Chemistry: Chemical Reaction Rates Lab Reaction rate is a
advertisement

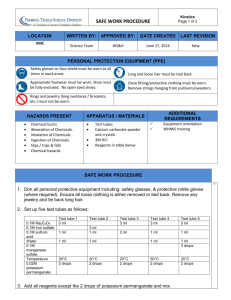
Name ________________________________________ Pd ____ Date ________________________ Chemistry: Chemical Reaction Rates Lab Reaction rate is a measure of how fast or how slow a chemical reaction occurs. In this lab you will investigate several variables to learn how they affect reaction rates. Ultimately, you want to relate your findings to the Collision theory of chemical reactions. Do your data support this model? Research Questions 1. How does the concentration of the reactants affect the reaction rate? 2. How does the temperature of the reactants affect the reaction rate? 3. How does the surface area of solid reactants affect the reaction rate? 4. How does the presence of a catalyst affect the reaction rate? Read through the experiment below and create the necessary data tables to record your qualitative and quantitative data. Your data tables must be attached to this lab report. Potassium Permanganate + Oxalic Acid Reaction 1. Put 4.0 mL of 0.03M potassium permanganate (KMnO4) in one test tube and 4.0 mL of 0.6M oxalic acid (H2C2O4) in another test tube. 2. Pour the oxalic acid into the potassium permanganate. Mix the solution by putting your thumb over the mouth of the test tube and inverting the test tube twice. 3. Record how long it takes for the color to change from purple to gold. 4. Measure the initial and final temperature of the solution at the start and end of the reaction. 5. Repeat steps #1 - #4 to run two additional trials. RQ #1 - Concentration Effect Perform the same reaction with concentration of the reactants diluted by one-half. 1. Put 2.0 mL of 0.03M potassium permanganate (KMnO4) and 6.0 mL of distilled water in one test tube, and 2.0 mL of 0.6M oxalic acid (H2C2O4) in another test tube. 2. Pour the oxalic acid into the potassium permanganate. Mix the solution by putting your thumb over the mouth of the test tube and inverting the test tube twice. 3. Measure how long it takes for the color to change from purple to gold. 4. Measure the initial and final temperature of the solution at the start and end of the reaction. 5. Repeat steps #1 - #4 to run two additional trials. RQ #2 - Temperature Effect Test tubes with 4.0 mL of 0.03M potassium permanganate (KMnO4) and 4.0 mL of 0.6M oxalic acid (H2C2O4) have been prepared and placed in a hot water bath. 1. Use a test tube holder to remove one test tube of each chemical. 2. Pour the oxalic acid into the potassium permanganate. 3. Measure how long it takes for the color to change from purple to gold. 4. Measure the initial and final temperature of the solution at the start and end of the reaction. 5. Repeat steps #1 - #4 to run two additional trials. Hydrochloric Acid + Zinc Reaction 1. Put 50 mL of 6.0M hydrochloric acid (HCl) into a 100 mL beaker. 2. Place the beaker of hydrochloric acid on the scale and zero out the scale. 3. Gently drop the three pieces of zinc in the hydrochloric acid and record the initial mass of zinc. 4. Record the mass of the zinc every 30 seconds for 3 minutes. RQ #3 – Surface Area Effect 1. Put 50 mL of 6.0M hydrochloric acid (HCl) into a 100 mL beaker. 2. Measure out the same amount of powdered zinc as you used in the first reaction. 3. Place the beaker of hydrochloric acid on the scale and zero out the scale. 4. Pour the powdered zinc into the hydrochloric acid and record the initial mass of zinc. 5. Record the mass of the zinc every 30 seconds for 3 minutes. Hydrogen Peroxide Decomposition Reaction 1. Put 80 mL of 3% hydrogen peroxide (the other 97% is water) into a 150 mL beaker. 2. Look for signs of a chemical reaction. Record your observations after 1 minute. 3. Keep the beaker and its contents for the next reaction. RQ #4 – Catalyst Effect Manganese oxide is a catalyst for the hydrogen peroxide decomposition reaction. Catalysts are not reactants or products. They facilitate the reaction, but do not undergo any chemical change during the reaction. 1. Measure out 0.5 g of manganese(IV) oxide (MnO2). 2. Pour the manganese(IV) oxide into the hydrogen peroxide. 3. Look for signs of a chemical reaction. Record your observations after 1 minute. Data Analysis 1. How could you tell a reaction was taking place between the potassium permanganate and the oxalic acid reaction? 2. How could you tell when the reaction was finished? 3. Calculate the average reaction time for the three potassium permanganate + oxalic acid reactions. 4. What type of reaction is the hydrochloric acid + zinc reaction? 5. Write the balanced chemical formula equation for this reaction. 6. How could you tell a reaction was taking place? 7. Why did the mass change as the reaction continued? 8. Calculate the average change in mass per minute for the two hydrochloric + zinc reactions? 9. Write the balanced chemical formula equation for the decomposition of hydrogen peroxide? 10. How could you tell a reaction was taking place? Conclusions 11. What effect does concentration have on the reaction rate? Use your data as specific examples that support your answer. 12. Use the Collision theory to explain why concentration has this effect on reaction rates. 13. What effect does temperature have on the reaction rate? Use your data as specific examples that support your answer. 14. Use the Collision theory to explain why temperature has this effect on reaction rates. 15. What effect does surface area of a solid reactant have on the reaction rate? Use your data as specific examples that support your answer. 16. Use the Collision theory to explain why the surface area of a solid reactant has this effect on reaction rates. 17. What effect does a catalyst have on reaction rate? Use your data as specific examples that support your answer. 18. Use the Collision theory to explain why the catalyst has the effect on reaction rates.