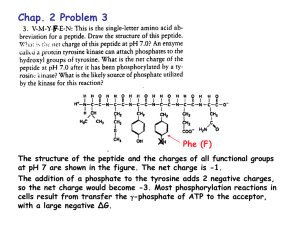
Polyprotic Acid Buffer Calculations: pH & Equilibrium
advertisement

Buffer Calculations for Polyprotic Acids
A polyprotic acid can form buffer solutions in presence of its conjugate base. For
example, phosphoric acid can form a buffer when combined with its conjugate base
(dihydrogen phosphate).
H3PO4 = H+ + H2PO4This buffer operates in the range:
ka1 = 1.1 x 10-2
pH = pka + 1 = 0.96 – 2.96
Also, another buffer which is commonly used is the dihydrogen phosphate/hydrogen
phosphate buffer.
H2PO4- = H+ + HPO42-
ka2 = 7.5 x 10-8
This buffer operates in the range from 6.1 to 8.1
A third buffer can be prepared by mixing hydrogen phosphate with orthophosphate as
the following equilibrium suggests:
HPO42- = H+ + PO43-
ka3 = 4.8 x 10-13
This buffer system operates in the pH range from 11.3 to 13.3
The same can be said about carbonic acid/bicarbonate where
H2CO3 = H+ + HCO3-
ka1 = 4.3 x 10-7
This buffer operates in the pH range from 5.4 to 7.4; while a more familiar buffer
is composed of carbonate and bicarbonate according to the equilibrium:
HCO3- = H+ + CO32-
ka2 = 4.8 x 10-11
The pH range of the buffer is 9.3 to 11.3.
Polyprotic acids and their salts are handy materials which can be used to prepare
buffer solutions of desired pH working ranges. This is true due to the wide variety of
their acid dissociation constants.
Example
Find the ratio of [H2PO4-]/HPO42-] if the pH of the solution containing a mixture of
both substances is 7.4. ka2 = 7.5x10-8
Solution
The equilibrium equation combining the two species is:
H2PO4- = H+ + HPO42Ka2 = [H ][HPO42-]/[H2PO4-]
ka2 = 7.5 x 10-8
+
[H+] = 10-7.4 = 4x10-8 M
7.5x10-8 = 4x10-8 [HPO42-]/[H2PO4-]
[HPO42-]/[H2PO4-] = 1.9
Fractions of Dissociating Species at a Given pH
Consider the situation where, for example, 0.1 mol of H3PO4 is dissolved in 1 L of
solution.
H3PO4 = H+ + H2PO4H2PO4- = H+ + HPO42HPO42- = H+ + PO43-
ka1 = 1.1 x 10-2
ka2 = 7.5 x 10-8
ka3 = 4.8 x 10-13
Some of the acid will remain undissociated (H3PO4), some will be converted to
H2PO4-, HPO42- and PO43- where we have, from mass balance:
CH3PO4 = [H3PO4] + [H2PO4-] + [HPO42-] + [PO43-]
We can write the fractions of each species in solution as
α0 = [H3PO4]/CH3PO4
α1 = [H2PO4-]/CH3PO4
α2 = [HPO42-]/CH3PO4
α3 = [PO43-]/CH3PO4
α0 + α1 + α2 + α3 = 1 ( total value of all fractions sum up to unity). The value of each
fraction depends on pH of solution. At low pH dissociation is suppressed and most
species will be in the form of H3PO4 while high pH values will result in greater
amounts converted to PO43-. Setting up a relation of these species as a function of [H+]
is straightforward using the equilibrium constant relations. Let us try finding α0 where
α0 is a function of undissociated acid. The point is to substitute all fractions by their
equivalent as a function of undissociated acid.
Ka1 = [H2PO4-][H+]/[H3PO4]
Therefore we have
[H2PO4-] = ka1 [H3PO4]/ [H+]
ka2 = [HPO42-][H+]/[H2PO-]
Multiplying ka2 time ka1 and rearranging we get:
[HPO42-] = ka1ka2 [H3PO4]/[H+]2
ka3 = [PO43-][H+]/[HPO42-]
Multiplying ka1 times ka2 times ka3 and rearranging we get:
[PO3-] = ka1ka2ka3 [H3PO4]/[H+]3
But we have:
CH3PO4 = [H3PO4] + [H2PO4-] + [HPO42-] + [PO43-]
Substitution for all species from above gives:
CH3PO4 = [H3PO4] + ka1 [H3PO4]/ [H+] + ka1ka2 [H3PO4]/[H+]2 + ka1ka2ka3
[H3PO4]/[H+]3
CH3PO4 = [H3PO4] {1 + ka1 / [H+] + ka1ka2 /[H+]2 + ka1ka2ka3 /[H+]3}
[H3PO4]/CH3PO4 = 1/ {1 + ka1 / [H+] + ka1ka2 /[H+]2 + ka1ka2ka3 /[H+]3}
αo = [H+]3 / ([H+]3 + ka1[H+]2 + ka1ka2[H+] + ka1ka2ka3)
Similar derivations for other fractions results in:
α1 = ka1[Η+]2 / ([H+]3 + ka1[H+]2 + ka1ka2[H+] + ka1ka2ka3)
α2 = ka1ka2 [H+] / ([H+]3 + ka1[H+]2 + ka1ka2[H+] + ka1ka2ka3)
α3 = ka1ka2ka3 / ([H+]3 + ka1[H+]2 + ka1ka2[H+] + ka1ka2ka3)
Example
Calculate the equilibrium concentrations of the different species in a 0.10 M
phosphoric acid solution at pH 3.00.
