is apparently
one of profound proliferative importance. The reason
why the cell must make this critical proliferative decision
during G2 phase might be no more complicated than the
fact that it must know whether to proceed into G1 phase
or to enter quiescence immediately following mitosis. On
the other hand, a decision made in G2 phase gives the cell
sufficient time to prepare for the consequences of that
decision well before it reaches the G1/S phase boundary
where the cell becomes committed to complete another
round of replication. This might be particularly important
for rapidly cycling cells where a shortened G1 phase is
necessary for a maximal growth rate. Whatever the explanation, it is proposed that for a cycling cell to continue
proliferation cyclin D1 must be induced in a Ras-dependent manner during G2 phase [14,15].
The molecular mechanism of the induction of cyclin D1
during G2 phase, therefore, is of central importance in
www.current-opinion.com
Cyclin D1 in actively proliferating cells Stacey 161
Figure 3
Cyclin D1
S
G2
M
G1
S
Ras required
Cyclin D1 required
Current Opinion in Cell Biology
The expression profile of cyclin D1 in actively cycling cells. This figure
indicates the expression levels of cyclin D1 as deduced from the data
presented. Note that cyclin D1 is induced specifically during G2 phase,
remains high through mitosis and into G1 phase, and then declines
again as the cells enter S phase. Ras activity is required for the
stimulation of cyclin D1 during G2 phase; while cyclin D1 itself is
required until the initiation of DNA synthesis, after which its levels
rapidly fall.
understanding the control of proliferation. On the basis of
in situ hybridization studies, it is clear that the levels of
cyclin D1 mRNA do not vary enough through the cell
cycle of actively cycling cells to account for the rapid
increase in cyclin D1 during G2 phase. Indeed, this
G2-phase increase in cyclin D1 levels has been observed
even in cells treated with a-amanitin to block new mRNA
synthesis [17]. Therefore, post-transcriptional control
mechanisms must be involved in the increase of cyclin
D1 levels during G2 phase. Moreover, it is clear that the
stability of cyclin D1 protein is altered during the cell
cycle, with a decreased half life observed in S phase [17].
Further study is necessary to determine the signaling
pathways and molecular mechanisms of post-transcriptional regulation of cyclin D1 levels during G2 phase.
In an effort to determine the role of altered protein
stability in cyclin D1 regulation, studies of protein degradation were initiated. It was demonstrated that the rapid
decline of cyclin D1 levels during S phase was dependent
upon proteasomal degradation, because when cells were
treated with MG132 or other proteasomal inhibitors the
low levels of cyclin D1 normally present during S phase
rapidly increased (DW Stacey, unpublished data). Even
though the increase in cyclin D1 levels was most evident
during S phase because of the normally low levels of
protein during this period, quantitative analyses demonstrated that the cyclin D1 levels increased also during G1
and G2 phases (DW Stacey, unpublished data).
Further study will be required to determine if altered
protein stability alone is sufficient to account for the
increased levels of cyclin D1 observed during G2 phase.
www.current-opinion.com
It appears, however, that the decline in S phase is not
dependent upon the signaling environment in the cell,
but is likely to be a regulated by cell cycle progression
directly. The necessity of this decline in cyclin D1 levels
during S phase can be explained by the observation some
time ago by Pagano et al. [18], who demonstrated that
cyclin D1 is inhibitory to DNA synthesis. When cyclin D1
levels were elevated by ectopic expression, DNA synthesis was blocked. It was further shown that this resulted
from the ability of cyclin D1 to bind and inactivate PCNA,
an essential component of the replication complex [18,19].
It is clear, therefore, that cyclin D1 levels must be low
during S phase, and that this decline is a fundamental
characteristic of normal cell cycle progression.
Model
On the basis of the data summarized above, we propose
the following model to explain the control of cell cycle
progression in actively cycling cells. Cyclin D1 is required
for transition from G1 to S phase, the point at which the
cell becomes committed to complete another round of
cell division. Once this decision is made, however, cyclin
D1 levels must be suppressed to low levels to allow the
cell to synthesize DNA. The fact that cyclin D1 levels are
low during S phase forces the cell to make a decision
regarding cyclin D1 levels when it reaches G2 phase. If
conditions are conducive for continued growth, the cell
elevates its cyclin D1 levels during G2 phase, allowing the
cell to continue through the next cell cycle. If conditions
are not conducive for continued proliferation, however,
cyclin D1 levels remain low during G2 phase (Figure 4a).
The fact that cyclin D1 levels must be reduced during S
phase has two important implications. First, because it is
suppressed during S phase the cell must make a positive
determination to increase those levels during G2 phase if
cell cycle progression is to continue. Since the switch is
automatically turned off at S phase, a pro-active decision
to turn it back on must be made at G2 phase (Figure 4a).
We therefore propose that cyclin D1 functions as a switch
in the control of cell growth. This switch is automatically
turned off in S phase, requiring that it be turned on again
in G2 phase for proliferation to continue.
