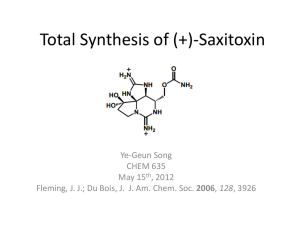
References
advertisement

References REFERENCES Adlington, R.M., Baldwin, J.E., Becker, G.W., Chen, B., Cheng, L., and Cooper, S.L., “Design, synthesis, and proposed active site binding analysis of monocyclic 2-azetidinone inhibitors of prostate specifics antigen,” J. Med. Chem., 2001, 44, 1491–1508. Alan, A.W., “Use of the berthelot reaction in the automated analysis of serum urea nitrogen”, Clin. Chem., 1966, 12, 151-157. Aliasghar, J., and Maaroof, Z., “Synthesis of novel n-sulfonyl monocyclic βlactams as potential antibacterial agents,” Molecules, 2006, 11, 49-58. Alistair, K., Sudharsan, S., Laurent, K., Sandra, L., Lynn, G., James, C. and Gurdyal, S., “Probing the mechanism of the Mycobacterium tuberculosis βketoacyl-acyl carrier protein synthase III : Factors influencing catalysis and substrate specificity. J. Biol. Chem., 2005, 280, 32539-32547. Amit, V., and Shailendra, K.S “4-Thiazolidinone – A biologically active scaffold,” Eur. J. Med. Chem., 2001, 43, 897-905. Antonio, C., Paolo, S., Laura, G., Donatella, U. and Stefania, Z., “Novel functionalized pyrido[2,3-g]quinoxalinones as antibacterial,antifungal and anticancer agents,” Farmaco, 2001, 56, 933–938. Asha, B., Abdul, R.B. and Amir, A., “Synthesis of new 2-(5-substituted-3phenyl-2-pyrazolinyl)-1,3-thiazolino[5,4-b]quinoxaline derivatives and evaluation of their antiamoebic activity,” Eur. J. Med. Chem., 2009, 44, 13171325. Asuncion, B., Eleni, P., Dimitra, H., Raquel, V., Esther, V., Beatriz, S., Saioa, A., Silvia, P., Ignacio, A. and Antonio, M., “Synthesis and antiinflammatory/ antioxidant activities of some new ring substituted 3-phenyl-1-(1,4-di-Noxidequinoxalin-2-yl)-2-propen-1-one derivatives and of their 4,5-dihydro(1H)-pyrazole analogues,” Bioorg. Med. Chem. Lett., 2007, 17, 6439–6443. Aysel, G., Taylan, I., Nalan, T. and Gulten T., “Synthesis and antimicrobial evaluation of some novel imidazolyl mercapto acetyl thiosemicarbazide and 4-thiazolidinone analogs,” Turkish J. Pharm. Sci., 2005, 2, 1-10. Balzarini, J., Rao, A., Carbone, A., Chimirri, A., Clercq, E.D., Monforte, A.M., Monforte, P., Pannecouque, C. and Zappala, M., “Synthesis of new 2,3diaryl-1,3-thiazolidin-4-ones as antiHIV agents,” Farmaco, 2004, 59, 33-39. Bharti, A.R., Nally, J.E. and Ricaldi, J.N., “Leptospirosis: A zoonotic disease of global importance,” Lancet Infect. Dis., 2003, 3, 757–771. 102 References Bhoot, D.P., Khunt, R.C., Shankhavara, V.K. and Parekh, H.H., “Synthesis of some new heterocyclic compounds with potential biological activity,” J. Sci. I. R. Iran, 2006, 17, 323-325. Chan, S.C., Wen, X.R. and Sang, C.S., “Ketones as a new synthon for quinoxaline synthesis,” Tetrahedron Lett., 2007, 48, 4665-4667. Chatrabhuji, P.M., Vyas, K.B., Nimavat, K.S. and Undavia, N.K., “Synthesis and antimicrobial activity of some 4 - oxo – thiazolidines-5- acetic acids,” Asian J. Biochem. Pharm. Res., 2009, 1, 2231-2560. Chavan, A.A. and Pai, N.R., “Synthesis and biological activity of nsubstituted 3-chloro-2-azetidinones,” Molecules, 2007, 12, 2467-2477. Colette, G. and Jean-Marie, G., “Biochemistry and comparative genomics of sxxk superfamily acyltransferases offer a clue to the