Enzyme Catalytic Strategies: Chymotrypsin & Mechanisms

BIOC 460, Spring 2008
Lectures 13-14
Enzymes: Catalytic Strategies
Reading: Berg, Tymoczko & Stryer, 6th ed.,
Chapter 9, pp. 241-254
hexokinase conformational change (Jmol): http://www.biochem.arizona.edu/classes/bioc462/462a/jmol/hexokinase/newhk.html
movie of chemical mechanism of serine proteases (from Voet & Voet,
Biochemistry , 3rd ed., 2004, Wiley): http://www.biochem.arizona.edu/classes/bioc460/spring/460web/lectures/LEC13-
14_EnzCatMech/15-3c_SerineProtease-b3/SerineProtease.htm
Serine proteases (Jmol) http://www.biochem.arizona.edu/classes/bioc462/462a/jmol/serprot/serprot1.htm
Key Concepts
• Mechanisms used by enzymes to enhance reaction rates include:
(1st 4 mechanisms based on BINDING of substrate and/or transition state)
1. Proximity & orientation
2. Desolvation (one type of electrostatic catalysis)
3. Preferential binding of the transition state
4. Induced fit
5. General acid/base catalysis
6. Covalent (nucleophilic) catalysis
7. Metal ion catalysis
8. (Electrostatic catalysis)
• The chemical mechanism of serine proteases like chymotrypsin illustrates:
– Proximity and orientation
– Transition state stabilization
– Covalent catalysis, involving a “catalytic triad” of Asp, His and Ser in the active site
– general acid-base catalysis
– electrostatic catalysis
LECTURES 13-14, Enzymes - Catalytic
Strategies 1
BIOC 460, Spring 2008
Learning Objectives
• Discuss (briefly explain): 8 general catalytic mechanisms used by enzymes to increase the rates of chemical reactions. (You won't be asked on an exam to simply LIST them, but you could be expected to explain any one -or 2 or 3 or 4 -- of them.)
• Terminology: proteolysis, serine protease , general acid, general base, catalytic triad, acyl group, tetrahedral intermediate, acyl-enzyme intermediate, acylation, deacylation, nucleophile, oxyanion hole
• Explain why peptide bonds are kinetically stable in the absence of a catalyst, given that equilibrium lies far in the direction of hydrolysis in 55.5 M H
(Why is any specific reaction a slow reaction?)
2
O.
• Describe the chemical mechanism of hydrolysis of peptide bonds by chymotrypsin , including the following:
– What is the "job" of the catalyst (the protease), i.e., what group needs to be made more susceptible to nucleophilic attack?
– Describe substrate binding, including the role and chemical nature of the
"specificity pocket" in chymotrypsin, and which peptide bond in the substrate (relative to the specificity group) will be cleaved.
– Draw the structure of the catalytic triad at the beginning of the reaction, and explain how the states of ionization and hydrogen bonding pattern of those 3 groups change step by step during catalysis.
•
Learning Objectives, continued
(chemical mechanism of chymotrypsin, continued)
– Explain the role of each member of the catalytic triad in the reaction.
– Identify the nucleophile that attacks the carbonyl carbon in acylation; identify the nucleophile that attacks the carbonyl carbon in deacylation.
– Describe the acyl-enzyme intermediate , including identifying the type of bond attaching the acyl group to the enzyme (Is it an amide linkage? anhydride? ester? etc.) and how that acyl group relates to the structure of the original substrate.
– Draw the structures of each of the tetrahedral intermediates in the reaction. (If you can do this, you understand the chemistry by which they formed.)
– Identify the leaving group coming from each of the tetrahedral intermediates as the intermediate breaks down.
– State what is being acylated and deacylated in the chymotrypsin reaction (be specific about the functional group involved).
– Explain the role of the "oxyanion hole" in the mechanism.
– Describe which type(s) of general catalytic mechanisms (first learning objective above) are used by chymotrypsin, and how.
LECTURES 13-14, Enzymes - Catalytic
Strategies 2
BIOC 460, Spring 2008
•
•
•
Learning Objectives, continued
Compare (very briefly, just the “bottom line”) the overall 3-dimensional structures of chymotrypsin, trypsin, and elastase, and compare the substrate binding specificities of those 3 enzymes, explaining the relationship of the “specificity site/pocket” structure to the differences in substrate specificity..