The second implication of the suppression of cyclin D1
during S phase also relates to the overall control of cell
growth. Since cyclin D1 plays such a central role in the
control of continued cell cycle progression, it might be
possible for a mutation to simply force expression of
cyclin D1 and thereby give its daughters a proliferative
advantage in the organism. The requirement for low
levels of cyclin D1 during S phase, however, reduces
the likelihood of this potentially disastrous situation by
requiring that simple overexpression of cyclin D1 is not
tolerated (Figure 4b). No cell, therefore, would be able to
proliferate unless cyclin D1 is subject to normal controls
over its expression. In this context, it is interesting to
Current Opinion in Cell Biology 2003, 15:158–163
162 Cell regulation
Figure 4
Conclusions
(+
Cyclin D1 levels
)G
ro
wt
hf
ac
tor
(a)
be maintained for optimal cell growth. It is possible that
both positive and negative influences over proliferation,
might define critical cell cycle regulatory molecules and
serve as a means to enforce their normal regulation.
(–) Growth factor
G2
M
G1
S
G2
Cell cycle phase
M
G1
S
(b)
Cell cycle
stimulation
Cyclin D1
G1
S
G2
Cell cycle
inhibition
p27Kip1
Current Opinion in Cell Biology
Models of cyclin D1 and cell cycle regulation. (a) Cyclin D1 is proposed
to serve as a switch to regulate the continuation of cell cycle
progression. Cyclin D1 must be present in G1 phase for the initiation of
DNA synthesis. Once DNA synthesis begins, however, cyclin D1 levels
are reduced to low levels throughout S phase. This reduction forces the
cell to make a positive decision during G2 phase to increase cyclin D1
levels if the cell is to continue cycling. If, however, conditions are not
conducive for continued proliferation, cyclin D1 levels remain low in G2
phase and the cell enters quiescence after mitosis. (b) Cyclin D1 is
required for entry into S phase, but must be reduced during S phase for
DNA synthesis to continue normally. This means that during each cycle,
cyclin D1 levels must be reduced to low levels and then stimulated
again. This control mechanism ensures that simple high levels of cyclin
D1 expression, which might otherwise lead to uncontrolled proliferation,
are not tolerated. Similarly, the levels of p27Kip1 are normally low in
cycling cells but are required during G1 phase for the assembly of cyclin
D1–CDK4. Again, simple elimination of p27Kip1, which might otherwise
lead to uncontrolled proliferation, is not tolerated. This dual-function
feature of these two critical control molecules requires their expression
during some cell cycle phases and their suppression during other
phases for the cell cycle to continue proliferation. In this way, the cell
ensures that normal regulatory patterns of these molecules are
maintained.
consider the situation with p27Kip1, another critical cell
cycle regulatory molecule. The levels of this growth
inhibitory molecule generally stay low in actively cycling
cells. Once again, a cell might gain a proliferative advantage by simply eliminating expression of this molecule
altogether. To ensure this does not take place, however,
p27Kip1 or a related growth suppressive molecule is
required for the formation of active cyclin D1–CDK4
complexes during G1 phase (Figure 4b) [20]. Thus, a
molecule that is normally growth suppressive has an
active role in promoting proliferation during G1 phase,
requiring that the normal regulation of p27Kip1 must also
Current Opinion in Cell Biology 2003, 15:158–163
The evidence suggests that cyclin D1 performs a critical
cell cycle regulatory function during G2 phase, an observation that was not made until studies in actively cycling
cells were performed. It is important to compare this
observation with other studies of cell cycle regulation
during G2 phase. DNA damage is known to induce a G2
phase arrest. The arrest presumably allows a cell to either
repair DNA or to block the proliferation of a potentially
genetically damaged cell. The pathways involved in this
arrest have been well characterized [21,22]. It is, however,
important to make a distinction between G2 phase arrest
and the control of cyclin D1 regulation discussed here. On
the one hand, DNA damage causes the cell to pause or
stop in its progression through G2 phase. On the other
hand, cyclin D1 expression has minimal consequences
upon the length of G2 phase [15]. Mitosis takes place
normally, but the proliferative fate of the cell following
division is altered depending upon the expression level of
cyclin D1 during G2 phase. Thus, two important and
fundamentally different control processes take place in
G2 phase. The decision to increase cyclin D1 is a normal
part of cell cycle progression, whereas G2 arrest is the
result of abnormal conditions.
Acknowledgements
I thank the members of the laboratory for helpful discussions of these
ideas — M Hitomi, Y Guo, K Yang, J Nye and J Harwalkar — and for
the experimental basis upon which they are based.
References and recommended reading
Papers of particular interest, published within the annual period of
review, have been highlighted as:
of special interest
of outstanding interest
1.
Cooper S: The Schaechter-Bentzon-Maaloe experiment and
the analysis of cell cycle events in eukaryotic cells.
Trends Microbiol 2002, 10:169-173.
2.
Olashaw N, Pledger WJ: Paradigms of growth control: relation to
Cdk activation. Science’s STKE: Signal Transduction Knowledge
Environment 2002, 2002:RE7.
3.
Sherr CJ, Roberts JM: CDK inhibitors: positive and negative
regulators of G1-phase progression. Genes Devel 1999,
13:1501-1512.
