Ostomy Guidelines Application Forms ( - 307 KB)
advertisement

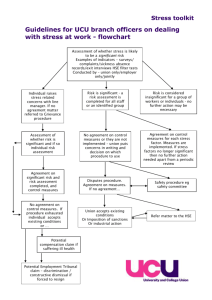
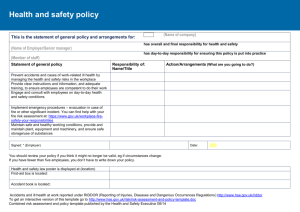
HSE PRIMARY CARE REIMBURSEMENT SERVICE OSTOMY PRODUCTS REIMBURSABLE UNDER THE GMS AND THE COMMUNITY DRUGS SCHEMES GUIDELINES FOR MANUFACTURERS/DISTRIBUTORS Primary Care Reimbursement Services Exit 5, M50 North Road Finglas Dublin 11 April 2014 INTRODUCTION These guidelines have been prepared by a HSE Expert Group, including Patient Representatives, for the information of Manufacturers/Distributors of Ostomy Products. Additions to the Reimbursable List will be considered in compliance with the Health (Supply and Pricing of Medical Goods) Act, 2013. 1. There are two types of submission for products: Type 1-New/Innovative Products and Type 2-Existing (Rollover/Minor Change) Products. 2. These guidelines are applicable to both Type 1 and Type 2 products. 3. Companies wishing to have a product listed for reimbursement under the GMS and Community Drugs Schemes must make application to:Ms Karen Burns HSE Urinary & Ostomy Review Group c/o HSE Procurement Units 1-3 Finisklin Business Park Sligo Ireland. 4. Applicants must demonstrate:(a) (b) (c) (d) Compliance with the appropriate British Standard, ISO Standard or any other equivalent standard in force in a Member State of the European Union. or Compliance with Directive 93/42/EEC, with each product carrying the CE marking. This Directive is implemented in Ireland via the European Communities (Medical Devices) Regulations 1994 (S.I. No.252 of 1994). and Compliance with the attached Criteria for Ostomy Products. and Consumer acceptability by the presentation of the results of User Trials to be conducted independently of the Manufacturer. Minimum Data requirements are set out in Appendix 1. 5. Products used on the body, or inserted into the body, must be safe and of good quality. 6. Must be suitable for self administration use under the supervision of a general medical practitioner or other relevant health professional and not be restricted to hospital or medical specialist use. 7. Must be cost effective. This may be demonstrated via published literature in peer reviewed journals. Although this is primarily applicable to Type 1 Innovative Products, the HSE may request same for premium price product on a case by case basis. 1 8. Must not be advertised or promoted to the public in everyday magazines/newspapers/TV/radio. However marketing activity which is aimed primarily at healthcare professionals is acceptable. Claims for patient outcome improvement should be supported by clinical evidence. On line advertising can present difficulties where claims for patient outcome improvement are made without the clinical evidence to support the claim. 9. The list of Ostomy Products reimbursable under the GMS and Community Drugs Schemes will be maintained in compliance with the Health (Pricing and Supply of Medical Goods) Act 2013. The HSE acknowledges that it will comply with the requirements of this Act. However the interchangeability or substitution clause is intended for those medicinal products deemed interchangeable by the Irish Medicines Board and will not be applied to devices. However, the HSE reserves the right to determine a bench mark price for products that are similar in nature and deliver the same functionality to patients. 10. The Categories / Classifications listed may be subject to change on an annual basis. APPLICATION PROCESS 1. Applicants should complete the price application form and appropriate application form for Type 1 or Type 2 products, as attached, for each product they seek to include on the Reimbursement List for GMS and Community Drugs Schemes. One signed copy of each form, along with appropriate backup material should be sent to the HSE – see Appendix 3, Application Checklist. 2. In the case of products similar to products already listed, the HSE will generally aim to ensure that the price of the new products is broadly in line with those already listed. If applicant companies want, at the time of application, to suggest factors supporting the price they are seeking they are free to do so. Similar products are those that, in the opinion of the HSE Expert Group, offer an equivalent technical solution and/or an equivalent level of clinical care for patients. 