Conductivity and TDS Lab Notes
advertisement

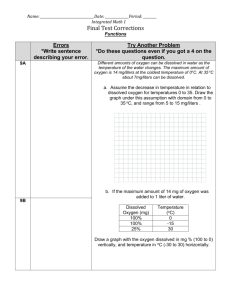
Environmental Chemistry Solution Conductivity and Total Dissolved Solids (TDS) Purpose: - To use a linear calibration curve to determine the concentration of Total Dissolved Solids (TDS) in a sample of stream water. Prelab Notes: Impurities in water samples are usually found in two forms; suspended and dissolved. Suspended impurities can be filtered out and affect the turbidity of the water sample. Dissolved impurities pass right through filter paper and affect the solutions conductivity. TDS or total dissolved solids is a measure of the total concentration of dissolved substances in a water sample. These dissolved substances are usually ionic compounds. Ionic Compounds – Consist of positive and negative ions in a crystal lattice structure. When water is introduced into this structure, the ions break out of it and “dissolve.” A high TDS concentration does not necessarily mean that a sample is “polluted” or dangerous. These ions come from a variety of sources. Sources of Total Dissolved Solids Hard-Water Ions - Ca2+ - Mg2+ - HCO3– Fertilizer in agricultural runoff - NH4+ - NO3– - PO43– - SO42– Urban runoff - Na+ - Cl– Salinity from tidal mixing, minerals, or returned irrigation water - Na+ - K+ - Cl– Acidic rainfall - H+ - NO3– - SO32–, SO42–