Translation and Post-Translational Modifications
advertisement

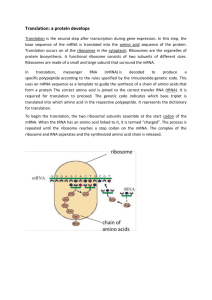
Translation Ribosomes synthesize a polypeptide according to the genetic instructions in mRNA. Translation in Eukaryotes occurs in 3 steps 1. Initiation • Ribosome binds to the mRNA and initiates at an AUG (methionine) codon 2. Elongation • The polypeptide is lengthened one amino acid at a time 3. Termination • Synthesis of the polypeptide terminates and the ribosome dissociates from the mRNA and the polypeptide Initiation of Translation (Eukaryotes) • The small ribosomal subunit forms an initiation complex with the initiator tRNA. • This complex binds the 5’ cap, then scans 5’ to 3’ in search of the first AUG codon. Initiation of Translation (Eukaryotes) • Once the first AUG is located then the large ribosomal subunit to binds to form the functional ribosome • An aminoacyl-tRNA then binds to the A site and a peptide bond is formed between methionine and the amino acid in the A site • Initiation is now complete and elongation can proceed Elongation • The nascent (growing) polypeptide is bound to the peptidyl-tRNA in the P-site • The incoming aminoacyl-tRNA binds to the A-site • Peptidyl transferase catalyzes a peptide bond between the new amino acid and the nascent polypeptide and breaks the bond between the nascent chain and the peptidyl-tRNA • The ribosome translocates by one codon along the mRNA Elongation • The tRNA in the E site exits the ribosome • The A site receives the next incoming aminoacyl-tRNA • A peptide bound is formed and the cycle continues until termination Termination • Releasing factors bind to termination codons (UAG, UAA, UGA) in the A-site. • Releasing factors facilitate hydrolysis of the nascent polypeptide from the peptidyl-tRNA thus freeing the polypeptide from the ribosome. Termination • Release of the polypeptide is followed by dissociation of the ribosomal subunits from the mRNA The 5’ cap and poly-A tail increase translation rates. • Specialized proteins bind to the 5’cap and 3’ poly-A tail of the mRNA. These proteins then bind to each other and thus bring the 5’ and 3’ ends of the mRNA together, forming a circle. Ribosomes that terminate translation are physically close to the 5’ cap where they bind and begin translation again. In this way the 5’ cap and poly-A tail function to increase translation rates. • Many ribosomes translate the same mRNA simultaneously. This complex is a polyribosome (polysome) Post Translational Modifications • The process of gene expression is not finished when an mRNA has been translated. • Many post translational modifications may be required for a polypeptide to fold into the shape required for function. Post Translational Modifications • Modifications may include: – Phosphorylation, the addition of a phospahate group – Methylation, the addition of a methyl group – Glycosylation, the addition of sugar groups – Disulfide bonds, the formation of covalent bonds between 2 cysteine amino acids. – Proteolytic Cleavage, the cutting of a sequence of amino acids from the polypeptide – Subunit binding to form a multisubunit protein Protein Folding begins during Translation • A polypeptide begins to fold as soon as it leaves the ribosome. • Some polypeptides can fold into their complete, mature conformation without help. • However, most polypeptides required chaperones to help them fold properly. Heat Shock Proteins (HSP) acts as Chaperones that aid protein folding • Chaperones bind to hydrophobic regions of the polypeptide and shield them from the aqueous environment until the entire polypeptide is translated. Then the chaperones help the protein to fold into its proper shape, with the hydrophobic R groups in the interior of the protein Misfolded proteins are destroyed by Proteosomes • The proteosome is a barrelshaped, multisubunit protease. Misfolded proteins enter one end and come out the other as small chains of amino acids (peptides) that are ultimately recycled. • Proteosomes are abundant, making up 1% of total cellular protein. Ubiquitin binds to misfolded proteins, targeting them for destruction by the proteosome •Misfolded proteins and proteins with oxidized or abnormal amino acids are seen as abnormal by the cell. Specialized enzymes attach chains of ubiquitin to these abnormal proteins. Ubiquitin has affinity for the proteasome and thus brings the abnormal protein to the proteasome for destruction. Almost without exception, proteins that enter the proteosome are first bound to ubiquitin. Plasma membrane proteins and secreted proteins are post-translationally modified in the Rough Endoplasmic Reticulum (RER) and the Golgi •Proteins bound for the plasma membrane must first enter the RER where they begin to fold and undergo chemical modification. •Proteins leave the RER in vesicles, which then fuse with the golgi, delivering their protein cargo. •Proteins are further modified in the golgi before being sent in vesicles to the plasma membrane where they are either secreted or integrated into the membrane. A signal sequence directs a nascent polypeptide to the rough endoplasmic reticulum • Only proteins that have an N-terminal signal sequence can enter the ER. • A signal recognition protein(SRP) binds to the signal sequence as it first exits the ribosome and brings the ribosome to the ER. • The polypeptide is threaded through the ER membrane and into the ER lumen A signal sequence directs a nascent polypeptide to the rough endoplasmic reticulum A signal sequence directs a nascent polypeptide to the rough endoplasmic reticulum A signal sequence directs a nascent polypeptide to the rough endoplasmic reticulum • Once the polypeptide is completely threaded into the ER, the signal sequence is cleaved off . • Now the polypeptide undergoes posttranslational modification and begins to fold Protein glycosylation occurs in the ER and the Golgi • Most proteins that enter the ER will be glycosylated, which means that an oligosaccharide (branched sugar group) will be covalently attached to the protein. • The sugar group may ultimately be important to the proteins function or it may simply act as an address label required to get the protein to its next cellular destination.