Cytotoxic, Antineoplastic and Other Hazardous Drugs
advertisement

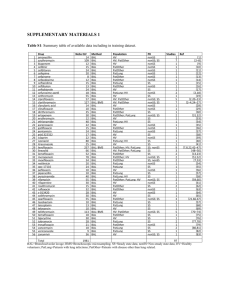
San Diego State University Environmental Health and Safety Department Cytotoxic/Antineoplastic and Other Hazardous Drugs National Institute of Occupational Safety & Health - List of Antineoplastic and Other Hazardous Drugs in Healthcare Settings 2012 Table 1. Sample List of Drugs that Should be Handled as Hazardous Drug Source Acitretin Aldesleukin Ambrisentan 7 4,5 7 Alefacept 6 Alitretinoin 3,4,5 Altretamine Amsacrine Anastrozole Arsenic trioxide Asparaginase Azacitidine Azathioprine Bacillus Calmette-Guerin (BCG)† Bendamustine HCl Bexarotene Bicalutamide Bleomycin Bortezomib Bosentan 1,2,3,4,5 3,5 1,5 1,2,3,4,5 1,2,3,4,5 3,5 2,3,5 1,2,4 Busulfan Cabergoline 1,2,3,4,5 7 Capecitabine Carbamazepine 1,2,3,4,5 7 Carboplatin Carmustine Cetrorelix acetate 1,2,3,4,5 1,2,3,4,5 5 Chlorambucil Chloramphenicol Choriogonadotropin alfa 1,2,3,4,5 1,5 5 7 2,3,4,5 1,5 1,2,3,4,5 6 6 AHFS Pharmacologic-therapeutic classification 88:04 Vitamin A 10:00 Antineoplastic agents 24:12.92 Vasodilating agents, miscellaneous 84:92 Skin and mucous membrane agents, miscellaneous 84:92 Skin and mucous membrane agents, miscellaneous 10:00 Antineoplastic agents Not in AHFS (antineoplastic agent) 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 92:44 Immunosuppressant agents 80:12 Vaccines 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 24:12.92 Vasodilating agents, miscellaneous 10:00 Antineoplastic agents 28:36.20.04 Ergot-derivative dopamine receptor agonists 10:00 Antineoplastic agents 28:12.92 Anticonvulsants, miscellaneous 10:00 Antineoplastic agents 10:00 Antineoplastic agents 92:40 Gonadotropin-releasing hormone antagonists 10:00 Antineoplastic agents 8:12.08 Chloramphenicols 68:18 Gonadotropins Drug Source Cidofovir ,5 Cisplatin Cladribine Clofarabine Clonazepam Colchicine Cyclophosphamide Cyclosporin Cytarabine Dacarbazine Dactinomycin Dasatinib Daunorubicin HCl Decitibine Degarelix Denileukin Diethylstilbestrol ,2,3,4,5 ,2,3,4,5 Dinoprostone Docetaxel Doxorubicin Dronedarone HCl Dutasteride Entecavir Epirubicin Ergonovine/methylergonovi ne Estradiol Estramustine phosphate Estrogen-progestin combinations Estrogens, conjugated Estrogens, esterified 8 Drug Estrone Estropipate Etoposide Everolimus ,2,3,4,5 ,2,3,4,5 ,2,3,4,5 ,2,3,4,5 ,2,3,4,5 ,4,5 ,2,3,4,5 ,2,3,4,5 ,2,3,4,5 ,5 ,2,3,4,5 ource 5 5 1,2,3,4,5 7 AHFS Pharmacologictherapeutic classification :18.32 Nucleosides and nucleotides 0:00 Antineoplastic agents 0:00 Antineoplastic agents 0:00 Antineoplastic agents 8:12.08 Benzodiazepines 2:16 Antigout agents 0:00 Antineoplastic agents 2:44 Immunosuppressive agents 0:00 Antineoplastic agents 0:00 Antineoplastic agents 0:00 Antineoplastic agents 0:00 Antineoplastic agents 0:00 Antineoplastic agents 0:00 Antineoplastic agents 0:00 Antineoplastic agents 0:00 Antineoplastic agents ot in AHFS (nonsteroidal synthetic estrogen) 6:00 Oxytocics 0:00 Antineoplastic agents 0:00 Antineoplastic agents 4:04.04 Antiarrythmics 2:08 5-alpha reductase inhibitors :18.32 Nucleosides and nucleotides 0:00 Antineoplastic agents 6:00 Oxytocics 8:16.04 Estrogens 0:00 Antineoplastic agents 8:12 Contraceptives 8:16.04 Estrogens 8:16.04 Estrogens HFS Pharmacologic-therapeutic classification 68:16.04 Estrogens 68:16.04 Estrogens 10:00 Antineoplastic agents 10:00 Antineoplastic agents Drug Source Exemestane Finasteride 1,5 1,3,5 Floxuridine Fludarabine Fluorouracil Fluoxymesterone Flutamide Fulvestrant Ganciclovir 1,2,3,4,5 1,2,3,4,5 1,2,3,4,5 5 1,2,5 5 1,2,3,4,5 Ganirelix acetate 5 Gemcitabine Gemtuzumab ozogamicin Gonadotropin, chorionic Goserelin Hydroxyurea Idarubicin Ifosfamide Imatinib mesylate Irinotecan HCl Ixabepilone Leflunomide 1,2,3,4,5 1,3,4,5 5 1,2,5 1,2,3,4,5 1,2,3,4,5 