mycobacterial paradox: presence of penicillin-susceptible target proteins versus lack of efficiency of penicillin as therapeutic agent,” Microbiol. Mol. Biol.Rev., 2002, 76, 702-738. Desai, N.C., Dave, D., Shah, M.D. and Vyas, G.D., “Synthesis and antibacterial activity of some novel 4-oxo-1,3-thiazolidines, 2-oxoazetidines and 5-oxoimidazolines,” Indian J. Chem., 2000, 39, 277-282. Desai, N.C., Shah, M.D. and Bhavsar, A.M., “Synthesis and QSAR studies of 4-oxothiazolidines and 2-oxoazetidines as potential antibacterial agents,” Indian J. Chem., 2008, 47, 1135-1144. Dharmchand, P.S., Sanjaykumar, D., Syed, R.H. and Ramgopal. S., “Synthesis and antimicrobial activity of some new quinoxaline derivatives,” Pharmaceuticals, 2010, 3, 2416-2425. Esther, V., Silvia, P.S., Lidia, M.L., Saioa, A., Asuncion, B., Beatriz, S., Raquel, V., Ignacio, A. and Antonio, M., “Selective activity against Mycobacterium tuberculosis of new quinoxaline1,4-di-N-oxides,” Bioorg. Med. Chem., 2009, 17, 385–389. Giri, S., Sharan, P. and Nizamuddin, A., “Syntheses of some 1-(substituted coumarin-3-carboxamido)-3-substituted-4-aryl-2-azetidinones as potential antifungal agents,” Agricul. Biol. Chem., 1989, 53, 1153-1155. Girija, S.S., and Boycie, J.M., “Synthesis of 2-azetidinones from 2-diazo-1, 2diarylethanonesand N-(2-thienylidene)imines as possible antimicrobial agents,” Farmaco, 2005, 60, 727–730. Goel, R.K., Singh, A., Naidu, P.S., Mahajan, M.P. and Kulkarni, S.K., “PASS assisted search and evaluation of some azetidin-2-ones as C.N.S. active agents,” J. Pharm. Pharmaceut. Sci., 2005, 8, 182- 189. 103 References Grinev, A.N ., “6-Substituted indolo[1,2-c]quinazolines as new antimicrobial agents”, Pharm Chem J, 1978,12, 233-236. Halve, A. K., Bhadauria, D., and Dubey, R., “N/C-4 substituted azetidin-2ones, synthesis and preliminary evaluation as antimicrobial agent,” Bioorg. Med. Chem., 2007, 17, 341-345. Hasaninejada, A.Z., Mohammadizadeha, M.R. and Karamia, Z., “Synthesis of quinoxaline derivatives via condensation of aryl-1,2-diamines with1,2diketones using (NH4)6MO7O24. 4H2O as an efficient, mild and reusable catalyst,” J. Iran. Chem. Soc., 2009, 6, 153-158. Hongyu, Z., Shuhong, W., Shumei, Z., Aifeng, L., Ying, S., Rongshi, L., Ying, Z., Sean, E., Peter, W., Swaan, O., Bingliang, F., Bin, Z., and Bing, Y., “Design, synthesis, cytoselective toxicity, structure–activity relationships, and pharmacophore of thiazolidinone derivatives targeting drug resistant lung cancer cells,” J. Med. Chem., 2008, 51, 1242–1251. Itamar, S., Ada, B. and Abraham, S., “Efficacy of ciprofloxacin against leptospira interrogans serogroup icterohaemorrhagiae,” Antimicrob. Agents Chemother., 1989, 33, 788-789. Jasmer, R.M., Nahid, P.and Hopewell, P.C., “Clinical practice: Latent tuberculosis infection,” N. Engl. J. Med., 2002, 347, 1860–1866. Jean, R., Michel, B. and Patrick, M., “Quinoxaline-5,6-(and 5-8)-dionespotential antitumoral agents,” Eur. J. Med. Chem., 1981, 16, 545-550. Johannes, G.V., Gerhard, H., and Gerhard, S., “Facile retro dieckmann reactions of 3-0xo-carbapenam esters”, Heterocycles, 1985, 23, 1081-1084. Jonathan, D.G. and Lowell, H.H., “Modeling antileukemic activity of carboquinones with electrotopological state and chi indices,” J. Chem. Inf. Comput. Sci., 1999, 39, 356-364. Julius, A.G., “The calorimetric determination of r-glutamyl transpeptidase with a synthetic substrate,” Arch. Biochem. Biophys. 