Unit # 9 Acids, Bases, and Salts
advertisement

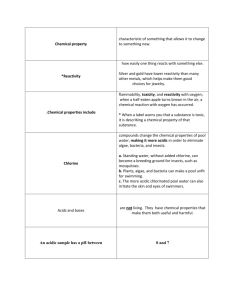
Unit # 9 Acids, Bases, and Salts I. Acid-Base Theories: A. Arrhenius Acids and Bases: Arrhenius acid = an acid that gives _________________________________________ - monoprotic acid = an acid containing ________________________________, example = ______________ - diprotic acid = an acid containing ___________________________________, example = ______________ - triprotic acid = an acid containing __________________ ionizable hydrogens, example = ______________ *** A hydrogen atom is only ionizable if it is _____________________________ ______________________________, otherwise it will not be released in solution. Question: How many of the hydrogen atoms in acetic acid are ionizable? H O H—C—C—O—H H Arrhenius base = a base that gives _________________________________________ Properties of Acids and Bases Acid Base Taste pH range Color change with universal indicator Unit # 9 (Book Chapter 19) Acids, Bases, and Salts -1- B. Bronsted-Lowry acids and Bases (a broader definition): Bronsted-Lowry acid = __________________________________________________ Bronsted-Lowry base = ________________________________________________ – __________________ definition than an Arrhenius base Example: NH3(aq) + H2O(l) NH4+(aq) + OH-(aq) Conjugate Acids and Bases: Example: NH3(aq) + H2O(l) NH4+(aq) + OH-(aq) - consider the reverse reaction of ammonium and hydroxide ion, which substance is the acid? __________ which is the base? __________ … so NH4+ is called a ________________________ and OH- is called a ________________________ - A conjugate acid-base pair is made up of the ____________________________ _______________________________________________________________OR the base and its corresponding conjugate acid in the products – consists of ______ __________________________________________________________________ Additional Example: HCl(g) + H2O(l) H3O+(aq) + Cl-(aq) acid = ___________ base = ___________ conjugate acid = ___________ conjugate base = ___________ amphoteric substances = substances that can _________________________________ _________________________________________________ – like _________________ C. Lewis Acids and Bases: Lewis acid = __________________________________________________________ Lewis base = __________________________________________________________ Example: NH3(aq) + H2O(l) NH4+(aq) + OH-(aq) Unit # 9 (Book Chapter 19) Acids, Bases, and Salts -2- II. Hydrogen Ions and Acidity: A. Hydrogen Ions: self-ionization = _______________________________________________________ H2O(l) H+(aq) + OH-(aq) note: H+(aq) = H3O+ - the ionization of water is _____________________ and Le Chatelier’s principle applies - the product of the hydrogen-ion concentration and the hydroxide-ion concentration = __________________ @ 25°C = _________________________ _____________________________________________________________ = Kw Kw = [H+] * [OH-] = 1.0 * 10-14 acidic solution = _______________________________________________________ basic solution (alkaline solution) = _________________________________________ _____________________________________________________________________ Example problem: If the [H+] in a solution is 1.0 * 10-5 M, is the solution acidic, basic, or neutral? What is the [OH-] of this solution? B. pH: pH = ________________________________________________________________ _____________________________________________________________________ pH = -log[H+] - in pure water [H+] = 1*10-7 M and the pH is 7. - Review table 19.5 on page 656. Example problem: What is the pH of a solution with a hydrogen-ion concentration of 4.2 * 10-10 M? Example problem: The pH of an unknown solution is 6.35. What is the hydrogen-ion concentration? Unit # 9 (Book Chapter 19) Acids, Bases, and Salts -3- Example problem: What is the pH of a solution if [OH-] = 4.0 * 10-11 M? C. Measuring pH: acid-base indicators or ________________________ can be used to measure pH. acid-base indicators = ___________________________________________________ _____________________________________________________________________ - review figure 19.8 on page 660. pH meter = ___________________________________________________________ _____________________________________________________________________ III. Strong and Weak Acids and Bases Acids and bases are classified as strong or weak depending on ______________________ ___________________________________________________________________________. A. Acids strong acid = an acid that ________________________________ in aqueous solution Examples: hydrochloric acid, sulfuric acid, nitric acid HCl(g) + H2O(l) H3O+(aq) + Cl-(aq) *** notice the completion arrow (rxn goes to completion) weak acid = an acid that _________________________________ in aqueous solution Examples: acetic acid, carbonic acid CH3COOH(aq) + H2O(l) H3O+(aq) + CH3COO-(aq) *** <1% ioniztion Review table 19.6 on page 664. Unit # 9 (Book Chapter 19) Acids, Bases, and Salts -4- B. Acid Dissociation Constant – Ka Write the Keq for the dissociation of CH3COOH(aq) above. *** for dilute aqueous solutions the concentration of water is a constant so, Ka = Keq * [H2O] = [H3O+] * [CH3COO-] [CH3COOH] the acid dissociation constant (Ka) measures the extent to which an acid dissociates in aqueous solution, and so can be used to represent the ___________ of an acid. The stronger the acid, the _____________________________ value. Example Calculation: At equilibrium a solution of acetic acid has the following concentrations: [CH3COOH] = 0.0987 M, and [H3O+] = [CH3COO-] = 1.34 * 10-3 M. Find Ka. Additional Example Calculation: In a 0.200 M solution of a monoprotic weak acid, [H+] = 9.86 * 10-4 M. What is the Ka for this acid? Unit # 9 (Book Chapter 19) Acids, Bases, and