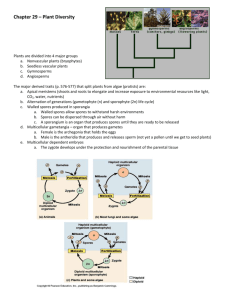
C-Fern® Web Manual
advertisement

C-Fern® Web Manual LESLIE G. HICKOK AND THOMAS R. WARNE CURRENT CONTACT INFORMATION For more information about using C-Fern – http:// www.c-fern.org Leslie G. Hickok - cfern@utk.edu Thomas R. Warne – warne@c-fern.org COPYRIGHT AND REVISIONS 2009, Revised and updated ©2008-present, Leslie G. Hickok and Thomas R. Warne 2004, Revised and updated ©1998-2008, University of Tennessee Research Foundation ACKNOWLEDGMENTS Development of C-Fern was supported, in part, by the National Science Foundation and the University of Tennessee. We gratefully acknowledge Stephenie Baxter for her superb laboratory and greenhouse assistance as well as illustrating the developmental stages of C-Fern. We also thank Jennifer Panter and Dr. Dale L. Vogelien for their contributions towards the optimization of Ceratopteris culture. C-Fern® Web Manual 2 Table of Contents Introduction...................................................................................5 Why Use C-Fern®?........................................................................................................ 5 C-Fern and the National Science Standards.................................................................6 What Is Ceratopteris? ...................................................................................................8 Taxonomy...................................................................................................................... 9 The C-Fern Life Cycle ................................................................................................ 10 A Perspective on Plant Reproduction— C-Fern and Teaching the Concept of Alternation of Generations ..........................................................................................14 Culture Instructions for C-Fern® Investigations .....................18 Introduction.................................................................................................................. 18 Quick Start .................................................................................................................. 18 Culture Instructions .....................................................................................................19 1. Getting Started.................................................................................................... 19 2. Determining Sowing Density and Number of Cultures....................................... 20 3. Preparing the Culture Medium............................................................................ 20 A. Using Pre-Made Bottled C-Fern Medium ...................................................... 20 B. Using Powdered Basic C-Fern Medium ....................................................... 21 C. Using a Microwave to Prepare C-Fern Powdered Medium...........................24 D. Preparation of Basic C-Fern Medium From Stock Solutions......................... 25 4. Inoculating (Sowing) Cultures Using Presterilized C-Fern Spores..................... 27 Seven Good Habits for Sowing C-Fern Cultures ....................................................... 27 5. Maintaining C-Fern Cultures............................................................................... 29 A. Culture Domes................................................................................................29 B. Growth Pod.....................................................................................................29 C. Temperature................................................................................................... 31 D. Light ............................................................................................................... 31 i. Growth Pod Requirements.......................................................................... 31 ii. Culture Dome Requirements ..................................................................... 31 iii. Constructing a C-Fern Light Stand ........................................................... 32 6. Observing Cultures..............................................................................................33 7. Sporophyte Culture............................................................................................. 34 A. Standard C-Fern (RN cultivar) Sporophytes.................................................. 34 B. C-Fern Express Sporophytes......................................................................... 35 8. Additional Information For Educators..................................................................36 C-Fern® Web Manual 3 A. Hints for Large Classes.................................................................................. 36 B. Independent Student Research Projects....................................................... 37 C. Questions for Discussion................................................................................38 Additional Culture Methods and Techniques..........................41 Surface Sterilization of C-Fern Spores .......................................................................41 DarkStart Method ..................................................................................................... 41 Dry Sowing of Presterilized C-Fern Spores................................................................ 44 Liquid Culture of C-Fern Gametophytes...................................................................... 