Learning Guide for Chapter 14 - Alcohols
advertisement

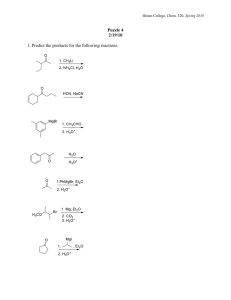
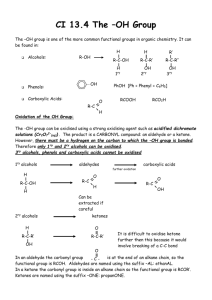
Learning Guide for Chapter 14 - Alcohols (I) I. Introduction to Alcohols and Thiols II. Acid/base Behavior of Alcohols, Phenols, and Thiols III. Nomenclature of Alcohols IV. Synthesis of Alcohols Previous reactions Reduction of C=O functional groups Organometallic reagents I. Introduction to Alcohols and Thiols What does an alcohol functional group look like? OH O How are alcohols different from carboxylic acids? OH carboxylic acids ahve C=O on same carbon as OH, alcohols do not What are some common alcohols? methanol ethanol isopropanol CH3OH wood alcohol; race car fuel; poisonous - causes blindness, then death; treatment - IV of ethanol grain alcohol; found in alcoholic beverages; comes from fermenting sugars and starches; denatured for use in labs; poisonous over time or in high doses OH OH found in rubbing alcohol; used in first aid for disinfecting What does a thiol functional group look like? What other name are they known by? SH mercaptan - captures mercury Why is their reactivity similar to alcohols? What is the main difference in their reactivity? S and O are in the same family size - S is larger than O thiols are more acidic, stronger Nu LG Ch 14 p 2 What is the most noticeable characteristic of thiols? strong, disagreeable odor What are some examples of this? SH SH SH natural gas additive Is this always the case? skunk odor no SH grapefruit mercaptan - citrusy smell Classification of Alcohols How are alcohols in which the carbon is sp3 hybridized classified? 1o, 2o, 3o OH 2o OH 3o OH 1o How are alcohols in which the carbon is sp2 hybridized classified? enols or aryl alcohols OH OH attached to C=C O tautomerize to form C=O enol OH aryl alcohol OH phenol OH directly attached to benzene ring other aryl alcohols are sometimes called phenols How would the following compounds be classified? OH OH allyl alcohol unsaturated alcohol not an enol aromatic alcohol not an aryl alcohol LG Ch 14 p 3 Physical Properties of Alcohols Match up the following compounds with their boiling points and justify your answers. similar masses dipole forces bp -42oC ethyl fluoride hydrogen bonding ethanol Van der Waals bp -38 oC bp -38 oC F OH bp 78oC bp 78oC much higher! bp -42oC propane Which of the following would you expect to be soluble in water? OH CH3OH OH OH OH HO OH HO OH OH OH OH soluble: 1-4 carbons with one OH; up to at least 6 C's with more than one OH IR Spectra of Alcohols Is IR a good way to identify alcohols? yes Label the important bands in the following spectrum. O-H C-H 3400-3200 cm-1 CH2 C-O bending 1300-1000 cm-1 OH LG Ch 14 p 4 NMR Spectra of Alcohols H Consider the following compound. H OH Where should you look for the H's next to the OH? 3-4 ppm, triplet Where should you look for the OH peak? 1-5 ppm, broad singlet What will happen if D2O is added to this sample? OH disappears DHO peak appears Label the peaks in the following spectrum. OH a - 5H, m, 7ppm b - 2H, t, 2-3 ppm c - 2H, t, 3-4 ppm d - 1H, s, 1-5 ppm a>c>b>d II. Acid-base Behavior of Alcohols, Phenols, and Thiols What happens when an alcohol reacts with an acid? base OH acid H-base pKa -2.4 OH2 + base pKa < -2.4 protonated alcohol What kind of acid would be required for this reaction to have a favorable equilibrium? pKa less than 2.4 H2SO4 -10 HI -9 HBr -8 HCl -7 What would happen if the protonated alcohol came in contact with a base? OH2 NaOH OH + H2O it would be protonated LG Ch 14 p 5 What happens when an alcohol reacts with an