3. The original hard signed copy of the application form must be received before an application can be accepted formally by the HSE and an acknowledgement letter sent. 4. Product samples should be identical to the final product, though not necessarily from a production run if this is impractical. The text of the proposed labelling should be final although it may be presented in mock up form if the finally produced version is not available. 5. In this first phase all applications submitted must be received complete by the closing dates of 23rd June 2014 (Type 2) and the 11th July 2014 (Type 1). Going forward the HSE will acknowledge each application within 10 working days of receipt and will be considered in compliance with the Health (Pricing and Supply of Medical Goods) Act 2013. An application will not be released for assessment until all necessary information has been received. If, after 28 working days, the full documentation has not been received by the HSE, a reminder will be sent asking for this to be sent within the next 28 days. Failure to send the missing paperwork / samples will prompt 2 the HSE to ask for collection of any paperwork / samples to be arranged within the next 7 days, after which they will be destroyed. Any further applications for the product will then have to start anew. 6. Manufacturers should inform the HSE if any changes are made to their products that affect their entry to the Reimbursable List e.g product name change, a change in Manufacturer / Distributor of the product, a change in packaging (including product reference codes), a change in product specification and if the product has been discontinued. 7. Products that the Manufacturer/Distributor wishes to have deleted/discontinued (Type 2-Existing (Rollover/Minor Change)) from the Reimbursement List must remain valid for a period of at least 12 months to allow for client transition to an alternative product. 8. Where an application has been rejected, either because (i) it does not satisfy the product criteria or (ii) a price premium has been requested. An appeal can be registered with the HSE with an opportunity to outline the patient benefits afforded by the product which cannot be satisfied by the products already on the Reimbursement List. In such circumstances, an appropriate cost effectiveness analysis, cost benefit analysis or cost-consequences analysis can be submitted in support of the application outlining the clear patient benefits for the price premium requested. Note: Where an application has been rejected, the applicant will be furnished with the reason why and representation will be allowed with the applicant having discretion to provide additional information in response to the initial rejection. 3 CRITERIA FOR OSTOMY PRODUCTS 1. Ostomy Collection Pouches 1.1 1.1.1 1.1.2 Size (All Pouches) All pouches except Fistula must be available in a wide range of gasket sizes. Fistula pouches must be available as a standard "cut to fit". 1.2 1.2.1 1.2.2 1.2.3 1.2.4 Plastic (All Pouches) The plastic must be of good quality and be odour proof. Must be flexible and comfortable. Must be discreet and unobtrusive. Must be quiet film. 1.3 1.3.1 Pouch (All Pouches) The pouch must have a soft, absorbent backing and be available in clear (including split cover variants) and opaque presentations. Each box of pouches must be packed in box sizes ranging 10 – 30 units. In the case of two piece systems, shelf containers and preferably unit and inner containers, must contain a statement as to which pouches and flanges will couple together. Each box of pouches must contain a leaflet with application directions. Directions must be easily understood by the client and include illustrations for ease of comprehension. A measuring guide must be available for correct fitting and must be included in each box or be available upon request. In the case of fistula pouches a template must be included. 1.3.2 1.3.3 1.3.4 1.3.5 . 1.4 1.4.1 1.4.2 1.4.3 1.4.4 1.4.5 Adhesive (All Pouches and Flanges) Must be easy to apply and remove. Must be secure. Must be comfortable. Must incorporate a skin protective. If the appliance incorporates an adhesive tape, the tape must be non allergenic. 