John L. Stickney Department of Chemistry University of Georgia
advertisement

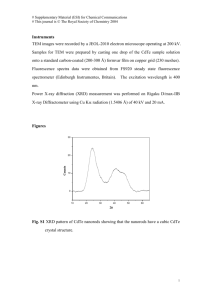
John L. Stickney Department of Chemistry University of Georgia Athens, GA 30602 Stickney@chem.uga.edu www.chem.uga.edu/stickney/ Acknowledgments: People Dr. Youn-Geun Kim Madhivanan Muthuvel (Madhi) Venkatram Venkatasamy (Ram) Steve Cox Jay Kim Nagrrajan Jayaraju (Nag) Dhego Banga Deepa Vairavapandian Chandru Thambidurai Professor Uwe Happek Moved on Dr. Raman Vaidyanathan Dr. Ken Mathe Dr. Nattapong Srisook (Natt) Funding NSF Materials and Chemistry NSF DMR NER nanoscale research DOE, Bio Fuels program University of Georgia Research Foundation I have a Dream That one day all ULSI structures will be grown from the bottom up, using solution phase reactions with atom level control. The electronic industries are already working on the 35 nm node, so lateral size control will be accomplished using Lithography. Vertical growth will be accomplished atomic layer by atomic layer. Ambient growth of metals and semiconductors has been demonstrated (electrodeposition in solution) using EC-ALE. Chemistries for the layer by layer growth of oxides and polymers are tractable Zinc Blende Atomic Layer Epitaxy (ALE): the use of surface limited reactions to form deposits an atomic layer at a time. Atomic Layer Deposition (ALD): Same as ALE, without emphasizing epitaxy. Electrochemical Atomic Layer Epitaxy (EC-ALE): The use of electrochemical surface limited reactions (UPD or SLR3) for ALE or ALD. • UPD: underpotential deposition Where an atomic layer of one element deposits on a second at a potential prior to that needed to deposit on itself • SLR3:Surface Limited Redox Replacement Reactions Use of UPD to form an atomic layer of a sacrificial metal to limit the deposition of a more noble metal by redox replacement at open circuit, to an atomic layer. Se4+ Se4+ EC-ALE Layer by layer underpotential deposition of compound semiconductors, performed at room temperature. Se4+ Se4+ Se4+ Se4+ Se4+ Au Pb2+ Pb2+ Pb2+ Pb2+ Pb2+ Pb2+ Pb2+ } One Cycle of PbSe Deposition of 200 cycles of CdTe: 0.25 mM TeO2, 10 mM Na2B4O7, 20 mM Na2SO4, E = -400 mM 0.25 mM TeO2, 10 mM Na2B4O7, 20 mM Na2SO4, E = -551 mM Ultrahigh Vacuum (UHV) System for UHV-EC x-y-z Manipulator AES LEED Main Chamber AnteChamber Sample Transporter Cryopump Ion Pump Electrochemical Cell Pictures of Ultrahigh Vacuum (UHV) Chamber and Antechamber 1.5 cycle, beginning with Cd, E= -550 mV CdTe 3 x 3 One cycle of CdTe formation: The bright areas are Cd UPD on Au Te followed by Cd. The darker areas are the CdTe too little Te in first step (0.33 ML). monolayer. 0.44 ML coverage of Te is needed to Cd stripping leaves the 1/3 coverage form a monolayer of CdTe Te structure, and pits where there was only Cd UPD on Au Compounds, Elements and Structures Formed Using EC-ALE II-VI CdS CdSe CdTe ZnS ZnSe ZnTe HgSe HgTe HgS III-V IV-VI GaAs? SnS? GaSb SnSe? InAs SnTe? InSb PbS PbSe PbTe Others In2Se3 CuSe Sb2Te3 Bi2Te3 Superlattices InAs/InSb PbSe/PbTe HgCdTe Metals: Pt, Cu Nanoclusters In2Se3 PbSe HgSe CdTe Compound Au GaAs GaSb InP InAs InSb ZnS ZnSe ZnTe CdS CdSe CdTe HgS HgSe HgTe PbS PbSe PbTe SnTe Band Gap, Lattice Constant, Interatomic eV nm Distance,nm 2.86 1.43 5.653 0.69 6.095 1.28 5.8687 0.36 6.058 0.17 6.4787 3.6-3.8 a 3.814, c 6.257 2.58 5.667 2.28 6.101 2.53 a 4.136, c 6.713 1.74 a 4.299, c 7.010 1.5 6.477 2.5 -0.15 6.086 0 6.42 0.37 5.936 0.26 6.124 0.29 6.46 0.18 6.328 Au Lattice Mismatch, % 4.29 3.997 4.31 4.15 4.284 4.581 3.814 3.823 4.314 4.136 4.299 4.58 6.83 -0.47 3.26 0.14 -6.78 11.10 10.89 -0.56 3.59 -0.21 -6.76 4.303 4.568 4.197 4.33 4.568 4.475 -0.30 -6.48 2.17 -0.93 -6.48 -4.31 Bohr Radius, nm 14 23.3 9.5 35.5 69 1.7 2.8 4.6 3.1 6.1 6.5 39.3 20 46 X-ray Diffraction Pattern for 100 Cycle Electrodeposited PbTe on Au. Rock salt structure, a = 0.646 nm. Angle of incidence is 1°. The deposit Is essentially all (200) oriented. Experimental method of measuring the absorption coefficient θB eliminates reflection off of sample surface Io I θB θB Io I }t Again, the band gap of even the 100 