How do 3 other classes of proteases (besides the serine proteases) generate nucleophiles potent enough to attack a peptide carbonyl group?
To which protease class does HIV protease belong? Describe the quaternary structure and symmetry of the HIV protease and where in the quaternary structure the active site residues are located.
General Catalytic Mechanisms
• Different enzymes use different combinations of mechanisms to reduce activation energy and thus increase rate of reaction.
• 7 (or 8) "types" of mechanisms below -- really "overlapping" concepts in many cases.
• 1st 4 mechanisms related to BINDING of substrate and/or transition state,
(reaction takes place in active site, not in bulk solution)
1. PROXIMITY AND ORIENTATION (catalysis by “approximation”)
– Proximity
• Reaction between bound molecules doesn't require an improbable collision of 2 molecules.
• They're already in "contact" (increases local concentration of reactants).
– Orientation
• Reactants are not only near each other on enzyme, they're oriented in optimal position to react.
• The improbability of colliding in correct orientation is taken care of.
LECTURES 13-14, Enzymes - Catalytic
Strategies 3
BIOC 460, Spring 2008
General Catalytic Mechanisms, continued
2. DESOLVATION
• Active site gets reactants out of H
2
O.
• Lower dielectric constant environment than H environment), so stronger electrostatic interactions (strength inversely related to dielectric constant).
2
O (more nonpolar
• Reactive groups of reactants are protected from H
2 compete with reactants.
O, so H
2
O doesn't
– H
2
O won't react to give unwanted byproducts, e.g., by hydrolysis of some reactive intermediate in the reaction that was supposed to transfer its reactive group to another substrate.
3. TIGHT TRANSITION STATE BINDING
• used to be called "strain and distortion"
• Enzyme binds transition state very tightly, tighter than substrate.
• Free energy of transition state (peak of free energy barrier on reaction diagram) is lowered because its "distortion" (electrostatic or structural) is
"paid for" by tighter binding of transition state than of substrate.
4. INDUCED FIT
• Conformational change resulting from substrate binding
• Binding may stabilize different conformation of enzyme or substrate or both.
• Conformational change
– orients catalytic groups on enzyme,
– promotes tighter transition state binding, and/or
– excludes H
2
O (obviously related to tight transition state binding, too )
• Example: hexokinase, binding of glucose (1st reaction in glycolysis)
– Ends of the U-shaped enzyme hexokinase pinch toward each other -conformational change induced by binding D-glucose (red)
– hexokinase conformational change (jmol)
“Open” conformation (no glucose) “Closed” conformation ( glucose bound)
LECTURES 13-14, Enzymes - Catalytic
Strategies
Hexokinase, induced fit
Nelson & Cox, Lehninger
Principles of Biochemistry ,
4th ed. Fig. 8-21
4
BIOC 460, Spring 2008
• 1st 4 concepts (above) in catalysis rather general, all related to BINDING of substrate and/or transition state
• Reaction takes place in active site, not in bulk solution.
• Other catalytic mechanisms (below) involve specific groups and chemical mechanisms that depend on the specific reaction .
5. GENERAL ACID-BASE CATALYSIS
• Specific functional groups in enzyme structure positioned to
– donate a proton (act as a general acid ), or
– accept a proton (act as a general base )
• helps enzyme avoid unstable charged intermediates in reaction
• Group that donates a proton (acts as a general acid) in catalysis has to then accept a proton (act as a general base) later in catalytic mechanism for catalyst to be regenerated in its original conjugate acid form.
• Likewise, general base that accepts a proton must give it up later.
• Amino acid functional groups that can act as general acids/general bases:
– His imidazole
– α -amino group
– thiol of Cys
– R group carboxyls of Glu, Asp
– ε -amino group of Lys
– aromatic OH of Tyr
– guanidino group of Arg
6. COVALENT CATALYSIS
• rate acceleration by transient formation of a COVALENT catalystsubstrate bond
• Covalent intermediate is more reactive in next step in reaction , so that step has lower activation energy than it would have for a noncovalent catalytic mechanism -- enzyme alters pathway to get to product .