4.
Girard F, Strausfeld U, Fernandez A, Lam NJC: Cyclin A is required
for the onset of DNA replication in mammalian fibroblasts.
Cell 1991, 67:1169-1179.
5.
Burke LC, Bybee A, Linch DC: The retinoblastoma protein is
partially phosphorylated during early G1 in cycling cells but not
in G1 cells arrested with alpha-interferon. Oncogene 1991,
6:317-322.
6.
Coder D, Varvayanis S, Yen A: Late dephosphorylation of the RB
protein in G2 during the process of induced cell differentiation.
Eur J Cell Biol 1997, 72:159-165.
7.
Shayman JA, Cooper S: Phosphorylation-dephosphorylation of
retinoblastoma protein not necessary for passage through the
www.current-opinion.com
Cyclin D1 in actively proliferating cells Stacey 163
mammalian cell division cycle. Cell Mol Life Sci 2001,
58:580-595.
8.
Lillycrop KA, Bybee A, Latchman DS, Thomas NS: The
phosphorylation state of the retinoblastoma (RB) protein in G0/
G1 is dependent on growth status. J Biol Chem 1991,
266:20888-20892.
9.
Shayman JA, Cooper S: Revisiting retinoblastoma protein
phosphorylation during the mammalian cell cycle.
Cell Mol Life Sci 2001, 58:580-595.
This study presents the evidence that phosphorylation of the Rb protein
takes place throughout the cell cycle in actively cycling cells. Those cells
with hypophosphorylated Rb are likely to be cells within the culture whose
proliferation is retarded. This result is quite different than the conclusions
reached in studies with serum-deprived cultures.
10. Ekholm SV, Zickert P, Reed SI, Zetterberg A, Xu X: Accumulation
of cyclin E is not a prerequisite for passage through the
restriction point chromosomal localization and 50 sequence of
the human protein serine/threonine phosphatase 50 gene.
Mol Cell Biol 2001, 21:3256-3265.
Studies of cyclin E expression were performed with time lapse analyses in
actively cycling cultures. In these cells, cyclin E expression took place
following passage through the restriction point. The conclusions were
quite different than reported in serum-stimulated cultures, where cyclin E
expression apparently was required for the restriction point.
11. Larsson O, Zetterberg A: Existence of a commitment program
for mitosis in early G1 in tumour cells. Cell Prolif 1995, 28:33-43.
12. Stacey DW, Hitomi M, Kanovsky M, Gan L, Johnson EM: Cell
cycle arrest and morphological alterations following
microinjection of NIH3T3 cells with Pur alpha. Oncogene 1999,
18:4254-4261.
13. Hitomi M, Stacey DW: Cellular ras and cyclin D1 are required
during different cell cycle periods in cycling NIH 3T3 cells.
Mol Cell Biol 1999, 19:4623-4632.
www.current-opinion.com
14. Hitomi M, Stacey DW: Cyclin D1 production in cycling cells
depends on ras in a cell-cycle-specific manner. Curr Biol 1999,
9:1075-1084.
15. Hitomi M, Stacey DW: Ras-dependent cell cycle commitment
during G2 phase. FEBS Lett 2001, 490:123-131.
16. Sa G, Hitomi M, Harwalkar J, Stacey AW, Chen G, Stacey DW: Ras
is active throughout the cell cycle, but is able to induce cyclin
D1 only during G2 phase. Cell Cycle 2002, 1:50-58.
From this study, it is clear that Ras activity is able to induce cyclin D1 only
during G2 phase. Whatever the mechanism for induction of cyclin D1,
therefore, it is able to take place only during G2 phase.
17. Guo Y, Stacey DW, Hitomi M: Post-transcriptional regulation of
cyclin D1 expression during G2 phase. Oncogene 2002,
21:7545-7556.
It is clear from this work that the induction of cyclin D1 during G2 phase is
dependent upon post-transcriptional mechanisms.
18. Pagano M, Theodoras AM, Tam SW, Draetta GF, Chen J: Cyclin
D1-mediated inhibition of repair and replicative DNA synthesis
in human fibroblasts. Genes Dev 1994, 8:1627-1639.
19. Chen J, Peters R, Saha P, Lee P, Theodoras A, Pagano M, Wagner
G, Dutta A: A 39-amino-acid fragment of the cell cycle regulator
p21 is sufficient to bind PCNA and partially inhibit DNA
replication in vivo. Nucleic Acids Res 1996, 24:1727-1733.
20. Cheng M, Olivier P, Diehl JA, Fero M, Rousell MF, Roberts JM,
Sherr C: The p21(Cip1) and p27(Kip1) CDK ‘inhibitors’ are
essential activators of cyclin D-dependent kinases in murine
fibroblasts. EMBO J 1999, 18:1571-1583.
21. Abraham RT: Cell cycle checkpoint signaling through the ATM
and ATR kinases. Genes Dev 2001, 15:2177-2196.
22. Taylor WR, Stark GR: Regulation of the G2/M transition by p53.
Oncogene 2001, 20:1803-1815.
Current Opinion in Cell Biology 2003, 15:158–163