Virtual-experiment
advertisement

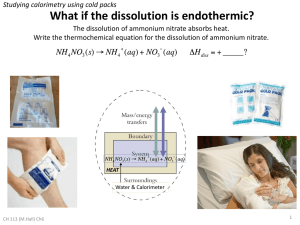
Virtual Experiment Worksheet Names: Experiment – Specific Heat Capacity of Copper Part 1, Calculating the calorimeter constant: Start with 50.0 g iron and 50.0 g water. What is the starting temperature of the water? ______ What is the final temperature? ______ Write out your heat equation including the calorimeter constant, showing just the variables and all the proper subscript labels (e.g., water, iron, calorimeter, final, initial). Remember, 0 = qmetal + qwater + qcalorimeter. qcalorimeter = Ccalorimeter(Tfinal-Tinitial) What is Ccalorimeter? Show proper units. ________ Change the mass of the iron and the mass of the water. Perform the same experiment and calculate Ccalorimeter. What do you get this time? Part 2: Now calculate the specific heat capacity of copper. Start with a copper mass of 50.0 g and a mass of water at 50.0 g. Record the following value: Starting Water Temperature: _____ Final Water Temperature: ______ Write out your heat equation (using only variables) including the heat transferred to the calorimeter. Then substitute in your known values. Calculate the specific heat capacity of copper. How does this value compare to the one you found in class? Going Further: Change the mass of copper and water used in the experiment. Mark down the starting temperature. Now write an equation for the final temperature of the water using the specific heat of copper. PREDICT what the final temperature of the water will be BEFORE running the experiment. Now run the experiment and compare your answer. Experiment – Heat of Solution of Ammonium Nitrate Purpose: Given the heat capacity of the calorimeter, determine the heat of solution of ammonium nitrate: NH4NO3 (s) NH4+ (aq) + NO3- (aq) When you write out your heat balance equation this time, remember that the heat produced by the reaction is absorbed by the surrounding water (qsolution) and the calorimeter (qcalorimeter). First, calculate the heat of solution (qsolution). Show your equations and given values (masses of water, ammonium nitrate, final and initial temperatures) Second, calculate the molar enthalpy of solution. This is just (qsolution)/n, where n is the number of moles of ammonium nitrate.