• Nucleophile : an electron-rich group that attacks nuclei
– examples of nucleophiles among protein functional groups:
• unprotonated His imidazole
• unprotonated α -amino group
• unprotonated ε -amino group of Lys
• unprotonated thiol (thiolate anion, -S – ) of Cys
• aliphatic -OH of Ser
• unprotonated R group (carboxylates) of Glu, Asp
– some coenzymes, e.g., thiamine pyrophosphate (TPP) & pyridoxal phosphate (PLP)
LECTURES 13-14, Enzymes - Catalytic
Strategies 5
BIOC 460, Spring 2008
•
•
7. METAL ION CATALYSIS (several catalytic roles)
Metal ions can be
– tightly bound (metalloenzymes), i.e., as a prosthetic group
(usually transition metal ions, e.g., Fe 2+ or Fe 3+ , Zn 2+ , Cu 2+ , Mn 2+ ....)
– loosely bound , binding reversibly and dissociating from enzyme
(usually Na + , K + , Mg 2+ , Ca 2+ ...)
Functions of metal ions in catalysis:
A.
Binding and orientation of substrate (ionic interactions with negatively charged substrate)
B.
Redox reactions (e.g., Fe 2+ / Fe 3+ in some enzymes)
C. Shielding or stabilizing negative charges on substrate or on transition state (electrophilic catalysis)
– example: Enzymes that bind ATP (adenosine triphosphate) require
Mg 2+ to be bound to nucleotide (so ligand is actually Mg 2+ •ATP) in order to
• shield negative charges, and
• orient the ATP substrate
– Example: Kinases (enzymes catalyzing phosphoryl transfer involving
ATP or other nucleoside triphosphates)
– All kinases require Mg 2+ for activity, but it's actually in complex with nucleotide (usually ATP).
– example: hexokinase (transfer of terminal phosphate group from
ATP to glucose, producing glucose-6-P and ADP as products)
•
•
[8. ELECTROSTATIC EFFECTS] concept not always "listed" separately because it’s involved in many other aspects of catalytic mechanisms
Some examples:
– providing lower dielectric constant of environment in active site
(hydrophobic environment)
– altering pK values of specific functional groups
– stabilizing a particular conformation of critical groups in active site by electrostatic interactions
– stabilizing (binding) a charged intermediate or transition state by providing an oppositely charged enzyme group nearby.
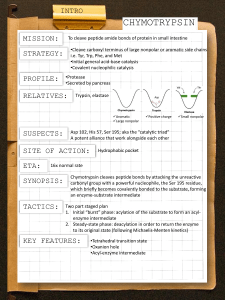
Enzyme Chemical Mechanisms (Chymotrypsin as an example)
• digestive protease
• synth. in mammalian pancreas
• secreted in inactive form as single
polypeptide chain (chymotrypsinogen)
• activated by proteolytic processing to
rearrange conformation to active enzyme,
which because of the activating peptide
bond "clips" has 3 chains
• structure stabilized by disulfide bonds
(true for many extracellular proteins)
Berg et al., Fig. 9-6
LECTURES 13-14, Enzymes - Catalytic
Strategies 6
BIOC 460, Spring 2008
PROTEASES
• Reaction catalyzed = hydrolysis of peptide bonds
• in vivo functions
• digestion of nutrient protein
• protein turnover (degradation of proteins that are old or no longer needed as conditions change)
• specific proteolytic “clips” for activation of enzymes (e.g., developmental sequence, or signal for special event like blood clotting)
• Peptide bond hydrolysis (S
N
peptide bond)
2 attack by : O of water on carbonyl C of the
• Equilibrium (in 55.5 M H
2
O) lies FAR to the right, but in absence of catalyst,
reaction is extremely slow (fortunately -- or our bodies would all be puddles
of amino acids in solution!)
• Peptide bonds "kinetically stable"
Mechanism of Peptide Bond Hydrolysis
• Mechanism of uncatalyzed reaction: simple nucleophilic attack by : O of H forming tetrahedral intermediate
2
O on carbonyl C of peptide bond,
• Tetrahedral intermediate then breaks down as the amine "half" of original peptide leaves.
• Reason uncatalyzed reaction is so slow: partial double bond character of peptide bond makes carbonyl carbon much less reactive than carbonyl carbons in carboxylate esters.
