Ideal Gas Law – PhET Simulation
advertisement

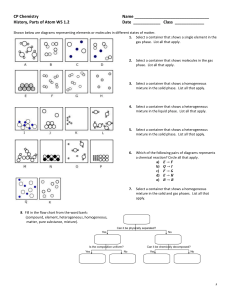
Ideal Gas Law – PhET Simulation Introduction In this simulation you will look at the motion of gas particles to describe the relationships that exist between pressure, volume, temperature, number and type of atoms in a contained gas. Procedure 1. Open: http://phet.colorado.edu/simulations/index.php?cat=Heat_and_Thermo 2. Select - gas properties simulation – run now! 3. Select constant volume 3. Pump handle just a little bit – wait for pressure to stabilize (gas well mixed) Note temperature and pressure Add more atoms by pumping some more. What happens to the temperature and pressure? _________________________________________________________________________________ Explain your answers in terms of mechanics of the gas atoms _________________________________________________________________________________ _________________________________________________________________________________ Sketch a graph of how you think the pressure of the gas in the container depends on the number of atoms in the container. P N Describe a real world situation that would be described by the graph you drew. ___________________________________________________________________________________ ___________________________________________________________________________________ 4. Press reset. 5. Put 40 “heavy species” atoms in the chamber at constant volume. Note the temperature and gas pressure when well mixed. Press Reset Put 40 “Light species” atoms in the chamber at constant volume. Note the temperature and gas pressure when well mixed. Compare the temperature and pressure for equal numbers of heavy or light gas atoms in the container ___________________________________________________________________________________ Explain your answers in terms of mechanics of the gas atoms _________________________________________________________________________________ _________________________________________________________________________________ 6. Press reset 7. Select constant volume 8. Put 100 gas atoms in the container – wait for pressure to stabilize. 9. Now select constant pressure 10. Pull arrow on heat control down, removing heat from the container. What happens to the temperature and volume? _________________________________________________________________________________ Explain your answers in terms of mechanics of the gas atoms _________________________________________________________________________________ _________________________________________________________________________________ Sketch a graph of how you think the volume of the gas in the container depends on the temperature of the gas in the container. V T (oK) Describe a real world situation that would be described by the graph you drew. ___________________________________________________________________________________ ___________________________________________________________________________________ 11. 12. 13. 14. Press reset Select constant volume Put 100 gas atoms in the container – wait for pressure to stabilize. Pull arrow on heat control down or up, removing or adding heat to the container. What happens to the temperature and pressure? _________________________________________________________________________________ Explain your answers in terms of mechanics of the gas atoms _________________________________________________________________________________ _________________________________________________________________________________ Sketch a graph of how you think the pressure of the gas in the container depends on the temperature of the gas in the container. P T (oK) Describe a real world situation that would be described by the graph you drew. ___________________________________________________________________________________ ___________________________________________________________________________________ 15. 16. 17. 18. Press reset Select constant temperature Put 100 gas atoms in the container – wait for pressure to stabilize. Click on the handle on the container and make the container smaller. What happens to the volume and pressure? _________________________________________________________________________________ Explain your answers in terms of mechanics of the gas atoms _________________________________________________________________________________ _________________________________________________________________________________ Sketch a graph of how you think the pressure of the gas in the container depends on the volume of the container. P V Describe a real world situation that would be described by the graph you drew. ___________________________________________________________________________________ ___________________________________________________________________________________