1960, 91, 61-70. Kaythara, P., Upadhayay, T., Doshi. R., and Parel, H.H., “Synthesis of some 2- azetidinones as potential antitubercular agents,” Indian J. Heterocycl. Chem., 2000, 10, 9-12. Kiktenko, V.S., Balashov, N.G. and Rodina, V.N., “Leptospirosis infection through insemination of animals,” J. Hyg. Epidemiol. Microbiol. Immunol., 1976, 21, 207-213. King, E.J., “A method for the determination of tartratelabile, prostatic acid phosphatase in serum,” Am. J. Cli. Path., 1959, 12, 85-89. 104 References Knut, B., “An alignment-independent versatile structure descriptor for qsar and qspr based on the distribution of molecular features,” J. Chem. Inf. Comput. Sci., 2002, 42, 26-35. Konstantinos, A., “Testing for tuberculosis,” Australian Prescriber, 2010, 33, 12-18. Kumar, V., Abbas, A.K., Fausto, N. and Mitchell, R.N., “Robins basic pathology” (8th ed.) Saunders Elseviers, Philadelphia, 2007, pp 516-522. Kyoung, S.K., Synthesis, biological properties, and structure-activity relationships of quinoxaline angiotensin II receptor antagonists. Bioorg. Med. Chem. Let., 1993, 3, 2667-2670. Kyoung, S.K., Synthesis, biological properties, and structure-activity relationships of quinoxaline angiotensin II receptor antagonists. Bioorg. Med. Chem. Let., 1993, 3, 2667-2670. Langston, C.E. and Heuter, K.J., “Leptospirosis. A re-emerging zoonotic disease,” Vet. Clin. N.- Am.- Small., 2003, 33, 791–807. Lowell, H.H. and Lemont, B.K., “Issues in representation of molecular structure-the development of molecular connectivity,” J. Mol. Graphics Modell., 2001, 20, 2-14. Mahendra, R.S., Mangesh, G., Kailash, G.B., Shashikant, V.B., Ana, N., Kalyan, C.A., Nisheeth, C.D. and Prashant, J.B., “Synthesis and anticonvulsant activity of clubbed thiazolidinone–barbituric acid and thiazolidinone–triazole derivatives,” Arkivoc, 2007, (xiv), 58-74. Majid, M.H., Khadijeh, B., Maryam, H.T., Negar, M.J. and Hossien, A.O., “Facile synthesis of quinoxaline derivatives using o-iodoxybenzoic acid (IBX) at room temperature,” Arkivoc, 2006, (xvi), 1424-6376. Majid, M.H., Shima, T., Khadijeh, B. and Hossein, A.O., “On water: A practical and efficient synthesis of quinoxaline derivatives catalyzed by CuSO4 · 5H2O,” Catal. Commun., 2007, 8, 211-214. Malawska, B., “New anticonvulsant agents,” Current Top. Med. Chem., 2005, 5, 69-85. Marisithambaram, K., Dasappa, J.P., Bojapoojary, K., Subrahmanya, B., Bantwal, S.H. and Nalilu, S.K., “Synthesis and biological activity of Schiff and Mannich bases bearing 2,4-dichloro-5-fluorophenyl moiety” Bioorg. Med. Chem., 2006, 14, 7482-7489. Michael, P. and Malcolm, M., “Measurement of serum creatinine – current status and future goals,” Clin. Biochem. Rev., 2006, 27, 173-184. 