Supporting Information
advertisement

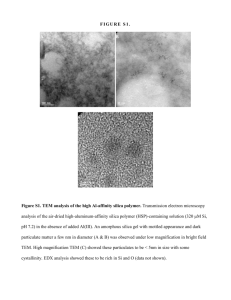
Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2012. Supporting Information for Adv. Funct. Mater., DOI: 10.1002/adfm.201202323 Hierarchical Assembly of Complex Block Copolymer Nanoparticles into Multicompartment Superstructures through Tunable Interparticle Associations Jiahua Zhu, Shiyi Zhang, Fuwu Zhang, Karen L. Wooley,* and Darrin J. Pochan* Submitted to Supporting Information for DOI: 10.1002/adfm.201202323 Hierarchical assembly of complex block copolymer nanoparticles into multicompartment superstructures through tunable inter-particle associations Jiahua Zhu1,‡, Shiyi Zhang2,3,‡, Fuwu Zhang2, Karen L. Wooley2*, Darrin J. Pochan1* 1 Department of Materials Science and Engineering, University of Delaware 2 Departments of Chemistry and Chemical Engineering, Texas A&M University 3 Department of Chemistry, Washington University in St. Louis 1. Experimental section Materials Poly(acrylic acid)90-b-poly(methyl methacrylate)100 (PAA90-b-PMMA100) diblock copolymers were synthesized by acidolysis of PtBA90-b-PMMA100 (PDI<1.20) precursors, which were prepared by sequential polymerization of methyl methacrylate and tert-butyl acrylate via atom transfer radical polymerization (ATRP).[1] IR: 3100-2900, 1750-1620, 1447, 1270-1080 cm−1. 1H NMR (DMF-d7, ppm): δ 3.92-3.75 (s, -OCH3), 2.76–1.26 (br,-CHCH2- of the polymer backbone), 1.23-0.91 (s, CH3). 13C NMR (DMF-d7, ppm): δ 177.8, 176.4, 54.4-51.7, 44.6-41.5, 36.7-35.5. DSC: (Tg)PMMA = 119.5 °C, (Tg)PAA = 133.5 °C. TGA in N2: 200–330 °C, 20% mass loss; 330–450 °C, 64% mass loss, 10% mass remaining above 450 °C. Poly(18-crown-6-g-acrylic acid)90-b-poly(methyl methacrylate)100 (P(crown0.4-g-AA0.6)90-b-PMMA100) diblock copolymers were synthesized by amidation of PAA90-b-PMMA100 with commercially-available 2-aminomethyl-18-crown-6. EDCI, 1-HOBt and 2-aminomethyl-18-crown-6 were used (0.45 equivalent to acid unit) and the mixture was allowed to undergo reaction for 24 hours. After dialysis for three days to remove byproducts and unreacted compounds, the white powder-like product was obtained from lyophilization with a 85% yield. 1H NMR confirmed that the conjugation efficiency was about 90%. IR: 3650-3020, 3020-2780, 1728, 1643, 1558, 1450, 1242, 1103, 964 cm−1. 1H NMR (DMF-d7, ppm): δ 3.92-2.98 (br,-OCH2CH2O- and -OCH3), 2.76–1.26 (br,-CHCH2- of the polymer backbone), 1.23-0.91 (s, CH3). 13C NMR (DMF-d7, ppm): δ 177.8, 176.4, 174.9, 72.0-67.4, 54.6-51.7, 44.6-41.3, 36.9-35.4. DSC: (Tg)PMMA = 120.8 , (Tg)crown-g-PAA = 157.2 . TGA in N2: 310–410 °C, 47% mass loss; 410–500 °C, 12% mass loss, 35% mass remaining above 500 °C. 1 Submitted to Poly(acrylic acid)75-b-polybutadiene104 (PAA75-b-PB104) diblock copolymers PAA-b-PB diblock copolymer was purchased from Polymer Source, Inc. and used as received. PDI = 1.10. 2,2’-(Ethylenedioxy)bis(ethylamine) (EDDA) was purchased from Sigma-Aldrich Corporation. Dimethylformamide (DMF) was purchased from Acros Organics. 