1.5 1.5.1 Filters Filters must be discreet and effective. (i.e. Odour or faecal material should not leak from the filter, they should prevent ballooning, designed to avoid blockage of the filter, waterproof in shower/bath or filter covers should be included in box of products). 1.6 1.6.1 1.6.2 1.6.3 Pouch Closures (Drainable, Fistula and Post-Op. Pouches) Pouch closures should be integrated into the pouch. Pouch closures should be easy to clean and allow for repeated opening and closing. If a soft wire tie is the method of closure a minimum of one tie per pouch must be included. If a bung type closure is the method of closure the bung should be securely attached to the pouch. Standard clip – 1 clip per ten pouches is acceptable. 1.6.4 1.6.5 4 1.7 1.7.1 1.7.2 Convexity Convex products should identify the degree of convexity e.g. shallow or deep or more preferably the depth of convexity in mm’s. This should be displayed on outer box. Literature included with the pouch should outline appropriate use of and precautions required when using convexity. E.g. should be used under the supervision of a medical heathcare professional such as a stoma clinical nurse. 1.8 1.8.1 Taps (Urostomy) The tap must be easily opened and closed i.e. user-friendly and must not pose a hazard when left in an open position i.e. when the pouch is used in conjunction with leg and night drainage bag. 1.9 1.9.1 Non-Return Valve (Urostomy) Urostomy pouches must have a non-return valve. 1.10 1.10.1 Connectors (Urostomy) Connectors on urostomy pouches must be universal to facilitate easy attachment to leg and night drainage pouches. One connector per pack of 10 pouches is acceptable. 1.10.2 1.11 1.11.1 Tubing (Urostomy Drain Tubing) Tubing must be constructed in material with anti kink properties, yet must be sufficiently flexible to allow easy manipulation and routeing. 1.12 1.12.1 Drainage Bags Faecal ostomy pouch free drainage bags must; 1.12.1.1 Contain an instruction leaflet. 1.12.1.2 Have a capacity between 2 and 3 litres. 1.12.1.3 Be made from a good quality plastic. 1.12.1.4 Have volume measuring markings on bag. 1.12.1.5 Have a durable and secure emptying tap/device. 1.12.1.6 Have universal eyelets to attach bag to a stand. 1.12.1.7 Be available in pack sizes between 10-30 units. Drainage bags for use with urostomy pouches - see "Criteria for Urinary Incontinence Products". 1.12.2 1.13 1.13.1 1.13.2 1.13.3 Flanges Two Piece pouches must be easy to attach to flanges. In the case of two piece systems, shelf containers and preferably unit and inner containers, must contain a statement as to which pouches and flanges will couple together within a range. However there may be instances whereby a client may not achieve the minimum wear time. Consequently, users should be advised that the minimum wear time is conditional e.g. on there being no leakage from under the flange or an adverse skin reaction occurring. 5 2. Adhesive Removers 2.1 2.2 2.3 2.4 2.5 2.6 Must include clear Client instructions i.e.- wash skin following use. Must not damage skin. Must not cause pain on application. Contents must be clearly marked. Must be easy to use. Must not compromise the adhesion of the new pouch being applied. 3. Belts 3.1 3.2 3.3 3.4 3.5 Must include clear Client instructions. Must be comfortable next to skin i.e. plastic tabs. Must be of washable fabric. Must be compatible with pouches. Must be easy to adjust. 4. Deodorants/Air Freshener 4.1 4.2 Must include clear Client instructions i.e. if applicable not to be sprayed on or near stoma. Must be ozone friendly. 5. Irrigation/Plug Devices 5.1 5.1.1 5.1.2 5.1.3 5.1.4 Irrigation/Plug Appliances Appliances must contain clear instructions with illustrations for client use. Appliances must clearly state approximate length of time for which it can be used i.e. Pouch, Tubing, Cone - not less than 1 year. Sleeves must be packed in boxes of 10-50 units. Cones must be packed singly. 5.2 5.2.1 5.2.2 5.2.3 5.2.4 Irrigation Pouch Plastic must be of good quality. Capacity must be 1000mls. to 2000mls. Volume capacity must be clearly labelled and easily read. Pouch must incorporate a good quality holder/handle. 