105 References Nagraja, G.K., Marlingaplara, G.P., and Mahadevan, K., “Synthesis of novel nitrogen containing naphtha-[2,1-b]-furan derivatives and investigations of their antimicrobial activities”, Arkovick, 2006, (xv), 664-674. Naoyuki, Y., “Influence of nonspecific reaction on determination of H2O2 using trinder reagents”, Clinica Chimica Acta, 2005, 360, 52-59. Navin, B.P. and Sarvil, D. P., “Synthesis and in vitro antimicrobial study of schiff base and thiazolidinone of 1-cyclopropyl-6-fluoro-7-[4-(2,3dichlorophenyl) piperazin-1-yl]-4-quinolone,” Acta Pol. Pharm. Drug Res., 2010, 67, 45-53. Paola, V., Athina, G., Kitka, A., Matteo, I. and Franca, Z., “Synthesis and antimicrobial activity of novel 2-thiazolylimino-5-arylidene-4thiazolidinones,” Bioorg. Med. Chem., 2006, 14, 3859-3864. Paolo, S., Antonio, C., Mario, L., Stefania, Z. and Leonardo, S., “Synthesis of substituted 2-ethoxycarbonyl- and 2-carboxyquinoxalin-3-ones for evaluation of antimicrobial and anticancer activity,” Farmaco, 1998, 53, 455-461. Parekh, H.H., Parikh, K.A. and. Parikh, A.R., “Synthesis of some 4thiazolidinone derivatives as antitubercular agents,” J. Sci. I. Rep. Iran, 2004, 15, 143-148. Parikh, K.A., Oza, P.S., Bhatt, S.B., and Parikh, A.R., “Synthesis of some new 2-azetidinones as potential antitubercular agents,” Indian J. Chem., 2000, 39, 716-718. Pierre, A., Hans, A., Yves, P.A., Serge, B., Silvio, O., Dirk, S. and Markus, S., “5-Aminomethylquinoxaline-2,3-diones: Arylamide derivatives as highly potent and selective glycine-site NMDA receptor antagonists,” Bioorg. Med. Chem. Lett., 1998, 8, 493-498 Predvoditeleva, G.S., Kartseva, T.V., Oleshko, O.N., Shvedov, V.I. and Pershin, G.N., “Synthesis and study of the tuberculostatic activity of alkyl derivatives of dihydropyridazino[3,4-b]quinoxalines,” J. Pharm. Chem., 1977, 11, 1349-1353. Rajesh, H.B., Mukul, R.J., Arun, A.G., Ashish, G., Pradip, A.J., Dipam, N.P., Vijay, M.P. and Pankaj, R.P., “Synthesis and antidiabetic activity of 3,6,7trisubstituted-2-(1h-imidazol-2-ylsulfanyl)quinoxalines and quinoxalin-2-yl isothioureas,” Archiv der Pharmazie, 2007, 340, 359–366. Rao, A., Balzarini, J., Carbone, A., Chimirri, A., Clercq, E.D., Monforte, A. M., Monforte, P., Pannecouque, C. and Zappalà, M., “Discovery of 2,3diaryl-1,3-thiazolidin-4-ones as potent antiHIV-1 agent,” Bioorg. Med. Chem. Lett., 2001, 11, 1793–1796. 106 References Rao, A., Carbone, A., Chimirri, A., Clercq, E.D., Monforte, A.M., Pietro, M., Pannecouque, C. and Zappala, M., “Synthesis and antiHIV activity of 2,3diaryl-1,3-thiazolidin- 4-(thi)one derivatives,” Farmaco, 2002, 57, 747-751. Ravichandran, V., Mohan, S. and Suresh Kumar, K., “Synthesis and antimicrobial activity of Mannich bases of isatin and its derivatives with 2[(2,6-dichlorophenyl)amino]phenylacetic acid,” Arkivoc, 2007, (xiv), 51-57. Sandip, J., Mahesh, P., Veeresh, M., Pradeepkumar, R., Shivalingarao M., Darbhamulla, S., and Mangala, G., “Synthesis and antimycobacterial activity of a novel series of isonicotinylhydrazide derivatives,” Arch. Pharm. Chem. Life Sci., 2009, 342, 723–731. Saraswathi, M., Rohini, R.M., Singh, V. and Naira, N., “Synthesis and evaluation of Mannich bases of benzimidazo [1,2-c] quinazolin- 6(5h)thionefor antimicrobial activity,” Pak. J. Pharm. Sci., 2010, 23, 459-462. Saravanan, R., Rajendran, P. and Garajan, S.P., Clinical, bacteriologic and histopathologic studies on induced leptospirosis in stray dog pups. Indian J. Patho. Microbiol., 