cycle PbTe deposit is blue shifted from its bulk value (0.29 eV) due to quantum confinement. The band gap of even the 50 cycle deposit is blue shifted from its bulk value (2100 cm-1) due to quantum confinement. PbSe energy gap data compared to the hyperbolic band model (Wang et al.) with theoretical film thickness multiplied by the coverage factor calculated for each film: Dotted line: using effective mass values of Dalven Solid line: using effective mass values of Kang et al. X-ray diffraction of 80 period 4PbTe / 4PbSe superlattice. Buffer layer is 10 cycle PbSe. Angle of incidence is 1°. (111) Period = 4.5 nm The band gap was found to be to be 0.48 eV. It is blue shifted from bulk value of PbSe (0.26 eV) and PbTe (0.29eV). Surface limited redox replacement reaction:SLR3 Replacement of Cu upd by Pt 320 nm×320 nm 105 nm×105 nm 260 nm×260 nm CV for the upd of Cu on Au(111) in 0.1M H2SO4 with 0.1M Cu2+, Cu upd adlayer formed at 0.005 V vs, Cu/0.1 M Cu2+. Pt submonolayer on Au(111) obtained by replacement of Cu upd monolayer S. R. Brankovic, J. X. Wang, R. R. Adžić, Surface Science 2001, 474, L173. Experimental Cu 2+ 4+ 4+ Pt4+ 4+ 4+ 4+ 2+ 4+ 2+ 4+ 2 e- 2+ 4+ 2 e- 2+ 2+ 2+ 4+ 4+ 2+ 2+ 2+ 4+ 4+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ 2+ Pt monolayer Iodine Cu UPD Gold Previously Prepared Cu UPD on Au(111) Surface in 0.05 M H2SO4 with 1mM CuSO4 Solution. Honeycomb (√3 x √3)R30° Structure (θCu = 0.67 and θanion = 0.33) [121] [121] Interfacial structure of the Cu UPD on Au(111) after the first UPD peak:. top view side view - Honeycomb (√3 x √3)R30° structure in which θCu = 0.67 and with (bi)sulfate anions occupying the centers of the honeycomb (θanion = 0.33) - Cu-(1x1)structure with (bi)sulfate adsorbed on top of the copper adlayer. * Toney, M. F.; Howard, J. N.; Richer, J.; Borges, G. l.; Gordon, J. G.; Melroy, O. R. Phys. Rev. Lett. 1995, 75, 4472. LEED Pattern (I) Clean Au (111) (1 x 1) Beam Energy : 55 eV Cu UPD (√3 x √3)R30º Beam Energy : 40 eV AES (I) Clean Au (111) vs 1st Cu UPD (01/21/05) dN(E)/dE Clean Au (111) 1st Cu UPD 100 200 300 400 500 600 Electron Energy (eV) 700 800 900 1000 EC-STM Images of Pt Submonolayer on Au(111) Obtained by Replacement of Cu UPD Monolayer Iodine-Induced Reordering of Pt After Iodine treated in 0.1 M HClO4 solution at 0.6 V vs. Ag/AgCl AES (II) Clean Au (111) vs I / Au (111) vs Cu / I / Au (111) (05/03/05) dN(E)/dE Clean Au (111) I / Au (111) I Cu Cl 100 200 Cu / I / Au (111) I 300 400 500 600 700 Electron Energy (eV) 800 900 1000 LEED Pattern (II) Clean Au (111) (1 x 1) Beam Energy : 55 eV I / Au (111) (√3 x √3)R30º Beam Energy : 48 eV Cu / I / Au (111) (√3 x √3)R30º Beam Energy : 48 eV EC-STM Images of Pt Layer After the 1st Replacement Cycle of UPD Cu with Pt(II) Iodine modified Pt submonolayer in 0.1 M HClO4 solution at 0.6 V vs. Ag/AgCl [121] [121] [121] [121] [121] [121] [121] High Tunneling Current Reveals Pt with Au(111)-(1×1) Substrate 10 x10 nm2 10 x10 nm2 10 x10 nm2 OCP change during Exchange with Pt (+2) Solution Cycle 1 to 10 Potential change during Pt exchange 2-27-06 0.650 E /V 0.550 0.450 0.350 0.250 0.150 0 100.0 200.0 300.0 t/s 400.0 500.0 600.0 Green -1 Blue -2 . . . OCP change during exchange using Pt (+4) solution platinum exchange 22806-pt 1 0.750 0.650 E /V 0.550 0.450 0.350 0.250 0.150 0 25.0 50.0 75.0 100.0 125.0 150.0 t/s Cycles 1 -4 (top one is first) EC-STM Images of Pt Layer After the 2nd Replacement Cycle of UPD Cu with Pt(II) Iodine modified Pt monolayer in 0.1 M HClO4 solution at 0.6 V vs. Ag/AgCl High Resolution EC-STM Images of the Iodine modified Pt Layer on Au(111) [121] After 1 min (√3 x √3)R30°- I on Pt layer Iodine modified Pt monolayer in 0.1 M HClO4 solution at 0.6 V vs. Ag/AgCl High Resolution EC-STM Images of the Mobility of Pt-complex After 1 min Pt I in Iodine and Pt(II) or Pt(VI)-free 0.1 M HClO4 solution High Resolution EC-STM Images of the Moire Pattern, (√3 x √3)R30°- I and Pt-Complex [121] EC-STM Images of Pt Layer After the 3rd Replacement Cycle of UPD Cu with Pt(II) Iodine modified Pt layer in 0.1 M HClO4 solution at 0.6 V vs. Ag/AgCl [121]