5.3 5.3.1 5.3.2 5.3.3 5.3.4 Irrigation Tubing Must be of good quality plastic. Must have anti-kink properties. Must be flexible and easy to use. Must incorporate a water controlling device which is easy to use. 6 5.4 5.4.1 5.4.2 5.4.3 5.4.4 5.5 5.5.1 5.5.2 5.5.3 5.5.4 5.6 5.6.1 5.6.2 5.6.3 5.6.4 5.6.5 Cone/Catheter Material must be of good quality. Must be soft. Tip must be rounded for comfortable insertion and removal, without causing damage to the stoma or rectum. Must be easy to remove from the tubing for easy cleaning. Sleeves (One Piece) Plastic material must be of good quality. Adhesive must be easy to apply and remove from skin. Sleeves must contain security clip i.e. for when the client wishes to walk around during the procedure. Instructions must state length of time each sleeve can be used for i.e. single use. Sleeves (Two Piece) Plastic material must be of good quality. Sleeve must be easy to apply to and remove from belt flange. Sleeves must contain security clip i.e. for when the client wishes to walk around during the procedure. Instructions must state length of time each sleeve can be used for i.e. single use. Must state approximate length of time each belt can be used for i.e. minimum of 6 months. 5.7 5.7.1 5.7.2 5.7.3 5.7.4 Plugs (One Piece) Material must be of good quality. Material must be soft and comfortable. Must be discreet and unobtrusive. Must be easy to insert and remove without traumatising the stoma or peristomal skin. 5.7.5 Must incorporate an effective filter. 5.7.6 Must be available in a selection of sizes. 5.7.7 Must contain clear instructions and illustrations for use and disposal. 5.7.8 Must be packed in boxes of 10-30 units. 5.7.9 Adhesive must be secure, easy to apply and remove from skin. 5.7.10 Adhesive must incorporate a skin protective. 5.8 5.8.1 5.8.2 5.8.3 5.8.4 5.8.5 5.8.6 5.8.7 5.8.8 5.8.9 5.8.10 Plugs (Two Piece) Base plate must be soft and comfortable. Must be discreet and unobtrusive. Adhesive must be secure, easy to apply and remove from skin. Adhesive must incorporate a skin protective. Must incorporate an effective filter. Plug must be easy to attach and remove from base plate. Must be packed in boxes of 10-30 units. Must contain clear instructions and illustrations for use and disposal. Must be available in a selection of sizes. Plug must be easy to insert and remove without traumatising the stoma or peristomal skin. 7 6. Skin Fillers and Protectives (Sprays, Powders, Pastes and Wipes) 6.1 6.2 6.3 6.4 6.5 Contents must be clearly labelled. Instructions must be clearly stated. Must be easy to apply to and remove from skin. Must not cause pain on application. Must not cause damage to skin. 7. Skin Protectors (Seals/Rings) 7.1 7.2 7.3 7.4 7.5 7.6 Must adhere to moist skin. Must be comfortable on skin. Must be flexible on skin. Must be available in a variety of sizes i.e. rings. Must be easy to apply to and remove from skin. Must contain Instructions. 8. High Output Pouches 8.1 8.2 8.3 8.4 Must have a bung type closure to allow for connection to free drainage if required. Must have an adhesive resistant to corrosive effluent. Convex high output pouches must have an option of attaching a belt. Must have a larger than average pouch capacity. 9. Anal Plugs 9.1 9.2 9.3 9.4 9.5 Contents must be clearly labelled. Instructions must be clearly stated. Must be available in a variety of sizes. Must be easy to apply to and remove from skin. Must be individually wrapped. 8 Appendix 1: Guidelines for Manufacturers/Distributors on the Design and Execution of User Trials of Ostomy Products This is applicable to Type 1: New/Innovative Products only. It is not a prerequisite that a User Trial must have been conducted in Ireland. Each Company who initiated a User Trial must have appointed a person who would have been responsible to the Manufacturer for co-ordinating each trial. The duties of a Co-ordinator should have included:o Preparing information booklets containing correct trial procedures; o Formulating a questionnaire; o Distributing the above documentation; o Collecting the questionnaires; o Collating the data; o Presenting the results. A User Trial should ideally have been conducted independently of a Manufacturer/Distributor by a Stoma Care Nurse or any other appropriately qualified medical or nursing person who has no direct or indirect association with any Manufacturer/Distributor. If there was any link between the Manufacturer/Distributor and the person who conduced the trial, this link should be declared on Type 1 Product