Molecular structure of myofibril proteins (Figs 10.5
advertisement

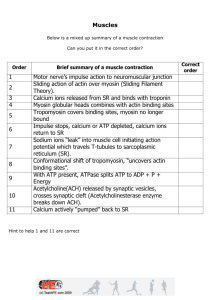
BS277 The Biology of Muscle. The molecular motor of muscle. Learning Outcome. Discuss muscle contraction [development, adaptation to training and fatigue]. Learning Objectives. After this lecture and associated reading you will be able to: [Describe myofibrillar and other key proteins of the muscle fibre]. Discuss the molecular interactions that result in myofilament sliding. Background reading. Jones, D., Round, J. and de Haan, A. (2004) Skeletal muscle; from molecules to movement. (Chapters 1-6) Churchill Livingstone. Martini, F.H. (2009) Fundamentals of Anatomy and Physiology.(8th edition). Chapter 10. Pearsons; Benjamin Cummings. Sweeney, H.L. (1996) Molecular Motors of Eukaryotic Cells Annu. Rev. Physiol. 58: 669-670 et seq. Muscle contraction. Just a special case of actin myosin interactions based on a highly specialised and ordered cytoskeleton containing the ubiquitous microfilaments (actin) and thick filaments of myosin that are specific to muscle (though myosin molecules are ubiquitous). Interaction of actin and myosin orientated along the axis of the muscle cell changes its shape, producing movement provided that the cell is anchored Skeletal muscle differentiation. Uninucleate myoblasts originate in the embryonic somites in week 4. Thyroid hormone, IGF and the withdrawal of other growth factors (FGF, TGF, proliferin) stimulate entry into Go and fusion into myotubes. Some remain as satellite stem cells for repair and growth. Myo D gene family of regulatory proteins coordinate the expression of structural genes. Myofilaments expressed at 9 weeks. Myotube formation ceases prenatally; subsequent increase in muscle bulk depends on hypertrophy, with satellite cells providing nuclei, not hyperplasia (Olsen, 1992). Cytology of skeletal muscle. Muscle fibres (Fig.10.2) are large syncitial cells (up to 100 x 50cm) that may run the entire length of the muscle. They are organised into bundles (within elements of the perimysium) called fascicles (fasciculae). Sarcolemma and T tubules transmit the action potentials that initiate contraction. Myofibrils are the characteristic organelles of skeletal muscle and contain thin myofilaments (~ microfilaments of the cytoskeleton) of actin and thick myofilaments of myosin. Each myofibril comprises multiple (thousands) units of myofilament arrays called sarcomeres, 1.52.5 in length, arranged in series and separated from each other by z-lines. Myofibrils are associated with mitochondria, lipid droplets and glycogen granules. Each myofibril is also surrounded by a fenestrated tube of sarcoplasmic reticulum (SR) that functions as a Ca++ store. Separate sections of SR are associated with each sarcomere and terminally they make contact with the T tubules to form triads (1 t-tubule + 2 sarcomere terminal cisternae). Overview of the structure of the Sarcomere (Figs 10.1-10.3, Eckert; Fig.10.5, Martini). Sarcomeres (~2.5um long) contain; 1. Thick and thin myofilaments. 2. Proteins that maintain their position. 3. Proteins that regulate their interaction. 1. 2. 3. Thin myofilaments principally actin polymers 6nm x 1. Thick myofilaments are myosin bundles, 12nm x 1.6. The arrangement of the myofilaments in the sarcomere is reflected by the striated appearance of the muscle; A (anisotropic – light polarising) band = myosin. Encompassing the central M line, the lighter H zone (wherein there is no overlap with the light myofilaments) and the terminal zones of overlap, generally associated with triads (2 per sarcomere). I (isotropic – not light polarising) band contains non-overlapping parts of actin light myofilaments between A bands and central Z lines (discs) that define the boundaries of the sarcomere and anchor the light myoflilaments. A sarcomere comprises an A band and 2x½ I bands. The myofilaments and the adjacent myofibrils are maintained in their relative positions by structural proteins, titin, nebulin, connectin and desmin. Thin myofilaments of actin, each subunit having a myosin binding site, are associated with a strand of tropomyosin that covers these sites, and troponin that binds them together and also binds Ca++. The arrangement of the thin and thick myofibrils is best appreciated in cross section (Fig 10.3 Eckert). I band. Hexagonal arrays of actin thin myofilaments. A band. Each myosin thick myofilament surrounded by 6 actins; each actin by 3 myosins, except in H