base? pKa > 18 pKa 16-18 OH base + H-base O alkoxide acid What kind of base would be required for this reaction to have a favorable equilibrium? pKa of the conjugate acid must be greater than 18 most commonly used base - NaH - pKa 36 How can alkali metals be used to form alkoxides? by a redox reaction 1o 3o OH OH Na O K O Na + 1/2 H2 Na - methyl, 1o o o + 1/2 H2 K - 2 , 3 (Na too slow) K remember our friend from E2? What would happen if an alkoxide came in contact with an acid? O HCl OH Cl it would be protonated What would happen if a protonated alcohol and an alkoxide were reacted with each other? O + OH2 2 they would neutralize each other How does the acidity of alcohols vary? methyl > 1o > 2o > 3o OH pKa 15.9 OH pKa 16.5 OH pKa 18.0 OH LG Ch 14 p 6 Why are aryl alcohols more acidic than aliphatic alcohols? conjugate base is stabilized by resonance OH O O base O O greatest resonance contributor pKa 10.0 + H-base Could NaOH be used as the base? yes O OH + NaOH + H2O products favored pKa 10.0 pKa 15.7 stronger acid Why won't this work with regular alcohols? alcohols not acidic enough O OH + NaOH + H2O pKa 15.7 stronger acid pKa 17 Why are thiols more acidic than alcohols? sulfur is larger than oxygen, H is further away, easier to remove base SH S pKa 10.5 + H-base thiolate ion Could NaOH be used as the base? S SH + pKa 10.5 stronger acid yes NaOH + H2O pKa 15.7 reagents favored LG Ch 14 p 7 III. Nomenclature of Alcohols Common names Based on the common name for the following alkyl halide, deduce the common name of the alcohol shown. Br propyl bromide OH propyl alcohol name of substituent + "alcohol" Give the common names of the following alcohols. methyl alcohol CH3OH ethyl alcohol OH OH OH isopropyl alcohol OH isobutyl alcohol OH sec-butyl alcohol tert-butyl alcohol OH cyclopentyl alcohol O OH OH allyl alcohol OH H vinyl alcohol unstable OH benzyl alcohol phenol IUPAC names What are the four steps in naming an organic compound? 1. Determine stereochemistry, if any. 2. Choose the principle chain, give it a name. 3. Number the principle chain. 4. Name the substituents, alphabetize them, add them to the name. LG Ch 14 p 8 What are the priorities for choosing the principle chain? 1. must contain the alcohol (or as many as possible) 2. double or triple bonds 3. longest chain 4. most substituents HO OH OH How does the alkane name change for each of the following? OH pentane pentanol drop "e", add "ol" OH cyclohexane OH cyclohexene cyclohexenol hexane hexadiene drop "ane" add "adiene" pentane pentanediol use entire alkane name, add "diol" OH What are the priorities for numbering the principle chain? 1. lowest number to alcohol(s) 2. lowest number to double or triple bonds 3. lowest numbers to substituents (first point of difference) 4. lowest number to first substituent alphabetically LG Ch 14 p 9 Number the following compounds. HO 2 6 OH 2 1 3 5 1 4 5 3 OH 4 3 1 2 4 If you have to name an alcohol as a substituent, what is it called? hydroxy OH OH 2-(1-hydroxyethyl)-1-cyclopentanol Draw structures for the following names. 