CHEMISTRY 111 LABORATORY
advertisement

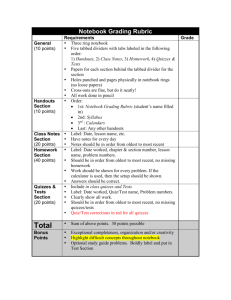
CHEMISTRY 111 LABORATORY Dr. Stuart Gentry gentry@lasalle.edu 215-951-1259 Holroyd 329 Office Hours: MWF 1 – 2pm, or drop-in as available Course materials and quizzes: Available on Canvas Credit: 4 credit hours (lecture + lab combined) INTRODUCTION This laboratory course is designed to complement and supplement your lecture course. Students will work in pairs. Your laboratory grade will constitute 25% of your final course grade. Students must separately pass both the lecture and lab portion to pass the combined course. SAFETY Safety is critical. Therefore safety glasses must be worn at all times. This includes sitting at the lab bench after completing the lab if other groups nearby are still working with chemicals. Failure to do so will result in your immediate expulsion from the laboratory and a grade of zero will be recorded as a lab grade for that experiment. TEXT Chemistry 111 Laboratory Manual (obtained from Chemistry Storeroom, Holroyd Rm 324) GRADING Your laboratory grade will be based on the following criteria: Lab Reports (80%) Lab Quizzes (10%) Lab Notebook / Technique / Safety (10%) MISSED LABS There will be no make-up of missed labs beyond one week after the lab was to be done. If you must make up a lab, you must contact Dr. Gentry to find a time within that week to make it up. This is only by special approval, and does not include labs missed because of studying for other classes or other non-emergency or non-illness reasons. LAB REPORTS Lab reports are due the following class period. 20% will be taken off the grade if turned in 1 week late. An additional 20% will be taken off if turned in 2 weeks late. Reports will not be accepted more than 3 weeks after the due date. Students will not receive credit for laboratory reports that have not been submitted by this time. Students will work in pairs BUT EACH STUDENT WILL SUBMIT AN INDEPENDENT REPORT. (Students who submit identical lab reports are guilty of plagiarism and the grade will be reduced accordingly. See below) PLAGIARISM To repeat what is written above, the lab report needs to be prepared individually. You are allowed to share raw data and you can talk about how to analyze the results or what they mean. But what goes in the written lab report in the way of calculations, answers to questions, and discussion must be IN THE WORDS OF THE INDIVIDUAL STUDENT. Any deviation from this is considered plagiarism and will be dealt with accordingly. This is true for both copee and copier, since it is not possible to know who wrote the initial report and who copied from it. Please refer to the university Academic Integrity policy for further information as well as the Student Guide to Rights and Responsibilities.. LAB QUIZZES It is of utmost importance that you be prepared when entering the laboratory for both your safety and the safety of those around you. In order to test your readiness for the upcoming experiment, a 10-question pre-lab quiz will be assigned. The quiz can be accessed through the course Blackboard page. The quiz must be completed before you come to lab. You will not be allowed to submit the quiz late since that circumvents the purpose of having you take the quiz. LAB NOTEBOOK This book should be a bound composition book or a spiral-bound notebook. (No 3-ring binders) ALL observations, calculations, in-lab discussions are to be included in this book. This book must be kept by each student AND initialed by your lab instructor at the end of the lab. Draw a line through any incorrect entries and place the correct entries next to them. The intent of a lab notebook is that is a real-time documentation of observations and results during the actual course of you doing the experiments. It is not something that is written up later or “prettied up” back in your dorm room. The entries should be complete enough that you could pick up the lab notebook 6 months later and know what the entries referred to. The lab notebook will not be graded on penmanship, it is after all a working lab document. But it needs to be structured enough to follow the train of work that was done in the lab on any given day. STUDENTS WITH DISABILITIES Students with disabilities should refer to the student handbook for resources that are available to them as well as compliance with the American Disabilities Act. SYLLABUS CHANGE POLICY This syllabus is a guide and every attempt is made to provide an accurate overview of the course. However, circumstances and events may make it necessary for the instructor to modify the syllabus during the semester and may depend, in part, on the progress, needs, and experiences of the students. The instructor will give notice when changes to the syllabus are made. General Rubric for Grading Lab Reports (see sample lab report in Chem. 111 lab manual) I) Objective (~5%) One to two sentence statement of objective of experiment. Should be more than stating the measurements to be made – what is the science or the theory that is being explored in the lab. II) Introduction (~10%) Discuss the chemistry that is to be explored in the chemistry. Include key concepts. III) Procedure (~5%) Refer to procedure in lab manual, then provide any differences from published procedure. IV) Experimental Results (~30%) Data table Graphs as appropriate - Axes labeled - Scales fully utilized (data fill the graph, not just in one corner) - Title on graph to know which set of data the graph refers to Data expressed with appropriate significant figures and units Include any qualitative observations Sample calculations included for each type of calculation in report (can be attached at end) V) Conclusions (~40%) All individual questions from lab report addressed - Must include both in-lab and post-lab questions - Include question as well as answer - Top marks only given if expand on answer and provide answer as a complete paragraph. Don’t just give one sentence answer like you would on an exam or homework. This is a report and as such answer should be a complete thought. Data compared to expected results (literature values) with % error given. Summary table required This not a repeat of earlier raw data, but may include a repeat of calculated values. Should include final experimental numbers as compared to literature values. Purpose is to help the reader understand what you learned, not just what you measured. Discuss possible sources of errors (not just human error) and possible improvements Overall conclusions *** This last item is critical, don’t just stop with the results. Instead must comment on what the results mean VI) Overall Impact of Report (~10%) Neatly typed or printed Data pages taken straight out of lab manual will not get top marks Correct spelling Evidence that time and thought was spent on preparing the report. CHEMISTRY 111 Laboratory Schedule Fall, 2014 Experiment Day M Tu W Th 8/25 8/26 8/27 8/28 ‒ 9/2 9/3 9/4 Physical Measurements / Introduction to Vernier Software 9/8 9/9 9/10 9/11 Chromatographic Separation 9/15 9/16 9/17 9/18 Percent Composition 9/22 9/23 9/24 9/25 Chemical Reactions of Copper 9/29 9/30 10/1 10/2 Solution Stoichiometry 10/6 10/7 10/8 10/9 Limiting Reagents 10/13 10/14 10/15 10/16 Analysis of an Iron Supplement ‒ 10/27 ‒ 10/28 10/22 10/23 Introduction to Spectroscopy ‒ 11/3 ‒ 11/4 10/29 10/30 Molecular Bonding and Shapes ‒ 11/10 ‒ 11/11 11/5 11/6 Heat of Combustion ‒ 11/17 ‒ 11/18 11/12 11/13 Enthalpy of Reaction ‒ 11/24 ‒ 11/25 11/19 ‒ 11/20 ‒ Math & Significant Figures Review Safety and Check-in (Fall Break) (Thanksgiving) Check Out 12/1 12/2 12/3 12/4