Base(s)
advertisement

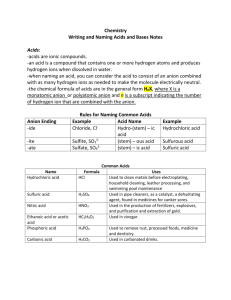
Compounds of TEST: 1. What is a Compound? - Chemical formulas, subscripts, & coefficients. 2. Covalent compound - What are they made of? - How do they form? - Naming & Formula writing. 3. Polyatomic Ions - - What are they made of? - Naming & formula writing. 4. Ionic compound - What are they made of? - How do they form? - Naming & Formula writing. 5. Acids & Bases - What are they made of? - naming & formula writing. 6. Hydrates & % Composition Bases: • Bases are aqueous ionic compounds that contain Hydroxide ions. (Dissolves in water) • Bases release Hydroxide ions into solution (when dissolved in water). • Naming Bases: (name like ionic compounds with a polyatomic hydroxide ion in it.) • All bases have names that end in hydroxide. Examples: • NaOH = sodium hydroxide • KOH = potassium hydroxide • Ca(OH)2 = calcium hydroxide All Example: Acid’s are aqueous, HCl (aqueous) Hydrochloric Acid & not Acidic HCl (liquid) Hydrogen Chloride [not an acid] Aqueous Acids: Acids are aqueous compounds that release Hydrogen Ions into solution. Hydrogen is always written 1st in the chemical formula for The general formula for all acids Example of Binary acids = all Acids. HnX Examples of oxy-acids HCl = Hydrochloric acid HNO3 = nitric acid HF = Hydrofluoric acid HNO2 = nitrous acid HBr = hydrobromic acid HBrO3 = bromic acid H 2S = Hydrosulfuric acid H2Se = Hydroselenic acid HBrO2 = bromous acid H2SO4 = Sulfuric acid H 3N = Hydronitric acid H2SO3 = sulfurous acid H 3P = Hydrophosphoric acid H3PO4 = phosphoric acid Binary Acids: • Are acids that contains hydrogen and 1 other element. Naming Binary acids: 1. To name, add the prefix hydro- for the hydrogen part of the molecule. 2. To the root of the second element name, add the suffix –ic followed by the word acid. Example: HCl = Hydrochloric acid H 2S = Hydrosulfuric acid H 3P = Hydrophosphoric acid Oxyacids (Acids with oxygen) Example: • HNO3 = nitric acid • H2SO3 = sulfurous acid • H2SO4 = Sulfuric acid • H3PO4 = phosphoric acid • HBrO3 = bromic acid • Oxyacids are acids that contains hydrogen and an oxyanion. Naming = – 1st – 2nd write the root name of the polyatomic oxy-anion. (no hydro) add to the ending of the root… -ate changes to –ic acid -ite changes to -ous acid Nitrate Nitric acid Nitrite Nitrous acid HCl = Hydrochloric acid Binary acids HF = Hydroflouric acid H 2S = Hydrosulfuric acid H2Se = Hydroselenic acid H 3N = Hydronitric acid H3P = Hydrophosphoric acid Oxyacids HNO3 = Nitric acid H2SO4 = Sulfuric acid H2SO3 = Sulfurous acid H3PO4 = Phosphoric acid HBrO3 = Bromic acid