Microscale chemistry
advertisement

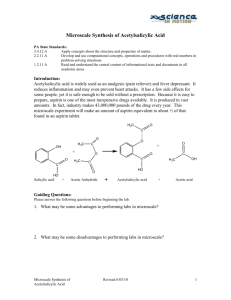
Microscale chemistry Current use and future opportunities An organic view from Strathclyde Dr. Colin Gibson Navigating the presentation: What is microscale all about and a brief history? Reasons for introducing microscale @ Strathclyde. The advantages/disadvantages:- Strathclyde/elsewhere. The available resources. What we do (time, what experiments, apparatus, work-up, purification & analysis). +/- of the teaching/learning experience. The future? What is microscale? “…an environmentally safe pollution prevention method of performing chemical processes using small quantities of chemicals without compromising the quality and standard of chemical applications in education and industry.” (www.microscale.org) “Thus, where students formerly worked with 10-50 g of starting material and 50-500 g of solvent, they may now work with 10-150 mg of reactant and 1-10 g of solvent” (Butcher et al. JCE, 1985, 62(2), 147.) Microscale timeline 1983 Introduced at Bowdoin College, Merrimack College and Brown University, USA to improve laboratory air quality which lacked high efficiency ventilation. 1986 Microscale inorganic chemistry developed @ Merrimack. 1987 First all microscale inorganic laboratory @ Merrimack. 1993 National microscale chemistry center (NMCC) established @ Merrimack. 2003 Funding for NMCC ceases. Why microscale @ Strathclyde? 1992 Asked to look at modernizing 3rd year organic labs. Prescriptive set of semi-microscale (1-5 g) preparations and qualitative organic analysis. No change from 2nd year labs. Modernize chemistry, remove unknowns (use nmr, ir) and introduce choice of experiments. It’ll cost too much for chemicals. We’ll use microscale to trim costs and as better preparation for placement year. Introduced in 1993. Advantages cited by others & in retrospect Reduction of hazardous waste disposal “…most of the product generated in the (semi-micro) laboratory is never employed for any useful chemical purpose.” (Szafran et al. JCE, 1989, 66(11), A263.) “…if you need 100 mg, make 100 mg, don’t make 5 g and throw away 4.9 g” (Breuer, CEJ, 2004, 79(2), 17.) Advantages cited by others & in retrospect Reduction of hazardous waste disposal “In 1992, a large (US) university taught both macroscale & microscale laboratory courses, simultaneously to over 1000 students. … The cost of waste disposal per student in this course was 25 times less than the corresponding macroscale course.” (Williamson, Macroscale & Microscale Organic Experiments, 1993, D. C. Heath & Co.) In 1986 Arizona State university observed an 80% reduction of waste for microscale compared to conventional labs. (Chloupek-McGough, JCE, 1989, 66(1), 92.) Advantages cited by others & in retrospect Pedagogic (A)- Reduced experiment times, increases number of manipulations per session:40% time reduction to complete Grignard experiment. (Pickering et al., JCE 1986, 63(6), 535.) Pedagogic (B)- Students develop better manipulative skills and teaches them to work carefully:limited tolerances for accidental losses with 100 mg of product. Advantages cited by others & in retrospect Reduced cost of chemicals:Allows introduction of choice of experiments. Reduced cost of chemicals:Opportunity to use more sophisticated/expensive reagents. Reduced apparatus footprint:More economical on fumecupboard use (≤ 5 experiments per hotplate-stirrer). Advantages cited by others Improvement of laboratory air quality Reduced contact with toxic chemicals Reduction of fire/explosion hazard Use of non-commercial reagents Glassware is more durable Reduces storage space Reasons cited against using microscale Our students are not skillful enough. Strathclyde students are no better/worse than others but do quite well with microscale. Students don’t meet standard equipment. True, if only microscale is introduced, why not a mixture of micro (100 mg)/semi-microscale (1-5 g)? It’s inappropriate training for research and industry. Organic research does use microscale