2-cyclohexyl-1-butanol 1,3-cyclopentanediol 5-bromo-4-methyl-2-hexanol OH OH OH HO 2-cyclopenten-1-ol Br 2-(1-hydroxyethyl)-1-cyclohexanol 5-methyl-3-hexanol OH OH OH OH Give the names of the following compounds. OH 2,3-pentanediol (3S,4S)-4-methyl-3-hexanol OH OH OH OH OH trans-3-penten-2-ol 4-(2-hydroxypropyl)-1-cyclohexanol LG Ch 14 p 10 IV. Synthesis of Alcohols Reactions from Previous Chapters What reagents would be required to convert the following compounds to alcohols using the reaction given? NaOH SN2 reactions Br 1o RX to ROH OH 1o H2SO4 Acid-catalyzed hydration OH H2O OH 1. Hg(OAc)2, H2O Oxymercuration-reduction 2. NaBH4 C=C to ROH 1. BH3-THF Hydroboration-oxidation OH 2. H2O2, Hydroxylation C=C to ROH KMnO4, KOH, H2O or OsO4, H2O2 OH- C=C to ROH OH C=C to vicinal diol OH In this chapter, we will learn two more: reduction of a C=O by adding hydrogen O H HO ketones aldehydes carboxylic acids esters to ROH reduction of a C=O by adding an alkyl group O HO R ketone aldehyde to ROH ester acid chloride LG Ch 14 p 11 Reduction of C=O groups to form alcohols How is oxidation defined in organic chemistry? gaining bonds to O and/or losing bonds to H How is reduction defined in organic chemistry? losing bonds to O and/or gaining bonds to H Which of these is occurring in the following reactions, and why? OH KMnO4 both C's gain bonds to O H2O, KOH 1. BH3-THF 2. H2-O2, OH- OH oxidation OH H one C gains bond to O, one C gains bond to H no net change in oxidation state H H2, Pd/C H both C's gain bonds to H reduction What type of alcohol can be formed by the reduction of the following compounds? lose a bond to O, gain a bond to H O reduction aldehyde OH 1o alcohol H ketone carboxylic acid O reduction O reduction 2o alcohol OH 1o alcohol OH O ester OH reduction O OH + OH 2 alcohols LG Ch 14 p 12 Which of the following reagents is effective in reducing the functional groups shown? C C C C to C C ketone, aldehyde C O to C OH COOH, ester C O to C OH H2, Pd/C yes no no NaBH4 no yes no most common reagent for ald/ket LiAlH4 no yes yes Raney Ni yes yes no most common reagent for ester, COOH Which is more reactive, NaBH4 or LiAlH4? Why? H NaBH4 = H B Na H H Li LiAlH4 = H Al H Al-H bond weaker than B-H H H less reactive solvent - methanol more reactive solvent - ether How do they react? H H B Na H !- O NaBH4 !+ H CH3OH H H Li H Al H H O LiAlH4 H 2. H3O+ O H Na H H H Li O OH H H OH H H What is Raney Ni? What is the advantage of using it? Ni catalyst with H2 molecules absorbed onto it it will also reduce C=C and C---C if they are present What safety hazards exist with these materials? LiAlH4 reacts vigorously with water, releases H2, can cause lab fires Raney Ni is pyrophoric - spontaneously ignites in air both steps taking place in the reaction first step has to finish, then add acid for second step LG Ch 14 p 13 What reagent(s) could be used to accomplish the following reactions? * NaBH4, CH3OH 4 functional groups O or Raney Ni or LiAlH4, 2) H3O+ O ketone --> 2o alcohol OH LiAlH4, 2) H3O+ OH COOH --> 1o alcohol OH O * NaBH4, CH3OH OH H aldehyde --> 1o alcohol or Raney Ni or LiAlH4, 2) H3O+ O LiAlH4, 2) H3O+ O O * NaBH4, CH3OH C=O vs C=C H O ketone vs ester O reduce aldehyde, leave C=C reduce both aldehyde and C=C OH H2, Pd/C O reduce C=C, leave aldehyde H LiAlH4, 2) H3O+ OH OH + O O O * NaBH4, CH3OH O O OH O can't be done! reduce both ketone and ester HO O reduce ketone, leave ester O or Raney Ni O O ester --> 2 alcohols OH Raney Ni H O OH or LiAlH4, 2) H3O+ H O HO OH + HO reduce ester, leave ketone LG Ch 14 p 14 Addition of Organometallic Reagents to C=O Groups to form Alcohols What is the advantage of using an organometallic reagent instead of a reducing agent to convert a C=O group to an alcohol? reducing agent - same carbon structure organometallic reagent - new carbon group added What are the two most common kinds of organometallic reagents used in organic chemistry? !- !+ Grignard reagents MgBr !+ Li organolithium reagents !