• Catalytic task of proteases is to make that normally unreactive carbonyl group more susceptible to nucleophilic attack by H
2
O .
LECTURES 13-14, Enzymes - Catalytic
Strategies 7
BIOC 460, Spring 2008
4 Classes of Proteases
• 4 classes of proteases based on different mechanisms to enhance the susceptibility of the carbonyl group to nucleophilic attack
1. Serine proteases (e.g., chymotrypsin) -- covalent catalysis , with initial nucleophilic attack carried out by enzyme Ser-O (H) group made into a potent nucleophile with assistance of nearby His imidazole that acts as a general base
2. Cys proteases -- again, covalent catalysis , with initial nucleophilic attack carried out by an enzyme Cys-S (H) group made into a potent nucleophile with assistance of nearby His imidazole that acts as a general base
3. Asp proteases -- nucleophile is HOH itself, assisted by 2 Asp residues, general base catalysis by 1st Asp carboxyl group and orientation/polarization of substrate carbonyl by 2nd Asp residue
4. Metalloproteases -- again, nucleophile is HOH , but assisted by binding to a metal (e.g. Zn 2+ ) and by general base catalysis by some enzyme base group, e.g. Glu-COO – .
detailed look at a serine protease, chymotrypsin
• Chymotrypsin makes carbonyl C of peptide bond more reactive by changing pathway of reaction.
• Covalent catalysis by Ser residue, with assistance of a general base (His)
• Overall reaction: 2 separate "half reactions" (2 "phases" of catalysis), with a metastable covalent intermediate ("acyl-enzyme intermediate") between the 2 half reactions.
• Overall chemical steps in the 2nd phase are almost an exact repeat of steps in the first phase.
What’s an “acyl group”?
LECTURES 13-14, Enzymes - Catalytic
Strategies 8
BIOC 460, Spring 2008
Overview of chymotrypsin mechanism: 2 half reactions
• First step/phase ("acylation")
– Enzyme provides potent nucleophile , a specific Ser O (H) group.
– Ser OH made more nucleophilic than usual with assistance of nearby
His residue as general base
– Nucleophilic attack on substrate --> covalent intermediate, the acyl enzyme intermediate (actually a carboxylate ester of carboxylate
"half" of original substrate, attached to enzyme's Ser R group that's the alcohol component of the ester).
– Amine "half" of original peptide/protein released as product (P1) at end of first phase.
Berg et al., Fig. 9-5
Overview of chymotrypsin mechanism: 2 half reactions
• Second phase ("deacylation")
– 2nd substrate, H
2
O, is nucleophile, attacking carbonyl C of the carboxylate ester of acyl enzyme, again with assistance of active site
His residue as general base.
– Ester bond of intermediate is hydrolyzed to regenerate alcohol component (the enzyme chymotrypsin, with its Ser-OH free again) and carboxylic acid component, the 2nd product (P2) (carboxyl "half" of original substrate peptide/protein).
Berg et al., Fig. 9-5
LECTURES 13-14, Enzymes - Catalytic
Strategies 9
BIOC 460, Spring 2008
THE CATALYTIC TRIAD
• 3 amino acid residues in active site in a hydrogen-bonded network:
– Ser (residue #195)
– His (residue #57)
– Asp (residue #102)
• essential for effective catalytic activity in chymotrypsin
• Catalytic triad action converts OH group of Ser 195 into a potent nucleophile.
Berg et al., Fig. 9-7
Movie of chemical mechanism of chymotrypsin: http://www.biochem.arizona.edu/classes/bioc460/spring/460web/lectures/LEC13-
14_EnzCatMech/15-3c_SerineProtease-b3/SerineProtease.htm
Whole Chymotrypsin Mechanism
(Berg et al., Fig. 9-8)
LECTURES 13-14, Enzymes - Catalytic
Strategies 10
BIOC 460, Spring 2008
FIRST PHASE OF CATALYSIS (PHASE I, ACYLATION )
• Formation of acyl-enzyme covalent intermediate and generation of the amine product
1.
Formation of ES Complex
• Enzyme binds substrate (peptide or
protein), with aromatic bulky
hydrophobic side chain "specificity
group" in "pocket”.