1,2,3,4,5 1,3,4,5 1,2,3,4,5 7 3,5 Lenalidomide 6 Letrozole Leuprolide acetate Lomustine Mechlorethamine Medroxyprogesterone acetate Megestrol 9 Drug 1,5 1,2,5 1,2,3,4,5 1,2,3,4,5 6 Melphalan Menotropins Mercaptopurine Methotrexate Methyltestosterone Mifepristone Mitomycin Mitotane 1,2,3,4,5 5 1,2,3,4,5 1,2,3,4,5 5 5 1,2,3,4,5 1,4,5 1,5 Source AHFS Pharmacologictherapeutic classification 10:00 Antineoplastic agents 92:08 5-alpha reductase inhibitors 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 68:08 Androgens 10:00 Antineoplastic agents 10:00 Antineoplastic agents 8:18.32 Nucleosides and nucleotides 2:40 Gonadotropin-releasing hormone antagonists 10:00 Antineoplastic agents 10:00 Antineoplastic agents 68:18 Gonadotropins 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 92:36 Disease-modifying antirheumatic agents 92:20 Biologic response modifiers 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 68:32 Progestins 10:00 Antineoplastic agents AHFS Pharmacologictherapeutic classification 10:00 Antineoplastic agents 68:18 Gonadotropins 10:00 Antineoplastic agents 10:00 Antineoplastic agents 68:08 Androgens 76:00 Oxytocics 10:00 Antineoplastic agents 10:00 Antineoplastic agents Mitoxantrone HCl Mycophenolate mofetil 1,2,3,4,5 1,3,5 Drug Source Mycophenolic acid 7 Nafarelin Nelarabine Nilotinib Nilutamide Oxaliplatin Oxcarbazepine 5 6 7 1,5 1,3,4,5 7 Oxytocin Paclitaxel Palifermin 5 1,2,3,4,5 6 Paroxetine** 6, 7 Pazopanib HCl Pegaspargase Pemetrexed Pentamidine isethionate 7 1,2,3,4,5 6 1,2,3,5 Pentetate calcium trisodium†† Pentostatin Phenoxybenzamine HCl 6 Pipobroman 3,5 Plerixafor Podofilox 7 5 10 Drug Source Podophyllum resin 5 Pralatrexate Procarbazine Progesterone Progestins Raloxifene 7 1,2,3,4,5 5 5 5 1,2,3,4,5 7 10:00 Antineoplastic agents 92:44 Immunosuppressive agents AHFS Pharmacologictherapeutic classification 92:44 Immunosuppressive agents 68:18 Gonadotropins 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 28:12.92 Anticonvulsants, miscellaneous 76:00 Oxytocics 10:00 Antineoplastic agents 84:16 Cell stimulants and proliferants 28:16.04.20 Selective serotonin uptake inhibitors 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 8:30.92 Antiprotozoals, miscellaneous Not in AHFS 10:00 Antineoplastic agents 12:16.04.04 Non-selective alpha-adrenergic blocking agents Not in AHFS (antineoplastic agent) 20:16 Hematopoietic agents 84:92 Miscellaneous skin and mucous membrane agents (mitotic inhibitor) AHFS Pharmacologictherapeutic classification 84:92 Skin and mucous membrane agents, miscellaneous 10:00 Antineoplastic agents 10:00 Antineoplastic agents 68:32 Progestins 68:12 Contraceptives 68:16.12 Estrogen agonists- Rasagiline mesylate Ribavirin 6 1,2,5 Drug Source Risperidone 6 Romidepsin Sirolimus 7 6 Sorafenib Streptozocin Sunitinib malate Tacrolimus 6 1,2,3,4,5 6 1,5 Tamoxifen Televancin Temozolomide Temsirolimus Teniposide Testolactone Testosterone Tetracycline HCl Thalidomide 1,2,5 7 3,4,5 7 1,2,3,4,5 5 5 7 1,3,5 Thioguanine Thiotepa Topotecan Toremifene citrate Tretinoin 1,2,3,4,5 1,2,3,4,5 1,2,3,4,5 1,5 1,2,3,5 Trifluridine Triptorelin Uracil mustard 1,2,5 5 3,5 11 Drug Source Valganciclovir 1,3,5 Valproic acid/ divalproex Na 7 Valrubicin Vidarabine Vigabatrin 1,2,3,5 1,2,5 7 Vinblastine sulfate 1,2,3,4,5 antagonists 28:36 Antiparkinsonian agents 8:18.32 Nucleosides and nucleotides AHFS Pharmacologictherapeutic classification 28:16.08.04 Atypical antipsychotics 10:00 Antineoplastic agents 92:44 Immunosuppressive agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 92:44 Immunosuppressive agents 10:00 Antineoplastic agents 8:12.28.16 Glycopeptides 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 68:08 Androgens 8:12.24 Tetracyclines 92:20 Biologic response modifiers 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 