44 Mutagenesis of C-Fern Spores................................................................................... 44 1. Ethyl Methane Sulfonate (EMS) Mutagenesis.................................................... 45 2. X-Ray Mutagenesis............................................................................................. 46 Controlled Fertilization: Selfing and Crossing Techniques ........................................ 47 1. Self-Fertilization...................................................................................................47 2. Cross-Fertilization............................................................................................... 48 Preparation of Semipermanent Slides for Observation and Analysis of C-Fern Gametophytes ............................................................................................................ 49 Greenhouse Culture of C-Fern Sporophytes.............................................................. 51 1. Transplantation....................................................................................................51 2. Fertilization.......................................................................................................... 52 Cloning of C-Fern Sporophytes ..................................................................................52 Collection of C-Fern Spores........................................................................................ 53 Sport Reports - Descriptions of C-Fern Mutants....................55 Glossary of Selected Terms...................................................... 63 Bibliography................................................................................68 User Service................................................................................77 C-Fern® Web Manual 4 Introduction Introduction A re plants alive? Do they do anything really interesting? Ask these questions to any number of high school or college students and be prepared for a variety of answers! Even biology majors may give surprising responses! It is no secret that teaching the basics of plant biology and capturing students’ interest can be a daunting task. To most students, plants are just not as naturally interesting or visually compelling as animals are. Nonetheless, plants are integral and essential components of our living world, and learning about them is a necessary part of any liberal or specialized education experience. Finding more effective ways to teach about them is a challenge. Why Use C-Fern®? Simple and Complex Development • haploid (gametophyte) and diploid (sporophyte) phases • cellular and whole plant observations and experiments Rapid Development • spore to sexually mature gametophytes in 14 days • spore to spore in less than 90 days (original C-Fern) or 60 days (C-Fern Express) Direct Observation of Development Haploid/Diploid Genetic System Optimized for Classroom Use • inexpensive • safe • miniaturized • very large numbers of individuals and treatments possible The advantages of using C-Fern® as a model plant system for teaching derive from the unique properties of its development and life cycle. Both haploid (gametophyte) and diploid (sporophyte) C-Fern® Web Manual 5 Introduction phases exist independently; therefore, observations and experimental studies are possible at both the cellular and whole plant levels without artificial manipulations of the life cycle. Development from spores to sexually mature gametophytes to young sporophytes can be observed within 2 to 3 weeks. The complete, spore-to-spore life cycle can occur in less than 90 days. The rapid growth and small size of gametophytes allows for the miniaturization of experiments, such that large numbers of individuals and treatments can be used in a small space and short time. The haploid/diploid genetic system is very simple. C-Fern combines the features of both higher and lower plant systems. The developmental simplicity and haploid nature of the independent gametophytic phase provides opportunities that are not available in angiosperm models. At the same time, the ability to study many processes within the complex vascular sporophyte phase allows direct comparisons with higher plant systems (e.g., Wisconsin Fast Plants™, Arabidopsis) that are not possible with developmentally simpler systems such as mosses (e.g., Funaria, Physcomitrella) or algae (e.g., Chlamydomonas, Euglena). This manual presents: • The protocols necessary to successfully culture and use C-Fern in educational settings. • Background information about the biology and natural history of Ceratopteris (the fern from which the C-Fern materials derive). C-Fern and the National Science Standards Laboratory-based education can serve many functions, such as the reinforcement and illustration of lecture and text material and the exposure of students to the uses of sophisticated equipment and current methodologies. However, an equally important and often neglected function is the use of the biological laboratory component to promote general literacy in the scientific method; i.e., how the process of science works, what constitutes scientific information, and how scientific information is acquired—in short, how to do science. Comprehension of the scientific method involves understanding and applying a large set of interrelated skills— problem identification, hypothesis formulation, experimental design, implementation, data collection, analysis and synthesis, communication of results—that necessitate