9.2 Network Covalent, Ionic, and Metallic Solids
advertisement

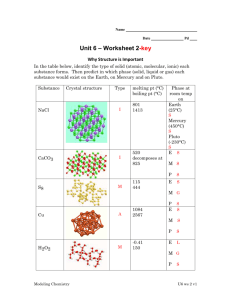
9.2 Network Covalent, Ionic, and Metallic Solids YOU ARE EXPECTED TO BE ABLE TO: • Classify non-molecular solids as either network covalent solids, ionic solids, or metallic solids. • Relate the physical properties of nonmolecular solids to the forces holding them together. Chemistry 1011 Slot 5 1 Network Covalent, Ionic, and Metallic Solids • Almost all substances that are gases or liquids at room temperature and pressure (25oC and 1 atm) are molecular • There are three distinct types of nonmolecular solids – Network covalent solids – Ionic solids – Metallic solids Chemistry 1011 Slot 5 2 Other Materials • There are also other materials with complex structures, for example – Polymers – natural and manufactured – Biological materials – Semiconductors Chemistry 1011 Slot 5 3 Network Covalent Solids • High melting points – To melt such a solid, covalent bonds must be broken • Insoluble in all common solvents – For solution to occur, covalent bonds must be broken • Poor electrical conductors – There are no mobile electrons (EXCEPT Graphite) Chemistry 1011 Slot 5 4 Some Examples - Carbon • Carbon can exist as both diamond and graphite • Both forms are network covalent solids • This site shows diagrams of both diamond and graphite structures: http://www.education.eth.net/acads/chemistry/allotropic_carb on-I.htm Chemistry 1011 Slot 5 5 Diamond • Diamond – pure carbon – Three dimensional structure based on tetrahedrally bonded carbon atoms. All single bonds • Very hard • Very high melting point • Does NOT conduct electricity – Diamond is transparent, strong, and very hard. It is a superb cutting tool. The atoms in diamond are very strongly held in position. Chemistry 1011 Slot 5 6 Diamond Structure • http://www.rensselaer.edu/dept/chemeng/WWW/faculty/cale/classes/SMP/lecture1/sld014.htm • http://iumsc11.chem.indiana.edu/common/Minerals/diamo nd/diamond.htm Chemistry 1011 Slot 5 7 Carbon • Graphite – pure carbon – Two dimensional, layer structure based on carbon atoms bonded in triangular planar arrangement. Each carbon atom forms 2 single and 1 double bond with its neighbours – Layers held together by dispersion forces • Very soft (layers slip) • Very high melting point • Conducts electricity as a result of delocalized electrons from double bonds Chemistry 1011 Slot 5 8 Graphite Structure • Graphite is black, soft, and an excellent lubricant. • It is easy to separate layers of atoms in graphite, to make them slide past one another. . Chemistry 1011 Slot 5 9 Other Network Covalent Solids • Quartz SiO2 – Three dimensional array of Si and O atoms – Major component of sand – Heated with CaCO3 and Na2CO3 gives glass • Other silicon – oxygen structures include – Mica – Asbestos – Talc Chemistry 1011 Slot 5 10 Ionic Solids - Observations • All ionic compounds: – are solids at room temperature – have high melting points – have wide temperature range for liquid phase – are hard, but brittle – conduct electricity in the liquid phase and in solution Chemistry 1011 Slot 5 11 Ionic Solids • High melting and non volatile – Strong interionic attractions must be overcome in order to separate ions of opposite charge • Do not conduct in solid phase – Ions must move in order for the material to conduct electricity. Ions can only move if the substance is melted or dissolved in water • Many dissolve in water, but not in organic solvents – Water is a highly polar substance Chemistry 1011 Slot 5 12 Attractions Between Ions • The force of attraction between oppositely charged ions depends on – The size of the charge • Greater charge = greater force of attraction, eg CaO vs NaCl – The size of the ions • Smaller ions get closer together = greater force of attraction, eg NaCl vs KBr http://www.scs.uiuc.edu/~chem315/solidstatestructures/naclf. htm Chemistry 1011 Slot 5 13 Metals - Observations • All metals: – – – – – – – are solids at room temperature (except for mercury); melting points vary have a wide temperature range for liquid phase are ductile and malleable conduct electricity in the solid and liquid phase, but not in the gaseous phase have high thermal conductivity are insoluble in water or other solvents are shiny Chemistry 1011 Slot 5 14 Metal Structures • Solid metals consist of regular arrays of metal atoms forming a crystal lattice • Delocalized valence electrons form a “sea of electrons” that can move freely throughout the metal • Delocalization provides stability • The “electron sea” is also present in the liquid phase Chemistry 1011 Slot 5 15 Explaining Metal Properties • Variable melting points – Melting point depends upon the strength of the bonds. This in turn generally depends upon the number of valence electrons. However, there is no simple correlation • High liquid temperature range – Vaporizing a metal requires the valence electrons to become localized, ie the metal bonds must be broken • Ductility and malleability – Metal crystal structures are flexible, layers in the crystal lattice can slide Chemistry 1011 Slot 5 16 Explaining Metal Properties • Conduction of electricity – Delocalized electrons are mobile. • Conduction of heat – Heat is carried through metals by colliding electrons • Solubility – Electrons cannot go into solution, nor cations by themselves • Shininess – Delocalized electrons absorb and emit light over a wide wavelength range Chemistry 1011 Slot 5 17 On-line materials • http://www.beyondbooks.com/psc92/3.asp Chemistry 1011 Slot 5 18