- What polarity do these reagents have? How reactive are they, and why? C - partially negative highly reactive - C doesn't like to be -, usually formed and used, not stored How are organometallic reagents formed? alkyl halide + metal Br Mg MgBr ether Cl Li 2 Li + LiCl ether What other kinds of halides can be used? vinyl halides Cl Mg ether Br aryl halides 2 Li ether MgCl Li + LiBr LG Ch 14 p 15 What are the two ways in which a Grignard or organolithium reagent can react? base MgBr H-base H + Mg Nu MgBr E E + Mg Br + base Br How strong of bases are organometallic reagents? Why is this so? conjugate acid - alkane pKa ~50 weakest acid - strongest base! C has low EN, unstable with partial neg charge What would happen if an organometallic reagent came in contact with water? it would be protonated Li H O H H + Li O H What other kinds of compounds will react as acids with organometallic reagents? alcohols, amines, amides, carboxylic acids, terminal alkynes If this usually a reaction we desire? no - it ruins our reagent! Is there any way in which it could be useful? converts RX to alkane Br MgBr CH3OH Mg + CH3O ether Mg Br adds a D Li Cl 2 Li hexanes D D2O + LiOD LG Ch 14 p 16 In the absense of any compounds with acidic H's, organometallic reagents react as nucleophiles. What type of compound will be formed when a Grignard or organolithium reagent is reacted with an aldehyde or ketone, followed by mild, aqueous acid? attack-push up O Mg Br O H 2. H3O+ aldehyde OH MgCl protonate react once 2o alcohol O Li O Li OH 2. H3O+ ketone 3o alcohol Is there any way to get a primary alcohol? MgBr O Mg OH Br O H H 2. H3O+ formaldehyde 1o alcohol What type of compound will be formed when a Grignard or organolithium reagent is reacted with an acid chloride, following by mild, aqueous acid? can't stop here! attack-push up push off reacts twice O O Cl 2 eq MgCl O Cl + Cl 2. H3O+ acid chloride OH protonate 3o alcohol w/2 identical groups Why doesn't this happen with aldehydes and ketones? no leaving group O attack-push up LG Ch 14 p 17 What will happen to an ester? same thing reacts twice O Li 2 O O O 2. H3O+ O + O Li OH + O HO reacts once What type of compound is formed when a Grignard or organolithium reagent is reacted with ethylene oxide? protonate attack-push off O Li OH O adds 2 C's 2. H3O+ What reagents would be needed to complete the following reactions? OH O H H3C MgBr 2. H3O+ 2. H3O+ O H3C H MgBr O OH MgBr H H 2. H3O+ O+ 2. H3 O Li O Li OH O 2. H3O+ 2. H3O+ How could the following compound be synthesized? OH O + H3C Li MgBr 2. H3O+ H 2o alcohol - RM + ald OH Li O + O H 2. H3O+ OH OH LG Ch 14 p 18 Summary of methods for synthesizing alcohols: SN2: 1o alkyl halide + NaOH --> 1o alcohol Acid-catalyzed hydration: alkene + H2SO4 + H2O --> 2o or 3o alcohol (more substituted side, rearrangments possible) Oxymercuration-reduction: alkene + 1. Hg(OAc)2, H2O 2. NaBH4 --> 2o or 3o alcohol (more substituted side, no rearrangements) Hydroboration-oxidation: alkene + 1. BH3-THF, 2. H2O2, NaOH --> 1o or 2o alcohol (less substituted side, no rearrangements) Reduction of an aldehyde or ketone: aldehyde or ketone + NaBH4, CH3OH --> 1o or 2o alcohol Reduction of a carboxylic acid or ester: carboxylic acid or ester + LiAlH4 --> 1o alcohol (ester has a second alcohol) Addition of an organometallic reagent to an aldehyde or ketone aldehyde or ketone + Grignard or organolithium reagent, 2. H3O+ --> 2o or 3o alcohol Addition of an organometallic reagent to and acid chloride or ester: acid chloride or ester + Grignard or organolithium reagent, 2. H3O+ --> 3o alcohol with two identical groups Addition of an organometallic reagent to an epoxide: ethylene oxide + Grignard or organolithium reagent, 2. H3O+ --> 1o alcohol