different approaches to teaching. The National Science Education Standards (National C-Fern® Web Manual 6 Introduction Table 1. Alignment of C-Fern with the National Research Council Life Science Standards. C-Fern is readily applicable to fundamental concepts or principles that underlie each of the following topics. The bulleted list (•) below each topic notes some specific C-Fern attributes that are relevant to each category. National Research Council Life Science Standards Levels K–4 Characteristics of organisms plant form and function basic requirements for plant growth Life cycles of organisms sexual and asexual reproduction non-flowering plant reproduction alternation of generations Organisms and environment Levels 5–8 Levels 9–12 Structure and function in living The cell systems structure and function in gametophyte C-Fern exists as two independent organisms gametophytes allow easy visualization of cell structure and differentiation comparative studies of gametophyte and sporophyte structure and function Reproduction and heredity tissues—vegetative, meristematic, absorptive / anchoring, specialized sperm cells cell motility (sperm) chemotaxis (sperm) spores as single-celled propagules Molecular basis of heredity C-Fern clearly shows the relationship although structure and function are of all aspects of sexual reproduction very different, C-Fern gametophytes and sporophytes contain the same genetic studies with C-Fern allow genetic instructions examination of both haploid (gametophytic) and diploid gametophytes have half the (sporophytic) ratios chromosomes of the sporophyte a wide variety of mutant stocks allows sexual differentiation in gametophytes extended investigation of the effects is related to a chemical control of of mutation gene expression mutants exist that have altered responses to the chemical (pheromone) that controls sexual type Regulation and behavior responses to changes in environment changes in sex ratio in response to (C-Fern can be grown both population size and/or environment terrestrially and aquatically) changes in sporophyte growthin response to aquatic vs. terrestrial environment Populations and ecosystems the effect of population size on sex ratio or early sporophyte development Diversity and adaptation of organisms the structure and function of the CFern life cycle relative to other plants adaptation to different environments Biological evolution ferns have adapted for a specific lifestyle C-Fern has specialize dadaptations for a “semi-aquatic” life-style Matter, energy, and organization of living systems C-Fern is autotrophic and exhibits distinct adaptations for photosynthesis and related activities C-Fern gametophyte and sporophyte generations are independently autotrophic Behavior of organisms chemotaxis by C-Fern sperm shows behavior at the single cell level C-Fern shows typical plant responses to the environment population pressures influence development in both gametophytes and sporophytes C-Fern® Web Manual 7 Introduction Committee on Science Education Standards and Assessment; and National Research Council 1996) clearly reinforce the need to more effectively teach the process of science through inquiry. However, normal time constraints and equipment needs can make it difficult to implement such an approach. One option is to introduce exercises in experimental science using an amenable and flexible model organism relevant to a broad range of disciplines and subject areas. In plants, only a few model systems have attributes that make them sufficient to meet the demands of a wide range of situations and levels. C-Fern is a broadly useful model system that possesses a suite of characteristics and developmental features that make it ideal for use in both the classroom and in student initiated research. The broad applicability of C-Fern for a variety of classroom uses means that it can be applied and integrated into several of the Content Standards. Life Science standards focus on the science facts, concepts, principles, theories, and models that are important for students to know, understand, and use. What Is Ceratopteris? Ceratopteris is a genus of homosporous ferns found in most tropical and subtropical areas of the world (Lloyd 1973). Species grow as either aquatics or subaquatics and are restricted in habitat to ponds, rivers, or intermittent wet areas such as ditches, rice paddies, or taro patches. Although some require an aquatic habitat, most species can be successfully grown in standard greenhouse pot culture under warm, humid conditions. Ceratopteris is eaten in areas of Southeastern Asia, and there was once an attempt to develop it as a crop in the Philippines (Copeland 1942). Currently, commercial applications are primarily limited to its use as an aquarium plant, where it is sold under the common name of water sprite and has even been immortalized in plastic replicas. In some environments, Ceratopteris can become an aggressive weed by clogging up freshwater streams and drainage systems. Instead of the long-lived rhizome and perennial habit of most homosporous ferns, Ceratopteris has a short, upright rhizome and grows as an annual. The fronds form small “fiddleheads” when young and are dimorphic at maturity. Initial fronds are sterile and from simple to three-lobed, but as development proceeds the fronds become increasingly more dissected and fertile. Sporangia occur in continuous rows on veins along the ventral edges of fertile fronds and are covered