Salts -5- C. Bases strong base = a base that ________________________________ into ____________ ___________ and _________________________________ in aqueous solution Examples: calcium hydroxide, magnesium hydroxide, sodium hydroxide weak base = a base that _________________________________________________ __________________________________________________________ – amount of dissociation is relatively __________________ Examples: acetic acid, carbonic acid NH4+(aq) + OH-(aq) NH3(aq) + H2O(l) *** this equilibrium favors the reactants D. Base Dissociation Constant – Kb Write the Keq for the dissociation of NH3(aq) above. *** for dilute aqueous solutions the concentration of water is a constant so, Kb = Keq * [H2O] = Or in general form: Kb = [conjugate acid] * [OH-] [base] the base dissociation constant (Kb) measures the extent to which a base dissociates in aqueous solution, and so can be used to represent the ___________ of a base. The stronger the base, the _____________________________ value. Unit # 9 (Book Chapter 19) Acids, Bases, and Salts -6- E. Differentiating Concentration and Strength: Strength refers to the extent of _________________________________________ of an acid or base, while concentration indicates how much of a substance is _______________________. Recall that substances __________________________________________________ (sugar). Acidic Solution Molar Concentration Hydrochloric acid 12 M HCl Relative Concentration Concentrated Relative Strength Strong Gastric Juice (stomach acid) Acetic acid 0.08 M HCl Concentrated Weak Vinegar 0.2 M CH3COOH 17 M CH3COOH *** Strength is _________________________ of concentration and vice versa. IV. Neutralization Reactions A. Acid-Base Reactions = Neutralization Reactions In general acids and bases react to produce a _____________ (ionic substance) and __________________. The complete reaction of a strong acid and a strong base produces a neutral solution – _________________________________________. Example: HCl(aq) + NaOH(aq) Mole Ratios and Neutralization Reactions (example problem) How many moles of sulfuric acid are needed to neutralize 0.50 moles of sodium hydroxide? Unit # 9 (Book Chapter 19) Acids, Bases, and Salts -7- B. Titration Titration = the process of ________________________________________________ _____________________________________________________________________ – using a neutralization reaction to determine concentration Steps: 1. a measured volume of an acid solution of _____________________________ is added to a flask 2. an ___________________________ is added 3. measured volumes of a _________________________________________ are mixed into the acid until the indicator just barely changes color The solution of known concentration is called the ______________________. Neutralization occurs (titration is complete) when _______________________ _________________________________________________________________. = the equivalence point = end point Determining the concentration by titration mathematically: Example: A 25 mL solution of H2SO4 is neutralized by 18 mL of 1.0 M NaOH. What is the concentration of the H2SO4 solution? The equation for the reaction is: H2SO4(aq) + 2NaOH(aq) Na2SO4(aq) + 2H2O(l) Conversion plan: Additional Example: How many milliliters of 0.45 M HCl will neutralize 25.0 mL of 1.00 M KOH? Unit # 9 (Book Chapter 19) Acids, Bases, and Salts -8- V. Salts in Solution A. Salt Hydrolysis Recall that some salts can hydrolyze water – _____________________________ ________________________________________________________ (Hydrolysis of Salts Lab) salt hydrolysis. salts that produce acidic solutions have _________________________ ____________________________________________________________ salts that produce basic solutions have __________________________ ____________________________________________________________ Example: ammonium chloride Ammonium chloride completely ionizes in water: NH4Cl(aq) the ammonium ion is a strong enough acid to donate a hydrogen ion to a water molecule: NH+4(aq) + H2O(l) the resulting H3O+ ions make the solution somewhat _______________ Strong acid + Strong base neutral solution Strong acid + Weak base acidic solution (salt’s cation releases hydrogens to water) Weak acid + Strong base basic solution (salt’s anion attracts hydrogens from water) Unit # 9 (Book Chapter 19) Acids, Bases, and Salts -9- B. Buffers Buffer = a solution that ___________________________________________ – a solution of a weak acid and one of its salts or a solution of a weak base and one of its salts a buffer contains ___________________________________________ _________________________ and _______________________________ _____________________________________________, therefore adding hydrogen ions and/or hydroxide ions has little effect on the pH of the solution Example: A solution of carbonic acid and sodium bicarbonate forms a buffer: If you add H+ ions: HCO3- + H+ If you add OH- ions H2CO3 + OH Buffer Capacity = the _______________________________________ ____________________________________________________________ _____________________________________ before a significant change in pH occurs (before your H+ ion acceptors and/or H+ ion donors run out) Important Buffer Systems Buffer name Ethanoic acid-ethanoate ion Dihydrogen phosphate ion-hydrogen phosphate ion Formulas Buffer pH* CH3COOH/CH3COO− 4.76 H2PO4−/HPO42− 7.20 H2CO3/HCO3− 6.46 NH4+/NH3 9.25 Carbonic acid-hydrogen carbonate ion (solution saturated with CO2) Ammonium ion-ammonia * Components have concentrations of 0.1M. Example Problem: A buffer consists of methanoic acid (HCOOH) and methanoate ion (HCOO-) Write an equation to show what happens when an acid is added to this buffer. And a base. Unit # 9 (Book Chapter 19) Acids, Bases, and Salts - 10 -