84:16 Cell stimulants and proliferants 52:04.20 Antivirals 10:00 Antineoplastic agents Not in AHFS (antineoplastic agent) AHFS Pharmacologictherapeutic classification 8:18.32 Nucleosides and nucleotides 28:12.92 Anticonvulsants, miscellaneous 10:00 Antineoplastic agents Not in AHFS 28:12.92 Anticonvulsants, miscellaneous 10:00 Antineoplastic agents Vincristine sulfate Vinorelbine tartrate Vorinostat Zidovudine Ziprasidone HCl 1,2,3,4,5 1,2,3,4,5 6 1,2,5 7 Drug Source Zoledronic acid 7 Zonisamide 6 10:00 Antineoplastic agents 10:00 Antineoplastic agents 10:00 Antineoplastic agents 8:18:08 Antiretroviral agents 28:16.08.04 Atypical antipsychotics AHFS Pharmacologictherapeutic classification 92:24 Bone resorption inhibitors 28:12.92 Anticonvulsants, miscellaneous *These lists of hazardous drugs were used with the permission of the institutions that provided them and were adapted for use by NIOSH. The sample lists are intended to guide health care providers in diverse practice settings and should not be construed as complete representations of all of the hazardous drugs used at the referenced institutions. Some drugs defined as hazardous may not pose a significant risk of direct occupational exposure because of their dosage formulation (for example, intact medications such as coated tablets or capsules that are administered to patients without modifying the formulation). However, they may pose a risk if solid drug formulations are altered outside a ventilated cabinet (for example, if tablets are crushed or dissolved, or if capsules are pierced or opened). BCG preparation should be done using aseptic techniques. To avoid cross-contamination, parenteral drugs should not be prepared in areas where BCG has been prepared. A separate area for the preparation of BCG suspension is recommended. All equipment, supplies, and receptacles in contact with BCG should be handled and disposed of as biohazardous. If preparation cannot be performed in a containment device, then respiratory protection, gloves and a gown should be worn to avoid inhalation or contact with BCG organisms. 1. The NIH Clinical Center, Bethesda, MD (Revised 8/2002). The NIH Health Clinical Center Hazardous Drug (HD) List is part of the NIH Clinical Center’s hazard communication program. It was developed in compliance with the OSHA hazard communication standard [29 CFR 1910.1200] as it applies to hazardous drugs used in the workplace. The list is continually revised and represents the diversity of medical practice at the NIH Clinical Center; however, its content does not reflect an exhaustive review of all FDA-approved medications that may be considered hazardous, and it is not intended for use outside the NIH. 2. The Johns Hopkins Hospital, Baltimore, MD (Revised 9/2002). 3. The Northside Hospital, Atlanta, GA (Revised 8/2002). 4. The University of Michigan Hospitals and Health Centers, Ann Arbor, MI (Revised 2/2003) 5. This sample listing of hazardous drugs was compiled by the Pharmaceutical Research and Manufacturers of America (PhRMA) using information from the AHFS DI monographs published by ASHP in selected AHFS Pharmacologic-Therapeutic Classification categories [ASHP/AHFS DI 2003] and applying the definition for hazardous drugs. The list also includes drugs from other sources that satisfy the definition for hazardous drugs [PDR 2004; Sweetman 2002; Shepard 2001; Schardein 2000; REPROTOX 2003]. Newly approved drugs that have structures or toxicological profiles that mimic the drugs on this list should also be included. This list was revised in June 2004. 6. NIOSH addition 2010 updated using ASHP/AHFS DI 2010. 7. NIOSH addition 2012 updated using ASHP/AHFS DI 2011. **2010, Paroxetine HCl; 2012, Paroxetine mesylate ††Refers to non-radio-labeled formulation only. †