Application Form. The minimum requirements of the HSE which must have been provided for in the design and organisation of such trials are set out in Page iii of this document. Each User Trial must have been conducted at not less than 3 centres. A trial cannot be judged to have been satisfactory unless there was a minimum number of at least 20 participants in total across the 3 centres. Product Accountability – The Company was responsible for the supply of all products used for the trial including the necessary items that allowed a product to have been used effectively e.g. flanges to have been used with two piece system pouches. Permission to carry out the trial must have been obtained by the Co-ordinator who conducted the trial from the relevant authority. Each enrolled participant must have been fully informed of the trial procedures and their written consent must have been obtained. Non GMS reimbursable accessories must have been excluded in the trial of products. Each product on trial must have been assessed on its own merit and without the benefit of any additional product e.g. adhesive spray when testing pouch adhesion. A trial participant may have been withdrawn from the User Trial at any time at the discretion of the person who conducted the trial. A Company initiating a User Trial must have supplied o all technical details about their products including recommended wear time. i specifications and o adverse reaction forms to the Co-ordinator who should have transmitted them to the person who conducted the trial. The following were prerequisites for participation in a User Trial:o o o o Participants must have been willing to use the product on trial. Participants must have provided informed consent. Participants must have been able to comprehend and complete the questionnaire provided. Participants must have been established for the period outlined in the table below on the type of product under trial. Exclusions:o Participants who were found to have had a skin assessment rating of 2, 3, or 4 must have been excluded from the User Trial 0 = Normal intact skin, 1 = Patchy redness, 2 = Extensive redness, 3 = Reddened blistered but not broken, 4 = Reddened with open areas of skin. o Participants who were undergoing Radiotherapy or Chemotherapy treatment must also have been excluded from the trial. o Products excluded from User Trials are Post-Op. and Fistula pouches. The following must have been complied with when conducting a User Trial:o Length of Trial - The minimum length of trial period must have been set for each specific product as indicated hereunderClassification One/Two Piece Closed Pouches One/Two Piece Open Pouches Urostomy Pouches Adhesive Remover Pouch Closures ·. Belts Deodorant/ Air Freshener Filters Irrigation /Plug Appliances(Pouch/Tubing/Cone/Sleeve) Skin Fillers & Protectives Skin Protectors Anal Plugs o Period 10-12 Days 18-20 Days 18-20 Days To be determined by the type of pouch being worn 18-20 Days To be determined by the type of pouch being worn To be determined by the type of pouch being worn 10-12 Days 28 Days To be determined by the type of pouch being worn To be determined by the type of pouch being worn 28 days The minimum wear time for Pouches/Flanges under trial will have been the participants normal wear time for this type of product. ii Minimum Dataset Questionnaire for each product on trial must have been provided by the Co-ordinator to the person who conducted the trial and this must have been completed by the trial participant. 1. Pouches: One Piece Closed/Open 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 1.9 1.10 1.11 1.12 1.13 1.14 1.15 Security. Comfort. Odour proof material. Noise/Rustle factor. Aesthetic appearance. Filter performance. Ease of application. Ease of removal. Adhesive residue. Backing paper - ease of removal. Flexibility. Capacity adequacy. Frequency of change - durability. Visibility/Profile. Assessment of backing material 1.16 1.17 1.18 Wear time/Reasons for change. Daily activities - Showering/bathing - Ease of drying, -Filter performance, -Adhesive performance. Status of peristomal skin at final assessment. 