by the inrolled margin of the frond, forming C-Fern® Web Manual 8 Introduction a “false indusium.” In addition to sexual reproduction via meiotic production of spores and subsequent gametophytes, Ceratopteris sporophytes have a prolific capacity for vegetative reproduction. Buds found in the axes of subdivisions of the frond can develop rapidly into plantlets and are likely the reason for its weedy nature in some habitats. Both in nature and under greenhouse culture, it is not unusual to find senescent fronds abundantly covered with developing plantlets that originated from these buds. On a practical level, the buds provide a convenient means for vegetative propagation of particular genotypes. Taxonomy Homosporous ferns like Ceratopteris belong to the order Filicales, within the class Filicopsida, in the division Tracheophyta, kingdom Plantae. Because Ceratopteris shows a variety of morphological characters that are exhibited by a number of different families, phylogenetic affinities at the family level are unclear. Some taxonomic treatments have placed Ceratopteris in its own monotypic family, Parkeriaceae, while others have included it as a subfamily or tribe within the Pteridaceae (Tryon and Tryon 1990). Robert M. Lloyd (Lloyd 1974, Lloyd 1993), in his taxonomic treatments, recognized four species: C. thalictroides, C. richardii, C. pteridoides, and C. cornuta. Ceratopteris thalictroides is described as a highly polymorphic species with pantropical distribution. Ceratopteris pteridoides and C. cornuta, show some similarities to each other but are both morphologically distinct from C. thalictroides. Ceratopteris pteridoides, typically thick-leafed and floating, is principally limited to Central and South America, while C. cornuta is mainly confined in distribution to mainland Africa. The fourth species, C. richardii, is morphologically similar to C. thalictroides; the only consistent difference noted by Lloyd (Lloyd 1974, Lloyd 1993) was the 16-spored sporangia of C. richardii as opposed to the 32-spored sporangia of all other species. Because of the plants’ similarities, Tryon and Tryon (1982) did not recognize C. richardii as being distinct from C. thalictroides. However, cytological observations show that C. thalictroides is tetraploid with n=77,78 and the other three species recognized by Lloyd are diploid with n=39 (Hickok 1977, Hickok 1979). Analyses of synthesized interspecific hybrids indicate incomplete reproductive isolation among the diploid species. The presence of herbarium specimens with both 16- and 32spored sporangia and with mixed C-Fern® Web Manual 9 Introduction features of both C. richardii and C. pteridoides suggest that hybridization occurs naturally (Lloyd 1993). Although a number of homozygous diploid and tetraploid accessions of Ceratopteris are available, the Hn-n strain has been used in most experimental studies (Hickok 1987, Hickok et al. 1995, Hickok et al. 1987). Hn-n is a diploid (n=39) derived from a collection of C. richardii from Cuba (Killip 44595 GH). In addition, two other diploid strains, 176D and ΦN8, are also in current use (see Hickok et al. 1995). All three strains show sexually dimorphic gametophytes, i.e., males and hermaphrodites, with males developing in response to the pheromone antheridiogen (ACe). However, 176D shows only slight sensitivity to antheridiogen as compared to Hn-n and ΦN8, and it consequently has fewer males in multispore cultures. The three strains can be crossed readily. Hybrids between Hn-n and 176D show ca. 90% spore viability while crosses of ΦN8 with either Hn-n or 176D show ca. 30% spore viability. During the past several years, our laboratory has developed an improved diploid cultivar, RN, from a series of crosses and backcrosses. All of the exercises presented here are based on the use of the RN cultivar. The C-Fern Life Cycle Like all homosporous ferns, C-Fern has two independent autotrophic phases: a structurally simple, haploid gametophyte and a vascular, diploid sporophyte. The gametophytic phase, which develops mitotically after germination of the single-celled spore, can be cultured axenically on a simple inorganic medium. Development of this haploid phase is very rapid. Germination occurs 3–4 days following inoculation, and full sexual maturity is attained within 6–8 days from germination. At maturity, the gametophyte consists of a small (2 mm), simple, essentially twodimensional thallus with rhizoids, vegetative cells, and sexual organs (archegonia and antheridia). The archegonium is the female organ; it contains one egg that lies at the base of a small neck sticking out from the surface of the gametophyte. The neck consists of four rows of cells, along with a few “neck canal cells” in the middle. The antheridium is the male sex organ; each contains 16 sperm. In the presence of water, the neck canal cells in mature archegonia burst open, creating a small, open canal leading to the egg. The canal cells’ contents are deposited near the top of the C-Fern® Web Manual 10 Introduction open neck. Meanwhile, the antheridia are also active. In the presence of water they also burst open, discharging motile sperm (spermatozoids). The rapidly swimming sperm are irresistibly attracted by the discharge from the archegonium. In a few minutes, hundreds of sperm can be seen swarming around the neck of the archegonium, and one of them eventually wiggles its way down the neck and fertilizes the egg. After fertilization of the egg, the