• Bound susbstrate is positioned for
the peptide bond on carbonyl side
(i.e., "carboxyl" side) of that residue
to be cleaved.
• Polypeptide chain of substrate also
forms a short β -sheet (hydrogen
bonds) with a β strand of enzyme in
binding site.
Berg et al., Fig. 9-8, Step 1
2. Formation of 1st Tetrahedral Intermediate (the chemistry begins)
• Oxygen atom of active site Ser-OH activated by hydrogen bond to His
(imidazole ring N: ) in catalytic triad
• His accepts proton from Ser-O H ( general base catalysis), to become
His H +
• Ser-O ( – ) (potent nucleophile) carries out nucleophilic attack on carbonyl C of substrate (nucleophilic catalysis, i.e. covalent catalysis) --> COVALENT bond to carbonyl C (1st tetrahedral intermediate) .
[Proton transfer and nucleophilic attack may be concerted (occur at same time), so Ser-O may not actually exist as an oxyanion/alkoxide ion (-O – )].
• Asp in catalytic triad : a) helps maintain perfect orientation of His and Ser residues in hydrogen bonded network, and b) facilitates H + transfer by electrostatic stabilization of HisH + after it has accepted the proton.
Berg et al., Fig. 9-8, Step 2
LECTURES 13-14, Enzymes - Catalytic
Strategies 11
BIOC 460, Spring 2008
Transition State Stabilization, the “Oxyanion Hole”
• Product of step 2 (nucleophilic attack in acylation half-reaction) = FIRST
TETRAHEDRAL INTERMEDIATE
• structure presumed similar to that of transition state for its formation and breakdown, with negatively charged "carbonyl" oxygen (not a carbonyl group anymore), an OXYANION
•The "oxyanion hole", an area in the
active site of serine proteases that binds
the transition state particularly tightly.
•Active site binds oxyanion more tightly
than it bound original carbonyl group of
the substrate.
• An additional hydrogen bond forms
between tetrahedral oxyanion and
enzyme groups around it (which form
"oxyanion hole" portion of active site)
•That hydrogen bond couldn't form to
carbonyl oxygen (=O) but can form now,
because of structural change
(lengthening of C–O bond) on forming tetrahedral intermediate .
•Also, hydrogen bonds to negatively
charged oxygen are stronger than to
neutral O.
Berg et al., Fig. 9.9
3. Formation of Acyl-Enzyme Intermediate
• First tetrahedral intermediate breaks down -- original amide (peptide) bond cleaves
• His H + donates a proton to the amino "half" of the original substrate ( His H + acts as general acid) to generate R
2
-NH
2
.
• Breaking of amide bond (departure of amine product) --> conversion of oxyanion back into a C=O, still covalently attached to Ser residue of enzyme, forming acyl-enzyme intermediate .
Berg et al., Fig. 9-8, Step 3
LECTURES 13-14, Enzymes - Catalytic
Strategies 12
BIOC 460, Spring 2008
4. Amine Product (R
2
-NH
2
(1st product leaves).
) dissociates from active site
• Breaking of amide bond
(departure of amine product) --> conversion of oxyanion back into a C=O, still covalently attached to
Ser residue of enzyme, forming acyl-enzyme intermediate .
• Original carbonyl group of peptide bond is now a carbonyl group again, but it's covalently attached to the Ser-O in the acyl-enzyme product of first half reaction
(acylation phase).
• What kind of linkage
does acyl-enzyme
have between
"carboxyl half"
of original peptide
substrate and O from
the Ser alcohol R
group?
Berg et al., Fig. 9-8, Step 4
SECOND PHASE OF CATALYSIS (PHASE II, DEACYLATION )
• Breakdown of acyl-enzyme (covalent intermediate) by reaction with
H O (HYDROLYSIS) and release of the carboxylic acid product
2
• This phase (second "half reaction") is almost an exact repeat of the first in terms of catalytic steps/mechanisms
5. Binding of Second Substrate,
H
2
O, in Active Site • nucleophilic attack facilitated by HisN:
acting as general base (but nucleophile is H
2
O , attacking
carbonyl C of acyl-enzyme )
• formation of second tetrahedral
intermediate with transition state
stabilization by binding of C-O – in
oxyanion hole
• breakdown of 2nd tetrahedral
intermediate (cleavage of what had
been ester bond in acyl-enzyme
intermediate, with HisH + as general
acid catalyst) to regenerate enzyme
Ser-OH and release the carboxylic
acid product (from the original
peptide/protein substrate).