2. Pouches: Two Piece System -Comfort, -Ability to absorb perspiration. In addition to points referred to under 1 above, the following must have been included in the Questionnaire2.1 2.2 2.3 2.4 Ease of attachment of flange to pouch. Ease of removal of flange from pouch. Incidence of "pop-off' occurrence. Flanges - Flexible, - Inflexible. 3. Urostomy Pouches In addition to points referred to under 1 above, the following must have been included in the Questionnaire3.1 3.2 3.3 3.4 3.5 3.6 3.7 Effectiveness of non-reflux valve. Effectiveness or tap, security. Ease of opening. Ease of closing. Ease of attachment to Night Drainage Bag/Leg Bag. Ease of removal from Night Drainage Bag/Leg Bag. Connectors - Ease of attachment, - Ease of removal, iii - Security. 4. Adhesive Removers 4.1 4.2 4.3 4.4 4.5 Clear instructions. Damage to skin. Pain on application. Ease of application. Compromising pouch adhesion. 5. Pouch Closures 5.1 5.2 5.3 5.4 5.5 5.6 5.7 Ease of application. Ease of opening. Ease of removal. Security. Ease of cleaning. Comfort. Discreet. 6. Belts 6.1 6.2 6.3 6.4 6.5 6.6 6.7 Clear instructions. Ease of application. Ease of removal. Ease of belt adjustment. Discreet. Comfort. Washable. 7. Deodorants/Air Fresheners 7.1 7.2 7.3 7.4 Clear instructions. Effectiveness. Ease of Usage. Acceptability of perfume. 8. Filters 8.1 8.2 8.3 8.4 Visibility profile. Effectiveness. Acceptable wear time. Leakage through filter. 9. Irrigation/Plug Appliances 9.1 9.2 Clear instructions. Ease of storage/carriage. iv 10. Irrigation Bag 10.1 10.2 10.3 10.4 Plastic - Durability. Capacity. Labelling. Holder - quality, - effectiveness. 11. Irrigation Tubing 11.1 11.2 11.3 11.4 Anti-kinking. Ease of handling. Flexibility. Water controlling device 11.5 Ease of removal from cone. 12. Cone/Catheter 12.1 12.2 12.3 12.4 Ease of insertion/removal. Comfortable. Ease of removal from tubing. Ease of cleaning. 13. Sleeves 13.1 13.2 13.3 13.4 13.5 13.6 13.7 13.8 Ease of application. Ease of removal. Comfortable. Ease of usage. Effectiveness. Ease of application of flange to belt. Ease of removal of flange from belt. Comfortable. - ease of usage, - effectiveness. (1-Piece) (1-Piece) (1-Piece) (Security clip) (Security clip) (2-Piece) (2-Piece) (2-Piece) v Appendix 2 - Pricing Rules HSE will apply to all Ostomy Product Applications 1) a. UK equivalent (at 12 month average exchange rate). OR b. Average of the lowest three European countries. OR c. Price proposed to the HSE. Note: whichever is the lowest. 2) UK equivalent based on either: a. C&D (the most current edition available at the time of pricing) b. BNF (the most current edition available), if C&D unavailable c. Submitted UK Price, if C&D and BNF not available 3) Benchmarked Pricing The HSE reserves the right to offer a reimbursement price which will be benchmarked against the prices(s) available to the HSE, for products that, in the opinion of the HSE Expert Group, offer an equivalent technical solution and/or an equivalent level of clinical care for patients. Appendix 3 – Application Checklist Type 1 - New/Innovative Products: Included Yes/No 1 Complete Type 1 Application Form. 2 Complete all sections of the Price Application Form. 3 Provide 3 samples, including patient information leaflet and outer packaging. 4 Provide CE Certificate. 5 Provide evidence – published literature. 6 Provide User Trial evidence. Note: The above must be completed for each Type 1 product for which an application is being made. Type 2 - Existing Products (Rollover/Minor Change) Included Yes/No 1 2 3 Complete appropriate Type 2 Application Form for each GMS Code. (Continuation, Minor Change or Discontinuation) Complete all sections of the Price Application Form. 4 Provide artwork or packaging (Minor Change). Sample is required if artwork not available. Provide Patient information leaflet (Minor Change). 