resulting diploid zygote develops rapidly by mitotic cell division, forming an embryo. Embryos are clearly visible after a few days, and in only 1–2 weeks roots and leaves can be seen on the small diploid sporophytes. The gametophyte soon dies and the sporophyte grows to maturity. It undergoes meiosis and produces spores to continue the life cycle. The pheromone-like substance, antheridiogen (ACe), secreted by developing gametophytes controls differentiation of two distinct sexual forms of gametophytes. ACe is likely biosynthetically related to gibberellins and is effective at extremely low concentrations (Warne and Hickok 1989). In the absence of ACe, gametophytes develop initially as heart-shaped (cordate) females with archegonia and subsequently as hermaphrodites with both archegonia and a few antheridia. In hermaphrodites, a defined meristematic region (notch meristem) is present, and growth is indeterminate until fertilization of an egg occurs. Meristematic activity ceases shortly after fertilization. In contrast to hermaphrodites, gametophytes that mature in the presence of ACe develop into tongue-shaped males that are small, determinate, lack a meristem, and produce large numbers of antheridia. At the vascular sporophyte stage, a C-Fern consists of a short upright stem (rhizome) with roots and leaves (fronds) and reaches a height of 10–40+ cm. In contrast to many ferns, the Ceratopteris sporophyte is not woody and grows rapidly as an annual. Spore production via meiosis occurs within sporangia that are located on the margins of fertile leaves. Upon maturity, spores are produced continually and are unlimited in number. Compared to many ferns, spores are quite large (ca.120 µm) and relatively easy to handle. Because individual haploid gametophytes can be self-fertilized, sporophytes completely homozygous across all genetic loci can be produced in one generation of selfing. Such sporophytes produce an unlimited number of genetically identical spores. If kept dry, spores remain viable for many years. C-Fern® Web Manual 11 Introduction B D A C Figure 1. C-Fern spores. A,B — ungerminated, proximal (A) and distal (B) views; C,D — germinated, showing splitting along trilete mark (C) and emergence of primary rhizoid (D). Spore diameter ca. 120 µm. Notch meristem Archegonium Figure 2. Young C-Fern gametophyte, 5 days from start (DFS) of culture. Spore coat diameter ca. 120 µm. Antheridium Egg Sperm Figure 3. Mature hermaphroditic C-Fern gametophyte with archegonia behind the notch meristem and a single antheridium on the margin, ca. 10 DFS. Spore coat diameter ca. 120 µm. Close-up: view of mature archegonium during fertilization. Sperm enter the open neck canal, uncoil, and move toward the egg. C-Fern® Web Manual 12 Introduction Mulitlayered structure Flagellum Dehisced antheridium Sperm body Figure 5. Individual mature sperm, ca. 8.8 × 5.5 µm. Undehisced antheridium Sperm De-differentiated meristem region Figure 4. Mature male C-Fern gametophyte, ca. 10 DFS. Sperm are released from the numerous antheridia on the surface of the male gametophyte. Spore coat diameter, ca. 120 µm. Young embryo Immature archegonium Young embryo Mature archegonium Region of old archegonial neck Figure 7. Close-up of hermaphroditic C-Fern gametophyte, ca. 3 days after fertilization, showing unfertilized (immature and mature) archegonia and young embryo developing within proliferated archegonial tissue. The remains of the old archegonial neck can be seen at the distal end of the embryo. Figure 6. Hermaphroditic C-Fern gametophyte, ca. 3 days after fertilization, showing a young sporophyte embryo. The embryo is covered by proliferated archegonial tissue. Following fertilization, cell division ceases in the notch meristem region of the gametophyte and cells enlarge. As the sporophyte continues to develop, the gametophyte eventually dies. Spore coat diameter, ca. 120 µm. C-Fern® Web Manual 13 Introduction A Perspective on Plant Reproduction— C-Fern and Teaching the Concept of Alternation of Generations Like other organisms, plants have to reproduce in order to survive as a species, increase in number, and colonize new habitats. Plants reproduce by two basic methods: asexual and sexual reproduction. Generally, the most efficient form of reproduction is asexual. This form typically does not involve a partner and allows an individual plant to produce duplicate copies of itself. Common examples of asexual reproduction can be seen in the ability of new plants to arise from stem cuttings, the production of “baby” plants from specialized buds on the mother plant (e.g., C-Fern and Kalanchoe, or “Mexican hat plant”) and specialized outgrowths from the mother plant, such as potato tubers or strawberry runners. In asexual reproduction, the offspring are genetically identical to the mother plant. Although asexual reproduction is an important aspect of plant biology, sexual reproduction is considered an essential activity of most types of plants. This is because during sexual reproduction the genes are shuffled to produce new combinations in the next generation. This genetic variation has been important in the successful evolution of plants over millions of years. It is also important to human agriculture because it allows the combination and modification of specific traits during the breeding of crop plants. Also, an important portion of our diets consists of the direct products of sexual reproduction—fruits and seeds. Sexual reproduction can involve