Research Protocol: Clinical Studies & Trials Guide
advertisement

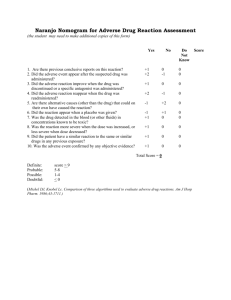
1. RESEARCH PROTOCOL A GUIDE FOR CLINICAL STUDIES, CLINICAL TRIALS AND HEALTH SERVICES RESEARCH A research protocol is the most important document relating to any research study It describes not only what the study is about and why it is important, but also all the practical, logistical and ethical issues involved It should be as detailed as necessary and provide a step-by-step guide to the study (although there may be some flexibility in the study process) The purpose of the protocol is not only for review, funding and approval processes, but for you and your team to develop your ideas, a robust methodology and a clear guide that can be referred to throughout the study; the protocol also provides a basis for production of any final reports and publications Developing a sound research protocol takes time; this is not only because it requires a sound evidence review, but also ‘thinking out the study’ as though it was taking place in ‘real-time’ and attempting to account for any potential issues and obstacles Development of the protocol also involves discussion with colleagues and management, where relevant (e.g. will they support your approach?) In line with the Research Governance Framework (RGF), it also involves peer-review (where you will need to address comments and make amendments, as appropriate) followed by ethical review and final R&D Approval If this is a clinical trial of an investigational medicinal product (IMP) then approval must be sought from the MHRA, in addition to approvals under the RGF See Clinical Trials Regulations Researchers must ensure they have training in Good Clinical Practice (GCP) before embarking on a clinical trial Clinical trial protocols may vary – this is just an example ‘Investigators Brochure’ may also be required for a clinical trial BACKGROUND INFORMATION PLEASE INSERT TEXT TO ALL RELEVANT SECTIONS. 1.1. BACKGROUND 1.2. INVESTIGATIONAL PRODUCT 1.3. REGULATORY STATUS 1.4. PRECLINICAL RESULTS 1.4.1. IN-VITRO TESTS 1.5. RISK AND BENEFITS 1.6. STUDY POPULATION 2. STUDY AIMS AND OBJECTIVES 3. TRIAL DESIGN 3.1. PRIMARY PERFORMANCE ENDPOINT 3.2. SECONDARY PERFORMANCE ENDPOINTS 3.3. SAFETY ENDPOINTS 3.4. TRIAL DURATION 3.5. STOPPING RULES 4. PATIENT SELECTION 4.1. INFORMED CONSENT 4.2. INCLUSION CRITERIA 4.3. EXCLUSION CRITERIA 4.4. SUBJECT WITHDRAWAL 5. STUDY PROCEDURES Visit 1: Inclusion Informed consent Baseline Medical history etc Visit 2 etc 6. DEVICE 7. ADVERSE EVENT REPORTING The following definitions are based on ISO 14155: 2011 Where the definition indicates “device”, it refers to any component of the system. 7.1. ADVERSE EVENT (AE) Any untoward medical occurrence, unintended disease or injury, or untoward clinical signs (including abnormal laboratory findings) in subjects, users or other persons, whether or not related to the investigational medical device NOTE 1: This definition includes events related to the investigational medical device or the comparator. NOTE 2: This definition includes events related to the procedures involved. NOTE 3: For users or other persons, this definition is restricted to events related to investigational medical devices. 7.2. ADVERSE DEVICE EFFECT (ADE) Any untoward and unintended adverse event related to the use of an investigational medical device is defined as an Adverse Device Effect. NOTE 1: This definition includes adverse events resulting from insufficient or inadequate instructions for use, deployment, implantation, installation, or operation, or any malfunction of the investigational medical device. NOTE 2: This definition includes any event resulting from use error or from intentional misuse of the investigational medical device. 7.3. SERIOUS ADVERSE EVENT (SAE) An Adverse Event that: a) led to death, b) led to serious deterioration in the health of the subject, that either resulted in 1. a life-threatening illness or injury, or 2. a permanent impairment of a body structure or a body function, or 3. in-patient or prolonged hospitalization, or 4. medical or surgical intervention to prevent life-threatening illness or injury or permanent impairment to a body structure or a body function, c) led to foetal distress, foetal death or a congenital abnormality or birth defect. NOTE: Planned hospitalization for a pre-existing condition, or a procedure required by the Clinical Investigation Plan (CIP), without serious deterioration in health, is not considered a serious adverse event. 