zone. Only triangular/hexagonal arrays of myosin thick myofilaments. (see Millman, 1996 for review and how these patterns have been worked out.) Molecular structure of myofibril proteins (Figs 10.5-10.7, Eckert) Myofibrils can be digested into component filaments by homogenisation in Mg++ -ATP and EGTA (Ca chelator) which breaks the cross links. Thin myofilaments (actin). F-(fibrillar, fibrous) actin is a polymer of G-(globular) actin subunits (42kD) arranged in a double helix. The F actin is 6-8nM thick and 1u long and comprises 380 G monomers in frog sartorius. The polymer is stabilised by a cap of -actinin (37kD). Associated regulatory proteins. Tropomyosin. Filaments in grooves in the actin helix. Troponin. A complex of globular proteins associated with tropomyosin and located at 40nm along the actin filament (2 per turn of the helix). Thick myofilaments (myosin). Polymer of myosin, itself a polymer; a homodimer of heavy chains each associated with a variable number of light chains. Each myosin “monomer” is 150nm long comprising a 2nm thick fibrous tail + neck, and a globular head 20 x 4nm that has actin binding and ATPase activity. The tail is 2 polypeptide helices coiled around each other. These polypeptide are not helical in the head region and are associated with a variable number of light chains that determine the enzyme characteristics. In low salt the myosins spontaneously aggregate to form heavy myofilaments that are identical to those found in vivo, 1.5u long and 12nm thick. There are about 300 myosins in the thick filament of the frog sartorius. Structural proteins (Fig 10.3, Martini). Nebulin determines the length of the thin myofilament. Titin stabilises the position of the thick myofilaments and prevents overstretching of the sarcomere. (Trinick, J. and Tskhovrevoba, L; 1999) Titin is attached to actin in the Z lines by connectin Myomesin, an elastic spring in the M-line stabilises thick myofilament lattice (Agarkova and Perriard, 2005). Z-Z intermediate filaments of desmin keep the myofibrils in register Triad proteins and excitation-contraction coupling. With particular reference to triad proteins, describe the steps by which a sarcolemmal action potential promotes the release of Ca++ from the SR and results in cross-bridge cycling. T-tubule action potential causes conformational change in the DHP receptor that opens the Ryanodine receptor Ca++ channel in the adjacent SR membrane, allowing efflux of Ca++ down its concentration gradient. Ca++ is constantly reclaimed into the SR lumen by the SERCA Ca++- ATPase pump. Other key proteins of the muscle fibre (Blake, et al., 2002) Genetic diseases affecting the muscle fibre are common, and many are classified as muscular dystrophies (‘wrongly nourished’). In Duchenne MD, an X-linked recessive condition that affects 1 in 3500 boys, muscle fibres begin to degenerate at an early age (2-5) and the condition is normally fatal before 30. Muscle wasting ultimately results in respiratory failure DMD is characterised by the lack of the protein dystrophin, part of a transmembrane complex that is associated internally with cytoskeletal actin and externally with the endomysium. Its function is unclear, but if any of the components of the complex is defective, membrane damage and poor calcium regulation lead to fibre degeneration, phagocytosis transient regeneration and ultimately fibrosis. There has been a suggestion that it might act like a shock absorber to protect the membrane during muscle contraction when force is transmitted from the myofibrils to the endomysium. The repair of the defect by gene therapy is a current research priority. Agarkova I, and Perriard J.C. (2005) The M-band: an elastic web that crosslinks thick filaments in the center of the sarcomere. Trends Cell Biol. 15 (9): 477-485 Blake,DJ., Weir, A., Newey, SE.,and Davies, KE. (2002) Function and Genetics of Dystrophin and Dystrophin-Related Proteins in Muscle Physiol. Rev. 82, 291-329. Millman, B.M. (1998) The Filament Lattice of Striated Muscle. Physiol. Rev. 78: 359–391 (see CMR for pdf) Olsen, E.N. (1992) Interplay between proliferation and differentiation within the myogenic lineage. Devl. Biol. 154, 261-272. Trinick, J. and Tskhovrevoba, L; (1999) Titin; a molecular control freak. Trends Cell Biol. 9 377-280. BS277.MHS.Nov09 BS277 The Biology of Muscle. The molecular motor of muscle. Learning Outcome. Discuss muscle contraction [development, adaptation to training and fatigue]. Learning Objectives. After this lecture and associated reading you will be able to: [Describe myofibrillar and other key proteins of the muscle fibre]. Discuss the molecular interactions that result in myofilament sliding. Background reading. Jones, D., Round, J. and de Haan, A. (2004) Skeletal muscle; from molecules to movement. (Chapters 1-6) Churchill Livingstone. Martini, F.H. (2009) Fundamentals of Anatomy and Physiology.