Berg et al., Fig. 9-8, Step 5
LECTURES 13-14, Enzymes - Catalytic
Strategies 13
BIOC 460, Spring 2008
6. Formation of the Second Tetrahedral Intermediate
• HOH forms hydrogen bond with HisN: in catalytic triad (just like Ser-OH did in first phase).
• His again acts as a general base , to become HisH + , activating O from H
2 to make it a potent nucleophile, to attack carbonyl C of acyl-enzyme intermediate (an ester).
O
• nucleophilic attack of HOH on carbonyl C of acyl-enzyme intermediate
(nucleophilic catalysis, i.e. covalent catalysis) → COVALENT bond between OH of water and carbonyl C --> 2nd tetrhedral intermediate).
• Asp in catalytic triad : a) helps maintain perfect orientation of catalytic triad, and b) facilitates H + transfer by electrostatic stabilization of His H + after it has accepted the proton.
Berg et al., Fig. 9-8, Step 6
Transition State Stabilization 2, the “oxyanion hole” again
• Product of step 6 (nucleophilic attack in deacylation half-reaction) =
SECOND TETRAHEDRAL INTERMEDIATE
• structure presumed similar to that of transition state for its formation and breakdown, with negatively charged "carbonyl" oxygen (not a carbonyl group anymore), an OXYANION
• Note that SECOND tetrahedral intermediate has an -OH group on it (from
H
2
O) instead of amido group of amine "half" of original substrate as in
FIRST tetrahedral intermediate.
• Otherwise, 2nd TI binding in
oxyanion hole is similar to
binding of first TI, with extra
hydrogen bond, etc.
• Again, oxyanion hole is
presumed to be stabilizing transition states for formation
and breakdown of 2nd
tetrahedral intermediate by
binding them tightly.
LECTURES 13-14, Enzymes - Catalytic
Strategies 14
BIOC 460, Spring 2008
7. Breakdown of 2nd Tetrahedral Intermediate:
Original ester bond (from acyl-enzyme) CLEAVES.
• His H + ( general acid ) donates proton back to Ser O, generating alcohol product of hydrolysis of acyl-enzyme, Ser-O H
• Ester bond from acyl-enzyme intermediate breaks --> carboxylic acid product ( R
1
-COOH ) from original substrate.
Berg et al., Fig. 9-8, Step 7
8. Carboxylic acid product dissociates from active site.
• Enzyme molecule now in its original state, with His imidazole in neutral form, catalytic triad appropriately hydrogen-bonded, and active site ready to bind another molecule of substrate and do it all again.
Does hydrolysis occur in the acylation or deacylation half
reaction of serine proteases?
What is the nucleophile in the acylation half reaction?
What is the nucleophile in the deacylation half reaction?
Berg et al., Fig. 9-8, Step 8
LECTURES 13-14, Enzymes - Catalytic
Strategies 15
BIOC 460, Spring 2008
Mammalian Serine Proteases are Homologous .
• 3-dimensional folds (tertiary structures) of chymotrypsin (red) and trypsin
(blue) . (Only the backbone tracings of
α
carbon positions are shown.)
• family of mammalian serine proteases:
– e.g., chymotrypsin, trypsin & elastase
• obviously homologous (share a
common evolutionary ancestral
gene) -- primary structures about
40% identical and 3-dimensional
folds nearly identical
• suggests common evolutionary
origin with a single ancestral gene
that duplicated a number of times,
after which sequences and substrate
specificities diverged (example of divergent evolution .)
• Family also includes many
proteolytic enzymes in the blood
clotting cascade .
Berg et al., Fig. 9-12
The hydrophobic “specificity pocket” of chymotrypsin
• area of active site responsible
for chymotrypsin’s substrate
specificity )
• Position of aromatic ring bound in pocket is shown in green in center.
• Note Gly residues in
“lining” of pocket (small, so
bulky, hydrophobic side chains
fit in binding site)
• Also note Ser 189 in bottom
of pocket. (This residue is
Asp in structure of trypsin .)