7.4. SERIOUS ADVERSE DEVICE EFFECT (SADE) Adverse device effect that has resulted in any of the consequences characteristic of a serious adverse event Note: This definition includes incidents and near incidents. An ADE that has resulted in any of the consequences characteristic of a SAE or that might have led to any of these consequences if suitable action had not been taken or intervention had not been made or if circumstances had been less opportune, is defined as a SADE. Note: This definition includes incidents and near incidents. 7.5. SERIOUS PROCEDURE RELATED ADVERSE EVENT (SPRAE) A SAE that occurs due to any procedure specific to the clinical investigation, including the implantation or modification of the system, is defined as a SPRAE. 7.6. UNEXPECTED SERIOUS ADVERSE DEVICE EFFECT (USADE) A SADE that is unexpected in nature is defined as an unexpected serious device effect. Serious adverse device effect which by its nature, incidence, severity or outcome has not been identified in the current version of the risk analysis report NOTE: Anticipated serious adverse device effect (ASADE) is an effect which by its nature, incidence, severity or outcome has been identified in the risk analysis report. 7.7. RECORDING AND REPORTING OF ADVERSE EVENTS AEs will be documented from the point of enrollment until the patient is exited from the study. …….All AEs, regardless of relatedness or outcome, must be reported. Information reported on the AE Form shall include a description of the event, the date of event onset, the relatedness of the event to the procedure, the relatedness of the event to the device, the expectedness of a SADE and a serious procedure related AE, actions taken as a result of the event, the outcome of the event, and the date the event was first noticed by, or reported to the investigator. For AEs which require immediate reporting, initial reporting may be done by telephone or e-mail, followed by the completed electronic AE Form. Contact information is given on each AE Form and is available in the investigator site file. All other AEs will be reported on the adverse event form in a timely manner after the investigator first learns of the event. All ongoing AEs will be followed-up until the last study visit. Following study exit, there will be a one month follow-up period for safety. During this period, if any AEs are reported as device or procedure related, they will be included in the study. 7.8. EMERGENCY CONTACT DETAILS IN CASE OF SAE/SADE In case of an immediately reportable AE the investigators can contact ……by phone. Contact details are available in the Investigator Site File. 8. DATA HANDLING 8.1. SOURCE DATA DOCUMENTATION The investigator must ensure accuracy, completeness and timeliness of the data reported in CRFs and in all other required reports. Data reported on CRFs, which are derived from source documents, must be consistent with the source documents or the discrepancies need to be justified in a documented rationale, signed and dated by the (principal) investigator, to be filed in the patient file. Only authorized persons may complete CRFs. CRFs shall be signed by investigators as specified on the Delegated Tasks List included in the Investigator Site File. The investigator must retain the Investigator Site File, patient data sources and CRFs in accordance with local law and regulations or for at least two years after closure of the study, whichever is longer. The investigator should take measures to prevent accidental or early destruction of the study related materials. 8.2. PATIENT CONFIDENTIALITY The investigators and institutions involved in this study will provide direct access to source data and documents only to the sponsor and appropriate authorities for the purposes of monitoring, audit, Ethics Committee review or regulatory inspection. Each subject taking part in the study will have agreed explicitly to such access by signing the informed consent. The sponsor will ensure that no subject will be identifiable either from the final report or published results. 9. ETHICS This clinical study was designed and shall be implemented and reported in accordance with the ISO 14155 (2011), with applicable local regulations, and with the ethical principles laid down in the Declaration of Helsinki. 9.1. QUALITY CONTROL AND QUALITY ASSURANCE 10. STATISTICS 10.1. ANALYSIS PATIENT SETS 10.2. SAMPLE SIZE CALCULATION 10.3. DEMOGRAPHIC AND BASELINE CHARACTERISTICS 10.4. ANALYSIS OF PERFORMANCE 10.5. ANALYSIS OF SAFETY 11. FINANCING AND INSURANCE 12. SPONSORSHIP AND INDEMNITY 13. PUBLICATION POLICY 14. REFERENCES 15. APPENDIX APPENDIX A - Schedule of Study Assessments