(8th edition). Chapter 10. Pearsons; Benjamin Cummings. Sweeney, H.L. (1996) Molecular Motors of Eukaryotic Cells Annu. Rev. Physiol. 58: 669-670 et seq. Skeletal muscle differentiation. What embryonic cell type gives rise to skeletal muscle fibres and what molecular signals are involved in their differentiation? How do muscles get bigger? What peptide ‘negative’ growth factor regulates muscle growth? What is the consequence of genetic defects in the structural genes for this factor or its receptor? (Olsen 1992) Cytology of skeletal muscle fibres What feature distinguishes muscle and nerve cell membranes from those of most other somatic cells? Which membrane structures carry the action potential from the sarcolemma deep into the muscle fibre? What are the contractile organelles of muscle cells and what are their serially arranged functional sub-units? What proteins constitute the thick and thin myofilaments? What is the role of the sarcoplasmic reticulum? Overview of the structure of the Sarcomere (Figs.10.1, 10.2, 10.3 Eckert). Sarcomeres (~2.5um long) contain; Thick and thin myofilaments. Proteins that maintain their position. Proteins that regulate their interaction. 1. Thin myofilaments principally actin polymers 6nm x 1. Thick myofilaments are myosin bundles, 12nm x 1.6 (see below). The arrangement of the myofilaments in the sarcomere is reflected by the striated appearance of the muscle; Describe how the disposition of the thin and thick myofilaments gives rise to the characteristic banding pattern of the sarcomere. What are the structural proteins that stabilise the relative positions of the myofibrils? What are the regulatory proteins that are associated with the thin myofilaments? The arrangement of the thin and thick myofibrils is best appreciated in cross section (Fig 10.3 Eckert; Millman, 1998). Describe and explain the patterns of myofilaments that appear in cross sections of different parts of the sarcomere. Molecular structure of myofibril proteins How may the myofibrils be disaggregated into myofilaments? Explain why this method works. Thin myofilaments (actin). F-(fibrillar, fibrous) actin is a polymer of G-(globular) actin subunits (42 kD) arranged in a double helix. The F actin is 6-8 nM thick and 1u long and comprises 380 G monomers in frog sartorius. The polymer is stabilised by a cap of -actinin (37 kD). Associated regulatory proteins. Name the two regulatory proteins that are associated with the thin myofilament and describe their position with respect to the structure of F-actin both in the absence of calcium and when calcium concentrations rise to >10-7M. 1. 2. Thick myofilaments (myosin). Draw an annotated diagram to illustrate the structure of myosin. What happens when a solution of myosin molecules in saline is dialysed against distilled water? Structural proteins (Fig 10.3, Martini). What are the roles of nebulin, titin, and actinin, myomesin and desmin? Biochemistry of contraction. Describe the roles of Ca and ATP in the cross bridge cycle. Outline the 5 steps of the cross-bridge cycle. 1. 2. 3. 4. 5. Triad proteins and excitation-contraction coupling. With particular reference to triad proteins, describe the steps by which a sarcolemmal action potential promotes the release of Ca++ from the SR and results in cross-bridge cycling. Other key proteins of the muscle fibre (Blake, et al., 2002) What is the role of Dystrophin? Agarkova I, and Perriard J.C. (2005) The M-band: an elastic web that crosslinks thick filaments in the center of the sarcomere. Trends Cell Biol. 15 (9): 477-485 Blake,DJ., Weir, A., Newey, SE.,and Davies, KE. (2002) Function and Genetics of Dystrophin and Dystrophin-Related Proteins in Muscle Physiol. Rev. 82, 291-329. Olsen, E.N. (1992) Interplay between proliferation and differentiation within the myogenic lineage. Devl. Biol. 154, 261-272. Millman, B.M. (1998) The Filament Lattice of Striated Muscle. Physiol.Rev. 78: 359–391 (CMR pdf) Trinick, J. and Tskhovrevoba, L; (1999) Titin; a molecular control freak. Trends Cell Biol. 9 377280. BS356#1.MHS.Nov08