2. Sample preparation and characterizations Complex nanoparticle assembly PAA90-b-PMMA100 or P(crown0.4-g-AA0.6)90-b-PMMA100 and PAA75-b-PS104 diblock copolymers at a certain blending ratio were first dissolved in DMF to produce a 0.1 weight percent solution of block copolymer. Fast water addition was accomplished by simply pipetting a certain amount of water into the polymer-DMF solution. After addition of a certain amount of diamine EDDA, sample solutions were sealed in vials at ambient condition before characterization. The time duration was recorded as aging time. Transmission electron microscopy (TEM) TEM imaging was performed on a Tecnai 12 microscope operating at an accelerating voltage of 120 kV. TEM samples were prepared by applying a drop of polymer solution (about 2–4 ul) onto a carbon-coated copper TEM grid and allowing the solvents to evaporate under ambient conditions. Images were collected on a Gatan CCD. A vapor staining method was applied to TEM samples. Dried TEM grids were placed on a 25x75x1 mm glass slide to sit in a sealed 100 mL glass bottle suspended above 1 mL 4 wt% osmium tetroxide aqueous solution (purchased from Electron Microscopy Sciences). OsO4 vapor staining process lasted from 6 hours to 24 hours for different samples. 1 H nuclear magnetic resonance (1H NMR) 1 H NMR spectra were recorded on an Inova 500 MHz spectrometer interfaced to a UNIX computer using VnmrJ software. Chemical shifts were referenced to the internal 3-trimethylsilylpropionate-d6 (TSP). The chemical shift of TSP was reported to be pH dependent.[2] The chemical shifts of TSP were referenced to the internal acetone, and showed no change over the pH range of the entire experiment. Solution A was 0.4630 g of EDDA and 0.6020 g of acetic acid dissolved in 20 mL of D2O. The molar ratio of amine to acid was 2.5 : 4, which was the final ratio in the polymer self-assembly condition. Solution B was 0.3320 g of 18-crown-6 dissolved in 10 mL of D2O. In the same volume of respective solutions, the molar ratio of acetic acid to 18-crown-6 was 4:1, which was the ratio in the polymer self-assembly condition. Solution C was 0.938 g of potassium chloride dissolved in 10 mL of D2O. In the same volume of respective solutions, the molar ratio of KCl to 18-crown-6 was 1:1. The chemical shift of (CH2OCH2CH2NH3+)2 was 3.220 ppm in solution A. The changes of this chemical shift were recorded as evidence of complexation and dissociation. Solution A and solution B were mixed in the ratios of 1:0.25, 1:0.5, 1:0.75, 1:1, 1:1.5, 1:2; 1:2.5; 1:3, 1:4, 1:5, 1:6, among which the ratio of 1:1 mimicked the ratio of EDDA, PAA and crown ether present in the polymer self-assembly condition (Figure 1). Solution A, solution B and 2 Submitted to solution C were mixed in the ratios of 1:1:0.5, 1:1:1, 1:1:2, 1:1:3, 1:1:4, 1:1:5, among which the ratio of 1:1:2 mimicked the ratio of the addition of KCl to trigger the dissociation. 3. Results and discussion Figure S1. TEM image showing the co-existing nanoparticle chains and rings with dispersed lengths and sizes from P(crown0.4-g-AA0.6)90-b-PMMA100 and PAA75-b-PB104 mixture with added EDDA diamine (amine to PAA acid and crown ether side chain) molar ratio = 0.5:1.0). Block copolymer mixing molar ratios (PMMA to PB volume ratio) are 1:1.5 (1:1). Samples were aged for 1 day and stained by OsO4 before imaging. 1 Dayananda, K.; Dhamodharanm R. Journal of Polymer Science: Part A: Polymer Chemistry, 2004, 42, 902-915 2 Wishart, D. S.; Bigam, C. G.; Yao, J.; Abildgaard, F.; Dyson, H. J.; Oldfield, E.; Markley, J. L.; Sykes, B. D. Journal of Biomolecular NMR 1995, 6, 135-14 3