Solution
The [H+] = 10-3.00 = 1.0x10-3 M
Substitution in the relation for αo gives
αo = [H+]3 / ([H+]3 + ka1[H+]2 + ka1ka2[H+] + ka1ka2ka3)
αo = (1.0x10-3)3/{(1.0x10-3)3 + 1.1x10-2 (1.0x10-3)2 + 1.1x10-2 * 7.5x10-8 (1.0x10-3) +
1.1x10-2 * 7.5x10-8 * 4.8 * 10-13}
αo = 8.2x10-2
α0 = [H3PO4]/CH3PO4
8.2x10-2 = [H3PO4]/0.10
[H3PO4] = 8.3x10-3 M
Similarly, α1 = 0.92,
α1 = [H2PO4-]/CH3PO4
0.92 = [H2PO4-]/0.10
[H2PO4-] = 9.2x10-2 M
Other fractions are calculated in the same manner.
pH Calculations for Salts of Polyprotic Acids
Two types of salts exist for polyprotic acids. These include:
1. Unprotonated salts
These are salts which are proton free which means they are not associated with any
protons. Examples are: Na3PO4 and Na2CO3. Calculation of pH for solutions of such
salts is straightforward and follows the same scheme described earlier for salts of
monoprotic acids.
Example
Find the pH of a 0.10 M Na3PO4 solution.
Solution
We have the following equilibrium in water
PO43- + H2O = HPO42- + OHThe equilibrium constant which corresponds to this equilibrium is kb where:
Kb = kw/ka3
We used ka3 since it is the equilibrium constant describing relation between PO43and HPO42-. However, in any equilibrium involving salts look at the highest
charge on any anion to find which ka to use.
Kb = 10-14/4.8x10-13
Kb = 0.020
Before Equilibrium
Equation
0.10
PO4 + H2O
0
HPO4-
0
OH-
At Equilibrium
0.10 –x
x
x
3-
Kb = x * x/0.10 – x
Assume 0.10 >> x
0.02 = x2/0.10
x = 0.045
Relative error = (0.045/0.10) x 100 = 45%
Therefore, assumption is invalid and we have to use the quadratic equation. If we
solve the quadratic equation we get:
X = 0.036
Therefore, [OH-] = 0.036 M
pOH = 1.44
pH = 14 – 1.44 = 12.56
2. Protonated Salts
These are usually amphoteric salts which react as acids and bases. For example,
NaH2PO4 in water would show the following equilibria:
H2PO4- = H+ + HPO42H2PO4- + H2O = OH- + H3PO4
H2O = H+ + OH[H+]solution = [H+]H2PO4- + [H+]H2O – [OH-]H2PO4[H+]solution = [HPO42-] + [OH-] – [H3PO4]
Now make all terms as functions in either H+ or H2PO4-, then we have:
[H+] = {ka2 [H2PO4-]/[H+]} + kw/[H+] –{[H2PO4-][H+]/ka1}
Rearrangement gives
[H+] = {(ka1kw + ka1ka2[H2PO4-])/(ka1 + [H2PO4-]}1/2
At high salt concentration and low ka1 this relation may be approximated to:
[H+] = {ka1ka2}1/2
Where the pH will be independent on salt concentration but only on the equilibrium
constants.
Example
Find the pH of a 0.10 M NaHCO3 solution. ka1 = 4.3 x 10-7, ka2 = 4.8 x 10-11
Solution
HCO3- = H+ + CO32HCO3- + H2O = OH- + H2CO3
H2O = H+ + OH-
[H+] = {(ka1kw + ka1ka2[H2PO4-])/(ka1 + [H2PO4-]}1/2
[H+] = {(4.3x10-7 * 10-14 + 4.3x10-7 * 4.8x10-11 * 0.10)/(4.3x10-7 + 0.10)}1/2
[H+] = 4.5x10-9 M
pH = 8.34
The same result can be obtained if we use [H+] = {ka1ka2}1/2
[H+] = {4.3x10-7 * 4.8x10-11}1/2 = 4.5x10-9 M
This is since the salt concentration is high enough. Now look at the following
example and compare:
Example
Find the pH of a 1.0x10-4 M NaHCO3 solution. ka1 = 4.3 x 10-7, ka2 = 4.8 x 10-11
Solution
[H+] = {(4.3x10-7 * 10-14 + 4.3x10-7 * 4.8x10-11 * 1.0x10-4)/(4.3x10-7 + 1.0x10-4)}1/2
[H+] = 7.97x10-9 M
pH = 8.10
Substitution in the relation [H+] = {ka1ka2}1/2 will give
[H+] = {4.3x10-7 * 4.8x10-11}1/2 = 4.5x10-9 M, which is incorrect
You can see the difference between the two results.