What Makes A Good Conductor?
advertisement

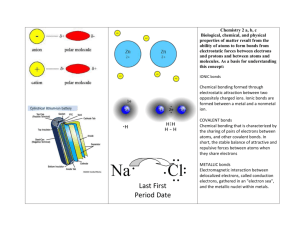
CHEMISTRY EXPERIMENTS ON FILETM EXPLORING PATTERNS IN CHEMISTRY • 4.08–1 What Makes A Good Conductor? Topic Chemical bonding Introduction An electrical current is the free movement of electrons. If a material allows the passage of electrons through it, it is a conductor. The way in which the atoms are combined within the material (its bond type) determines whether or not the material will allow the flow of electrons. Elements classified as metals lose electrons easily. The electrons in a metallic element are free to move along all the atoms in the metal, and these materials are good electrical conductors. The atoms are held together by attractive forces between the nuclei and these freely moving electrons. These substances are said to have metallic bonds. Elements classified as non-metals and compounds composed of non-metallic elements are held together by covalent bonds. Atoms in these substances share one or more specific pairs of electrons. Their electrons are localized (they cannot move freely throughout the substance), and these materials do not therefore conduct electricity. A few elements exhibit the properties of both metals and non-metals. They are called semi-metals or metalloids. When a metal and non-metal element combine to form a compound, a different type of bonding takes place. Atoms of the metal lose electrons and atoms of the non-metal gain electrons, forming charged particles called ions. The metals form positive ions and the non-metals form negative ions. The oppositely charged ions in the compound attract each other. This is called ionic bonding. Although the compound is made up of charged particles, the numbers of negative and positive particles are always equal and the compound is electrically neutral. In its solid form, the substance’s charged particles are not free to move around, so solid ionic compounds do not conduct electricity. However, when these compounds are melted or dissolved in water, the charged particles (ions) are separated and free to move about. We say they are dissociated. In solution these compounds are electrical conductors. Substances that conduct electricity in solution are called electrolytes and those that do not are called non-electrolytes. In this experiment you will test the conductivity of some solids and solutions, and relate your results to the bond types of the samples tested. Time required 45 minutes © Diagram Visual Information Ltd. Published by Facts On File, Inc. All electronic storage, reproduction, or transmittal is copyright protected by the publisher. 4.08–2 • EXPLORING PATTERNS IN CHEMISTRY CHEMISTRY EXPERIMENTS ON FILETM Materials conductivity apparatus (3 pieces of insulated copper wire, 6V flashlight bulb, socket for bulb, 6V DC lantern battery) aluminum foil Mylar® (from helium balloon) soapless steel wool glass rod wooden stick paper clip charcoal stick graphite pencil “lead” plastic ruler small piece of rubber tubing small piece of stripped copper wire 1 tbls table salt 1 tbls baking soda 1 tbls granulated sugar 1 liter water small saucepan stove or hotplate Safety note Take care when heating and testing solutions. Procedure Part A: Metallic and covalent bonds 1. Set up the conductivity apparatus as shown in the diagram below. 2. Test the conductivity of the solid substances listed in data table A on the next page. To do this, touch both wires of your testing device to each sample. If the bulb lights up, the sample is a conductor. Write “yes” or “no” in the column labelled “Does it conduct?” 3. When you have tested all the substances, go over your results and write whether each substance exhibits the properties of a metal or a non-metal. Write down what bond type you think each has. battery in holder light bulb 6V DC battery sample © Diagram Visual Information Ltd. connecting wire crocodile clip Published by Facts On File, Inc. All electronic storage, reproduction, or transmittal is copyright protected by the publisher. CHEMISTRY EXPERIMENTS ON FILETM EXPLORING PATTERNS IN CHEMISTRY • 4.08–3 DATA Substance (elements it is composed of) TABLE A Does it conduct? Exhibits properties of metal or non-metal? Bond type (metallic or covalent) glass rod (silicon, oxygen) aluminum foil steel wool Mylar® (a polyester – carbon, oxygen, hydrogen) paper clip (nickel, iron) plastic ruler (polypropylene – carbon, hydrogen) rubber tubing (butadiene – carbon, hydrogen) copper wire charcoal stick (carbon) wooden stick (cellulose) graphite pencil ‘lead’ (carbon) Part B: Electrolytes and non-electrolytes 1. Set up the conductivity apparatus as in part A. 2. For each of the substances listed in data table B, test a solid sample for conductivity and note the results. 3. Put 1 tablespoon of sugar into about 250 ml of water in a small saucepan. Bring the solution to the boil, stirring to be sure the sugar is dissolved. 4. Test the solution for conductivity and note the results in data table B. 5. Repeat stages 3 and 4 for the samples of table salt and baking soda. 6. Based on your results, decide whether each of these compounds is an electrolyte or a non-electrolyte and if it contains ionic or covalent bonds. DATA Substance (what is it composed of) Conduct as solid? TABLE Conductivity in solution B Electrolyte or non-electrlyte Bond type sugar (dextrose – carbon, hydrogen, oxygen) salt (sodium chloride) baking soda (sodium hydrogencarbonate) © Diagram Visual Information Ltd. Published by Facts On File, Inc. All electronic storage, reproduction, or transmittal is copyright protected by the publisher. 4.08–4 • EXPLORING PATTERNS IN CHEMISTRY CHEMISTRY EXPERIMENTS ON FILETM Analysis 1. Which of the solids in part A were good conductors? Which were poor conductors? Which of the samples exhibited metallic properties? 2. Which bond type did the most of the samples with metallic properties have? 3. Can you explain the results for carbon? 4. The wires of your conductivity device are copper coated with rubber. Explain the role of the copper and the rubber in your device. 5. Did any of the solid substances in part B conduct electricity? If yes, why? If not, why not? 6. Which of the solutions in part B conduct electricity? Explain why. Want to know more? Click here to view our findings. © Diagram Visual Information Ltd. Published by Facts On File, Inc. All electronic storage, reproduction, or transmittal is copyright protected by the publisher. 4.08 What Makes A Good Conductor? 1. The aluminum foil, steel wool, paper clip, copper, charcoal stick, and graphite pencil were good conductors and displayed metallic properties. The glass rod, Mylar®, plastic, rubber, and wood were poor conductors. 2. Most of the samples with metallic properties contained metallic bonds. The exception was carbon. 3. Carbon is a non-metal and therefore has covalent bonds. However, in this experiment it exhibited metallic properties. Carbon in the form of graphite is the only non-metallic element that conducts electricity. This can be explained by looking at its structure (see diagram below): strong carbon bonds weaker bond between layers carbon atom As you can see, graphite is composed of sheets of hexagons of carbon atoms joined by covalent bonds. Between each sheet, there are weaker (pi) bonds along which the electrons are free to move. It is the movement of electrons between the layers that explains this unusual property of graphite. 4. Copper contains metallic bonds and so conducts electricity. Rubber contains covalent bonds and so does not conduct electricity. It protects the user from getting an electric shock. 5. None of the solids in part B conducted electricity because none contained charged particles that were free to move. Although salt and baking soda contain ions, these ions were locked in position within the ionic lattice and could not move freely. 6. Solutions of salt and of baking soda conducted electricity. They are electrolytes. Both compounds contain ionic bonds. Sugar contains covalent bonds (it is composed entirely of nonmetals) and so did not conduct electricity even when dissolved in water. It is a non-electrolyte.