Berg et al., Fig. 9-10
LECTURES 13-14, Enzymes - Catalytic
Strategies 16
BIOC 460, Spring 2008
The specificity pockets of chymotrypsin, trypsin, and elastase
• Substrate binding sites on enzymes where "R1" group of the substrate binds
("R1" = R group of the amino acid residue contributing the carbonyl group of
the peptide bond to be cleaved).
• Trypsin cleaves peptide bonds on carbonyl side ("after") long + charged
residues (R1 = Lys + or Arg + )
•specificity "assisted" by Asp – residue in bottom of S1 site.
• Pocket of elastase is partly closed off so only small side chains may enter (Val
residues in stead of Gly residues).
Elastase cleaves after small neutral residues (e.g., Gly and Ala).
Berg et al., Fig. 9-13
Subtilisin: a bacterial protease
• no apparent evolutionary relationship to mammalian chymotrypsin/trypsin family (no primary structure or tertiary structural resemblance)
• same catalytic mechanism as mammalian serine proteases: a catalytic Ser assisted by a His and an Asp residue ( catalytic triad ), in same orientation, and an oxyanion hole to stabilize oxyanion intermediates (transition states)
• example of convergent evolution : independent evolution of same catalytic strategy
• (Chymotrypsin mechanism must be a very effective hydrolytic mechanism!)
LECTURES 13-14, Enzymes - Catalytic
Strategies
Berg et al., Fig. 9-14
17
BIOC 460, Spring 2008
site-directed mutagenesis studies on structural alterations in subtilisin active site
• gene for subtilisin was cloned
• specific mutations in the catalytic triad residues (individually and in combination) introduced by molecular biological methods
• mutant enzymes expressed, purified and studied
• Results: Mutations in catalytic triad residues have dramatic effects on
k cat
(turnover number) for subtilisin.
• Note log scale for k cat
about 10 6 !
cat
;
mutation of Ser or His
reduces k by a factor of
1st letter is normal (wild type) residue in that (number) position; 2nd letter is mutant amino acid residue replacing normal residue at that position.
Berg et al., Fig. 9-16
• However, also note that
what might seem to be
"fatally" modified
(mutated) enzymes still
have higher k
than k cat cat
values
for uncatalyzed
reaction, by factor of ~10 3 .
Activation strategies for 3 more classes of proteases
(besides Ser proteases)
• problem faced by proteases: activation of carbonyl C of peptide bond for attack by a nucleophile
• All generate a potent nucleophile to attack peptide carbonyl group.
– Cys proteases : nucleophile a Cys thiol activated by His (gen. base)
– Asp proteases : nucleophile is HOH itself assisted by 2 Asp residues: general base catalysis by 1 Asp carboxyl group and orientation/polarization of substrate carbonyl by 2nd Asp residue
– Metalloproteases : nucleophile is HOH assisted by binding to a metal
(e.g. Zn 2+ ) and by general base catalysis by some enzyme base group, e.g. Glu-COO – .
Berg et al., Fig. 9-18
LECTURES 13-14, Enzymes - Catalytic
Strategies 18
BIOC 460, Spring 2008
HIV protease: an Asp protease
• Homodimer : 2 identical subunits, each contributing an Asp to active site.
• 2 catalytic Asp residues , 1 from each subunit, on opposite sides of 2-fold axis of symmetry (below the bound crixivan in Fig. 9-21).
• Structure in Fig. 9.19 has substrate binding pocket indicated, with the 2 catalytic Asp residues in ball-and-stick structures.
• "Flaps" (a portion of each polypeptide chain, labeled) close down after substrate binds ( induced fit ).
• Structure shown in Fig. 9.21 is in complex with an inhibitor , crixivan , which has a conformation that approximates the 2-fold symmetry of the enzyme.
• Crixivan thus inhibits HIV protease without affecting normal cellular Asp proteases, which don't have the 2-fold symmetry that HIV protease has.
• Crixivan designed to mimic tetrahedral intermediate (transition state) -- it's a transition state analog, with groups to bind various sub-pockets in substrate binding site.
Berg et al., Fig. 9-19 Berg et al., Fig. 9-21
LECTURES 13-14, Enzymes - Catalytic
Strategies 19