5 Provide CE Certificate. Note: The above must be completed for each Type 2 existing GMS Code for which an application is being made. An application will not be released for assessment until all necessary information has been received. Appendix 4 – Forms 1. Price Application Form – Type 1 and Type 2 Products 2. Product Application Form – Type 1 New/Innovative Products to be added to the HSE List of Reimbursable Items 3. Product Application Form – Type 2 Declaration Form for Continued Listing of Product/s 4. Product Application Form - Type 2 Minor Change of Product on the HSE List of Reimbursable Items 5. Product Application Form – Type 2 Notice of Intention to Discontinue Product from the HSE List of Reimbursable Items ===================== HEALTH SERVICE EXECUTIVE PRICE APPLICATION FORM: Reimbursement of Ostomy Products Product Name & Description GMS Code*: Pack Size Type of Application (Type 1/Type 2) Reimbursement Price Proposed to HSE(€) Product Reference Code: European Pricing United Kingdom price should be quoted in Pound Sterling. State the European Country and Reimbursement Price in Euro where this product is marketed and reimbursed under the country’s Schemes/Insurance System. HSE will require independent validation of the European prices submitted which must accompany this form. Where this information is not available, please provide explanatory footnote/s in the table provided below. If this product is not available, specify N.A. United Kingdom Country Country Country £ € € € Country Country Country Country € Country € Country € Country € Country € Country € Country € Country € Country € € € € Country Average of the three lowest European Countries Country Country Average € € European Pricing Footnotes: *GMS Code where applicable € € United Kingdom Equivalent C&D £ (the most current edition available at time of application) BNF (if C&D price is not available) (the most current edition available at time of application) £ Name and Address of Key Contact Name: Position: Address: I confirm that the information provided in this application is correct. Signature: __________________________ Telephone No: ____________________________ Date: _______________ E-mail Address: ________________ The completed form along with application information should be returned to; Ms Karen Burns HSE Urinary & Ostomy Review Group c/o HSE Procurement Units 1-3 Finisklin Business Park Sligo Ireland. HEALTH SERVICE EXECUTIVE TYPE 1 Product Application Form: New/Innovative Ostomy Products to be added to the HSE List of Reimbursable Items Product Name: Product Description: Product Pack Size: Product Reference Code: Product Specification: Manufacturer/Distributor: Proposed launch date for product: Please confirm that the product will be made readily available to a dispensing contractor through the normal wholesale network. I confirm that the product will be made readily available to a dispensing contractor □ Are comparable products already listed including those from other manufacturers/suppliers Y/N, If Y please detail here: User Trial A User Trial should ideally have been conducted independently of a Manufacturer/Distributor by a Continence Care Nurse or any other appropriately qualified medical or nursing person who had no direct or indirect association with any Manufacturer/Distributor. If there was any link between the Manufacturer/Distributor and the person who conducted the trial, please declare and give details of the link here: Application Checklist All sections of Type 1 Product Application Form complete □ Price Application Form submitted □ The correct/relevant documents submitted □ CE Certification submitted □ Sample of the final packaging submitted □ Three samples of the product submitted (including Patient Information Leaflet) □ This application will not be released for assessment until all necessary information has been received. Name and Address of Key Contact Name: Position: Address: I confirm that the information provided in this application is correct. Signature: __________________________ Telephone No: ____________________________ Date: _______________ E-mail Address: ________________ The completed form along with application information should be returned to; Ms Karen Burns HSE Urinary & Ostomy Review Group c/o HSE Procurement Units 1-3 Finisklin Business Park Sligo Ireland. HEALTH SERVICE EXECUTIVE TYPE 2 Product Application Form: Declaration Form for Continued Listing of Ostomy Product/s I declare that Product Name & Description Pack Size Product Reference Code GMS Code Current HSE Reimbursement Price € Is still available under the terms agreed when it was first listed and there have been no changes to these. All certification is up to date. In the event of any changes to the above, the HSE will be notified immediately in writing. Name and Address of Key Contact Name: Position: Address: I confirm that the information provided in this application