Separation of Mixtures - Waterford Public Schools
advertisement

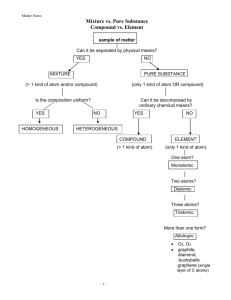
Separation of Mixtures Ms. Grobsky Separating Mixtures Remember, substances in a mixture are physically combined So, processes that are based on differences in physical properties can be used to separate individual components of the mixture Physical properties include: Odor Color Volume (size) Physical state Density Melting point Boiling point Solubility Different techniques are used to separate mixtures into individual components Filtration Separates heterogeneous mixtures composed of an insoluble substance and a soluble substance Uses a porous barrier to separate the solid from the liquid Mixture of solid and liquid Stirring rod Funnel Filter paper traps solid Liquid passes through, leaving the solid in the filter paper Filtrate (liquid component of the mixture) Filtration Distillation Used to separate homogeneous mixtures with liquids that have different boiling points of the components In the process, the mixture is boiled The liquid with the lower boiling point will vaporize first and the vapor is collected and condensed The liquid collected is the distillate Gasoline, kerosene, fuel oil, and lubricating oil are produced from petroleum by distillation A Distillation Apparatus How to Separate a Sand-Salt Water Mixture Evaporation and Crystallization Technique that can be used to separate a solute from the solvent in a solution Results in the formation of pure solid from a solution containing dissolved particles of that substance As the liquid evaporates, the dissolved substance comes out of solution as crystals Produces highly pure solids Rock candy is an example of this! Using a Separatory Funnel A separating funnel can be used to separate two liquids which are immiscible The more dense liquid can be collected first by opening the stopcock Centrifugation Can be used with liquid or solid mixtures AFTER Before Spin sample very rapidly Denser materials go to bottom Example Separate blood into serum and plasma Serum Blood Serum (clear) Plasma (contains red blood cells ‘RBCs’) Check for anemia (lack of iron) RBC’s A B C Magnetism Can be used to separate a magnetic substance from a non-magnetic substance Magnets are used in the recycling industry to separate iron and noniron metals Chromatography Broad category of separation techniques for obtaining pure substances from homogeneous mixtures like dyes, ink, gas mixtures, drug analysis or even DNA! Some chromatography techniques are low-tech Paper chromatography Whereas others are more high-tech and expensive Ion exchange chromatography HPLC chromatography Gas chromatography Each technique differs in their mobile and stationary phases Stationary phase The platform on which chemicals are separated Mobile phase Solvent that carries the chemicals through the stationary phase Separation by Gas Chromatography sample mixture a chromatographic column stationary phase mobile phase selectively absorbssweeps sample components down column http://antoine.frostburg.edu/chem/senese/101/matter/slides/sld006.htm detector Separation by Chromatography sample mixture a chromatographic column stationary phase mobile phase selectively absorbssweeps sample components down column http://antoine.frostburg.edu/chem/senese/101/matter/slides/sld006.htm detector Ion Chromatogram of Orange Juice detector response K+ Na+ 0 5 Mg2+ Fe3+ 10 15 time (minutes) Ca2+ 20 25 How Does Chromatography Work? Separation is based on different substances having differences in physical properties like: Solubility of the sample in the mobile phase “Like Dissolves Like” Molecule size Separation occurs because various components travel at different rates Taking a Closer Look at Paper Chromatography Paper chromatography is one easy technique used to separate the soluble components of a homogeneous mixture We will perform paper chromatographic separations of the pigments in FD&C dyes next class! A Brief Overview of Paper Chromatography • Compound is placed on stationary phase • Mobile phase passes through the stationary phase • Mobile phase solubilizes the components • Mobile phase carries the individual components a certain distance through the stationary phase, depending on their attraction (affinity) to both of the phases Paper Chromatography Mark the solvent front & allow paper to dry Solvent front Purple spots develop located at different distances from the origin line Dye Spots origin solvent Illustration of Chromatography Stationary Phase Separation Mobile Phase Mixture Components Components Affinity to Stationary Phase Affinity to Mobile Phase Blue ---------------- Insoluble in Mobile Phase Black Red Yellow The Rf Value and How It’s Used The chromatogram can be analyzed by measuring the distance travelled by the solvent front, and the distance from the origin to the center of each spot This is used to calculate the Rf (relative front) value for each spot: Distance moved by spot Rf Distance moved by solvent An Rf value is characteristic of a particular solute in a particular solvent It can be used to identify components of a mixture by comparing to tables of known Rf values X Rf Y The Rf value is always a value less than one as the solvent front always moves further than the solute molecules X3 Y X1 X4 X2 X5