as does some areas of pharmaceutical research. Few places can teach pilot scale chemistry! Expensive equipment. True, but using a mixture of micro/semi-microscale (1-5 g) then a kit is not needed for each student. Reasons cited against using microscale Unsatisfactory lab experience through ‘lack’ of product. Usually not the case with solid products. Can be an issue with colourless oils but they learn that there is enough product for tlc and an ir. Difficulty students have learning small scale techniques without having mastered large scale manipulations. Maybe. Why not semi-microscale (1-5 g) then a mixture of micro/semi-microscale? Lack of experience with macroscale exothermic experiments. Macroscale = 500 g, few places can teach pilot scale chemistry!!! Why not a mixture of micro/semi-microscale? What resources are available? There are 139 articles published between 1983 - March 2010 (mainly in Journal of Chemical Education (JCE)) of experiments, equipment and the philosophy of microscale. Ca 14 textbooks techniques. of microscale experiments and National microscale chemistry center (NMCC). www.microscale.com Plus 11 international microscale chemistry centres throughout the World, but not the UK. What we did/do 1993 set up microscale (ca 100 mg) and semi-microscale (1-5 g) for our 3rd year students (= 2nd year in rest of UK). Microscale were based on experiments from Mayo et al., “Microscale Organic Laboratory”, 2nd Edn., Wiley, 1989. Subsequent experiments from Williamson, “Macroscale and Microscale Organic Experiments”, 2nd Edn., D. C. Heath & Co., 1994. What we did/do In 1993/94:- 6 weeks of 4 afternoons @ 4 h = 96 h. In 2009/10:- 4 weeks of 4 afternoons @ 3 h = 48 h. 2009/10:- introduced microscale PCC oxidation into 2nd year. Uses 135 mg of alcohol and a silica column, analysis by ir. 2009/10:- 2nd year carry out prescribed course of 1 semimacroscale (25 g), 3 semi-microscale experiments (1-5 g) and microscale PCC oxidation. What we do in 3rd year Compulsory multistep Wittig reaction @ semi-macro and semi-microscale. 3 Steps and worth 20 marks 2 Semi-microscale (ca 1-2 g) experiments from a choice of five, worth 15 or 20 marks (crossed aldol, Diels-Alder (anthracene), acylation of ferrocene, fenbufen preparation and enantioselective yeast reduction). 2 Microscale experiments from a choice of four, worth 15 marks (Diels-Alder (sulfolene), Beckmann, enol acetate preparation and PCC oxidation). The microscale kits The apparatus On work-up, purification & analysis Biphasic extractions are carried out in 3 or 5 ml reaction vials using a Pasteur pipette to separate phases. Recrystallizations are carried out in the reaction vials, followed by filtration with a 10 mm ∅ Hirsch funnel. We do not use Craig tubes! Oils can be purified by silica column using pipette. On work-up, purification & analysis Analysis by solution cell ir using CH2Cl2 (20 mg) and mp, tlc. Remainder is handed in for marking On the pluses We have introduced choice. Waste reduction. Improved manipulative skills:- project students happily carry out microscale transformations. now Students learn to take more care with experimental conditions. Curbs the free-spirited experimentalists! Students learn that chemistry is not just about making grammes of white solid. On the pluses Students learn that ca 50 mg of a colourless oil is just visible to the human eye and is enough for ir and tlc. Reduced apparatus footprint is a huge advantage. Over the last 4 years we have struggled to cope with student numbers in the laboratory. No mass failure of students. Just as many pass as did in 1992! Strathclyde students are just as popular as ever as one year industrial placement students. On the minuses Cost of the kits. On the minuses:- cost of kits £££ 3 ml Reaction vial 14.60 5 ml Reaction vial 14.60 Drying tube 16.10 Claisen adapter 33.50 Air condenser 19.60 Water jacketed condenser 41.80 250 µl Syringe 42.08 2 ml Syringe + needle 6.73 Spin vane/stirrer 5.50 Caps, O-ring, septa 1.79 196.30 On the minuses Cost of the kits. Cost of losses and breakages. 