is correct. Signature: __________________________ Telephone No: ____________________________ Date: _______________ E-mail Address: ________________ The completed form should be returned to; Ms Karen Burns HSE Urinary & Ostomy Review Group c/o HSE Procurement Units 1-3 Finisklin Business Park Sligo Ireland. N.B. If the information provided is found to be incorrect by the HSE, your product will be immediately deleted from the HSE List of Reimbursable Items. HEALTH SERVICE EXECUTIVE TYPE 2 Product Application Form: Minor Change of Ostomy Product on the HSE List of Reimbursable Items This form has been prepared to enable manufacturers to inform the HSE of a proposed change of Ostomy product(s) on the HSE List of Reimbursable Items. Changes to a product can include, for example; ● Packaging of product (including pack size) ● Product Specification ●Name of the product ● Manufacturer/Distributor of the product ● Product Reference Code GMS Code: Current Listing Proposed Changes Product Name: Product Description: Product Pack Size: Product Reference Code: Product Specification: Product Packaging: Manufacturer/Distributor: Other: Proposed date for minor change: Date (month and year) when it is estimated that stocks of currently listed product will be depleted: A copy of any letter(s) sent or proposed to be sent to Health Care Professionals in relation to the minor change of the product: NOTE: For ALL Minor Change Applications, the HSE will require copy of the outer packaging artwork, CE cert/license and patient information leaflet for currently listed product AND proposed minor change product. Name and Address of Key Contact Name: Position: Address: I confirm that the information provided in this application is correct. Signature: __________________________ Telephone No: ____________________________ Date: _______________ E-mail Address: ________________ The completed form should be returned to; Ms Karen Burns HSE Urinary & Ostomy Review Group c/o HSE Procurement Units 1-3 Finisklin Business Park Sligo, Ireland. N.B. If the information provided is found to be incorrect by the HSE, your product will be immediately deleted from the HSE List of Reimbursable Items. HEALTH SERVICE EXECUTIVE TYPE 2 Product Application Form: Notice of Intention to Discontinue Ostomy Product from the HSE List of Reimbursable Items This form has been prepared to enable manufacturers, importers or their agents to notify the HSE of the withdrawal of an ostomy product. At least 3 months is required where there is a reimbursed alternative for the approved indication available on the market. At least 12 months is required where there is no reimbursed alternative for the approved indication available on the market. This is in the interest of maintaining an uninterrupted supply of ostomy products to patients. Product Name & Description: Product Reference Code: GMS Code: Pack Size: HSE Reimbursement Price: Expiry date of last manufactured batch: Proposed date for product discontinuation: Date (month and year) when it is estimated that stocks of product will be depleted: Where the product discontinuation is of a particular pack size within a range of products provide details of those products that will continue to remain available: Give reasons for the proposed product discontinuation of the product (s) with appropriate substantiating information: If there is a reimbursed alternative to the product being discontinued please provide details: Provide an evaluation of likely impact that the proposed discontinuation will have on the quality of patient care, including an estimate of the number of patients it will affect: Provide details of the current status and availability of the product in the various Member States of the European Union: A copy of any letter(s) sent or proposed to be sent to Health Care Professionals in relation to the discontinuation of the product. € Name and Address of Key Contact Name: Position: Address: I confirm that the information provided in this application is correct. Signature: __________________________ Telephone No: ____________________________ The completed form should be returned to; Ms Karen Burns HSE Urinary & Ostomy Review Group c/o HSE Procurement Units 1-3 Finisklin Business Park Sligo Ireland. Date: _______________ E-mail Address: ________________