Document
advertisement

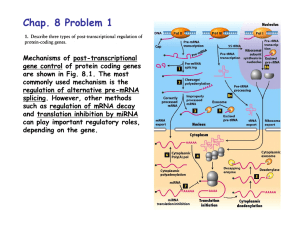
Summary 12: RNA Maturation & Processing Slide 1: We continue our RNA series with RNA processing. The primary focus will be on premRNA modifications at the 5’ and 3’ ends and on splicing, which will convert pre-mRNA into “mature” mRNA. Importantly, 5’ and 3’ modifications happen only in eukaryotes, whereas splicing has been observed in both pro- and eukaryotes. However, coding RNAs in bacteria usually do not require splicing (as shown in this slide), splicing in prokaryotes is confined to non-coding regulatory RNAs. Slide 2: This overview puts all processing events into cellular context. We are focusing on process 1, 2 and 7. Please note that pre-mRNA splicing and polyadenylation occur cotranscriptionally. This slide also informs you about the localization of the three different RNA polymerase complexes (nucleus vs. nucleolus). Slide 3: To better understand the differences between transcription in pro- and eukaryotes, we are taking a step back and look at the gene structure in bacterial DNA. Genes that work in the same biochemical pathway are often clustered together in an operon. The trp operon contains five genes that encode proteins required for tryptophan biosynthesis. All genes in this operon are transcribed from a single promoter into a single mRNA. This mRNA has five docking sites for ribosomes and thus five different proteins are produced from one mRNA. Slide 4: In eukaryotes, the five corresponding genes are located in different chromosomal regions. Each gene gives rise to a transcript that is then translated into protein. Slide 5: This is an overview of the different pre-mRNA processing steps: 5’ capping, splicing and 3’ polyadenylation. Splicing occurs during transcription, so strictly speaking, the order of events is not described quite accurately in this slide. Polyadenylation is triggered by a specific sequence in the transcript. An endonuclease cleaves this sequence and a specific polymerase adds a tail of As. This poly (A) tail protects the transcript from 5’ exonucleolytic degradation and is also required for translation initiation. Slide 6: The capping enzyme adds a 7-methylguanylate to the 5’ phosphate of the transcript. This is the only 5’ to 5’ linkage in nucleic acids. The cap protects from degradation and is important for translation. The capping enzyme is recruited by the phosphorylated CTD of RNA polymerase II. Slide 7: This slide illustrates the splicing of intron DNA. The mature mRNA is intronless and only contains the information encoded in the exons. Typically, there is a short stretch at the 5’ and 3’ ends that is not translated into protein, termed UTR for untranslated region. Slide 8: Introns are encoded by specific sequences at the exon intron junctions. Each intron has a 5’ splice site, a 3’ splice site and a branch point. The conserved nucleotides within the intron are: GU (5’), A (branch point) and AG (3’). The 5’ exon usually ends with an AG and the 3’ exon starts normally with a G. Slide 9: We’ve discussed this table before, and I included it as a reminder that intron DNA amounts to a total of more than 50% of our genome. So the likelihood that a mutation occurs in an intron is quite high. Some of these mutations can affect the recognition sequences for splicing, which could cause a pre-mRNA not to be properly processed. The table in slide 19 lists diseases that arise from improper splicing. We will get back to this point later in the context of “alternative splicing”. Slide 10: Introns in human genes are all excised by spliceosome-catalyzed splicing. However, evolutionarily, splicing likely originated as an RNA-catalyzed process. Group I and II selfsplicing introns utilize a similar splicing mechanism without any requirement for proteins to catalyze the reaction. Group I introns need a guanosine nucleotide co-factor. Group II employs the 2’OH of the adenosine at the branch point to “launch” the attack of the 5’ splice site. This is also true for spliceosome-catalyzed intron removal. Slide 11: This slide shows how the intron forms a lariat structure. This lariat is eventually degraded. Slide 12: Here you can follow the process step-by-step: 2’OH attack of the 5’ splice site, 3’OH is formed after the first transesterification, the 3’OH then attacks the 3’ splice site followed by a second transesterification. In the end, the two exons are linked and the lariat intron is excised. Slide 13: The spliceosome consists of 5 small nuclear ribonucleoprotein particles (snRNPs) all of which contain a specific small nuclear RNA. The snRNAs provide the specificity, as they hybridize with the signal sequences of the intron. In vitro, splicing can be carried out in the absence of proteins. In vivo, the proteins make the process faster and more efficient, but in the absence of the snRNAs the snRNPs would be useless. Slides 14 - 17: These slides provide an overview of the step-by-step assembly of the spliceosome. The individual snRNPs bind specific sequences and bring them close together so that the transesterifications can occur. For example, U1 and U2 bring the branch point and the 5’ splice site together. Once this has been accomplished, U1 is dispensable. U6 is the catalytic center, but it is only active once U4 has been removed. Slide 18: Alternative splicing depicts the process by which a pre-mRNA can yield two different mature RNAs. This is highly tissue-specific. Alternative splicing creates a lot of protein variants or isotypes that are not encoded by their own genes. Calcitonin expression in neuronal and thyroid cells is a well-known example. Slide 19: Alternative splice variants that produce non-functional or not fully functional proteins can cause diseases. In most of the cases listed the pre-mRNA is spliced in an alternative incorrect way, because of mutations in the common splicing signal sequences. Remember BRCA1 or Lesh Nyhan syndrome from previous lectures? Slide 20: I hope you can appreciate the complexity of pre-mRNA processing to obtain a mature and “functional” mRNA. However, not every properly spliced mRNA is necessarily designated for protein translation. miRNAs are short regulatory sequences that can interfere with the fate of an mRNA. Most miRNAs target transcripts in their 3’UTRs (slide 7). If base-pairing between the miRNA and mRNA is discontinuous and incomplete, translation of the transcript is usually inhibited. If base-pairing is perfect, the target mRNA is degraded or “silenced”. The class of miRNAs that can trigger silencing through degradation is called siRNA. RNA interference plays an important role during development, as it can reversibly or irreversibly suppress specific transcripts.