2008/9:- 7 microsyringes, 5 drying tubes (microkit for whole 4 week period). Now students must sign in/out kits after each experiment and kits are monitored by a technician. 2009/10:- 1 microsyringe, 9 stirring vanes. On the minuses Cost of the kits. Cost of losses and breakages. Need to use a ir solution cell with CH2Cl2. Now have an ATR ir for small samples but KBr disks may be a way forward. On the minuses Cost of the kits. Cost of losses and breakages. Need to use a ir solution cell with CH2Cl2. Less tolerant to experimental errors. Hotplate too high, or the ‘free-spirited’ student. Students are less keen on microscale. ? I don’t have enough/any product. Usually there is! ? Is this done in industry? So, that’s it sorted? “In the three years since 1985…., nearly 300 institutions moved to convert to this type of program. The magnitude and rapidity of adoptions really does mean that we are in the midst of an educational revolution.” (Mayo et al., “Microscale Organic Laboratory”, 2nd Edn., Wiley, 1989. ) “The revolution is over. The microscale era has arrived. We are confident that future students will be assured the opportunity to experience the thrill of coaxing one complex molecular structure into another and to bring about these transformations on quantities of material that are just visible to the human eye!” (Mayo et al., “Microscale Organic Laboratory”, 3rd Edn., Wiley, 1994. ) So, that’s it sorted- Sadly, no! “At the time of writing about 66% of US universities and colleges……use microscale experiments to some extent.” (Breuer, CEJ, 2004, 79(2), 17.) “At the time of writing about 66% of US universities and colleges and about 33% of UK universities use microscale experiments to some extent.” (Breuer, CEJ, 2004, 79(2), 17.) March 2010:- straw poll of OrgNet, still about 33% of UK universities using microscale. Future opportunities? Encourage the 67% to adopt some microscale element? Future opportunities? Really do need more modern and up-to-date experiments. Microscale Organic Papers Experiments Apparatus Principles 12 10 8 6 4 2 2010 2005 2000 1995 1990 1985 0 Future opportunities? Really do need more modern and up-to-date experiments. More kudos for demonstrating/developing undergraduate laboratories. Currently a poor relation to research and lecturing and tutorials. Future opportunities? Really do need more modern and up-to-date experiments. Broaden to inorganic experiments. Strathclyde will introduce 2 microscale inorganic experiments over next 2 years. To reduce cobalt salt use. Perhaps some using expensive catalysts or introduce catalysis? Perhaps some organometallic examples? Future opportunities:- Microreactors Network of micron sized channels etched into solid surface (10-500 µm 1-8 cm) Flow rates ~ 1-250 µl min-1 (10 ml min-1) Reagent concentration ~ 0.01-2 M Reaction times ~ 10-20 min (10 h) Future opportunities:- Microreactors Control reagent mixing. Control thermal (high/low) or concentration gradients. Better conversion & selectivity than batch reactors. So better yields and purity. More atom efficient. Only small amounts of solvents and reagents needed. Improved sustainability. Watts et al., Chem Comm., 2007, 443 Wirth et al., OBC, 2007, 5, 733 Future opportunities:- Microreactors Professor T. Wirth @ Cardiff University has University funding to introduce microreactor experiments for chemistry & pharmacy students. T. Wirth, private communication, April 2010. Who did/do all the hard work? Lilias Rees Dr. Debbie Willison Dr. Marion Currie Dr. Chris Dodds Alasdair Shaw Dr. John Haddow Strathclyde 3rd year 1993-2010 Thank you for listening & any questions? What is microscale? … “an environmentally safe pollution prevention method of performing chemical processes using small quantities of chemicals without compromising the quality and standard of chemical applications in education and industry” (www.microscale.org) What is microscale? The apparatus (© Alasdair Shaw, Strathclyde University)