1999, 42, 463-469. Sathe, B.S., Jaychandran, E., Jagtap, V. A. and Sreenivasa, G.M., “Antitubercular activity of fluoro benzothiazole comprising potent thaizolidinone,” Int. J. Pharma. Bio Sci., 2010, 1, 332-337. Seshaiah, K.S., Muniyandy, S. and Atmakuru R., “Synthesis and antibacterial screening of hydrazones, Schiff and Mannich bases of isatin derivatives,” Sharma, M.C., Sahu, N.K., Kohli, D.V., Chaturvedi, S.C., Smita, S., “Synthesis, characterization and biological activities of some 1(nicotinylamino) -2 substituted azetidine-4 -ones as potential antibacterial agents,” Dig. J. Nanomat. Bios., 2009, 4, 361 – 367. Sheela, J., Navita, K., Deepak, K. and Rakesh, S., “Synthesis and in vitro study of novel Mannich bases as antibacterial agents,” Bioorg. Med. Chem. Lett., 2005, 15, 221-226. Shu-Kun, L., “A facile synthesis of quinoxaline–2,3–diones as NMDA receptor antagonists,” Molecules, 1996, 1, 37-40. Smythe, L., “Leptospirosis – A review of laboratory diagnostic methods”, Aust. J. Med. Sci., 2006, 27, 16-19. Sriram, D., Bal, T.R. and Yogeeswari, P., Newer aminopyrimidinimino isatin analogues as non-nucleoside HIV-1 reverse transcriptase inhibitors for HIV and other opportunistic infections of AIDS: Design, synthesis and biological evaluation ,” Farmaco, 2005, 60, 377–384, 107 References Srivastava, S.K., Srivatsava, S., and Srivastava, S.D., “Synthesis of new carbazolylthiadiazol-2-oxoazetidines,” Indian J. Chem., 1999, 36, 183-187. Subudhi, B.B., Panda, P.K., Tosh, B.K., Sahu, S., and Majhi, P., “synthesis and biological activity evaluation of some azetidinone and thiazolidinone derivatives of coumarins,” Dhaka Univ. J. Pharm. Sci., 2005, 4, 87-92. Tom, D., “Early historical milestones in HDL-cholesterol assay,” Clin. Chem., 1998, 44, 898-899. Vazzana, I., Terranova, E., Mattioli, F., and Sparatore, F., “Aromatic schiff bases and 2,3-disubstituted-1,3-thiazolidin-4-one derivatives as antiinflammatory agents,” Arkivoc, 2004, (v), 364-374. Vishnu, K.T., Dharmendra, B.Y., Hardesh, K.M., Ashok, K.C. and Praveen, K.S., “Design, synthesis, and biological evaluation of 1,2,3-trisubstituted-1,4dihydrobenzo [g]quinoxaline-5,10-diones and related compounds as antifungal and antibacterial agents,” Bioorg. Med. Chem., 2006, 14, 6120–6126. Viswajanani, J.S., Ajay, S., Smita, S., Seema, K., Manisha, P., Pragya, B., Tarun, M., Ashok, R., JagMohan, K. and Anita, M., “Synthesis and antimicrobial activity of novel thiazolidinones,” Arkivoc, 2005, (ii), 46-59. Xu, H., Julie, D., Christian, B., Philippe, M.L., Xavier, F.,Reynald, H. and Bruno, F., “Synthesis and antiprotozoal activity of some new synthetic substituted quinoxalines,” Bioorg. Med. Chem. Lett., 2006, 16, 815–820. Yanofsky, S.D., Shen, E.S., Holden, F., Whitehorn, E., Aguilar, B., Tate, E., Holmes, C.P., Scheuerman, R., MacLean, D., Wu, M.M., Frail, D.E., López, F.J., Winneker, R., Arey, B.J., and Barrett, R.W., “Allosteric activation of the follicle-stimulating hormone (FSH) receptor by selective nonpeptide agonists,” J. Biol. Chem., 2006, 281, 13226-13237. Yun, B.K., “Synthesis and biological activity of new quinoxaline antibiotics of echinomycin analogues,” Bioorg. Med. Chem. Let., 2004, 14, 541-544. 108