either one or two plants. When a single plant is involved, different organs develop on the same plant to produce male and female gametes. When two plants are involved, one produces male and the other female gametes. Just as in animals, these gametes can be referred to as egg and sperm. The fertilization of an egg by a sperm to produce a zygote is a critical event in the reproductive process. An important difference between most plants and animals is that gametes are not the immediate products of meiotic cell division, as they typically are in animals. Instead, meiosis in plants yields spores. Spores develop into gametophytes that in turn produce gametes. Union of the gametes (fertilization or syngamy) results in a diploid embryo that gives rise to a mature sporophyte. Mature sporophytes produce spores via meiotic division. This cycle of C-Fern® Web Manual 14 Introduction gametophyte to sporophyte, each with its own role in the process of sexual reproduction, is commonly referred to as the alternation of generations. This reproductive complexity, while advantageous for the plants, can be a difficult concept for students to understand. Nevertheless, there’s a good reason for the added step! Because plants are for the most part stationary, they have developed a different approach than animals to the problem of getting opposite sexual types together to mate. The solution? Spores! As opposed to the direct meiotic production of vulnerable gametes, spores have an increased potential for transport and for survival over extended periods of time and in a variety of environmental conditions. They are suited to a plant’s mode of existence, where a mate may not be immediately available or close at hand when meiosis occurs. When conditions are favorable, meiotically-produced spores give rise to gametophytes and, subsequently, to gametes. To complete the process, plants have evolved a variety of fascinating ways to ensure that fertilization occurs! In C-Fern, students can directly observe the sexual reproductive process and the alternation of generations. The development of gametophytes and sporophytes, production of sperm and eggs, and fertilization can all be easily seen. Although ferns are typically known only by the conspicuous, green, leafy, sporophyte stage, this stage represents only a portion of the sexual life cycle. Meiosis occurs in ferns’ sporangia, borne on the sporophyte, sometimes on the underside of the sporophyte leaf. In contrast to the microspores and megaspores of flowering plants and heterosporous ferns, homosporous ferns such as C-Fern produce only one type of spore. When they are ripe, spores are released from the sporophyte, and, if they land in a suitable environment, they germinate and develop into small (1–2 mm) green, free-living gametophytes. Fern gametophytes may develop as hermaphrodites with both antheridia and archegonia or as distinct male plants with antheridia. When mature and in the presence of water, antheridia burst open and release the sperm cells, which swim in search of a receptive egg. In the archegonium, the egg is located at the end of a short, closed tube or neck. When mature and when water is present, the neck opens and spews out cellular material, including a chemical attractant that draws masses of sperm to the neck opening. The sperm then attempt to swim down the neck and fertilize the egg. After fertilization by a single sperm, the zygote develops by mitotic divisions into a young embryo that can be seen within the now “pregnant” archegonium. Continued development produces a leafy C-Fern® Web Manual 15 Introduction green sporophyte to complete the sexual cycle. All of these stages are clearly observable in C-Fern cultures and are explored fully in the C-Fern investigations dealing with reproduction and differentiation. An understanding of alternation of generations gained from study of the C-Fern life cycle can help students understand the life cycle of flowering plants. In flowering plants, the alternation of generations is more difficult for students to observe. This is because the male and female gametophyte generations have been reduced through evolution to only a few cells. Meiotically produced microspores mature into the pollen, contained within the stamen. The pollen grain contains the much-reduced male gametophyte, consisting of only two cells at maturity. When pollen is transferred to a compatible stigma, the pollen tube grows down the style of the flower. When it reaches the vicinity of the egg, a mitotic division produces sperm. In the developing ovary, meiosis results in a megaspore that then develops by mitotic division into a much-reduced female gametophyte, the embryo sac. The embryo sac contains the egg, along with other nuclei important to the process of embryo formation and seed development. The production of pollen, which can be easily transported, allows mating (pollination) by a variety of mechanisms, including wind, water, insects, and other animals. The specialization of many types of flowering plants, with their intricate adaptations to ensure sexual reproduction, has resulted in a diverse and fascinating group of organisms. If students’ concepts of plant sexual reproduction are first thoroughly grounded in the dynamic representation of alternation of generations represented by C-Fern, they will better understand the flowering plant life cycle as well. C-Fern® Web Manual 16 Introduction Figure 8. Life Cycle of C-Fern C-Fern® Web Manual 17 Culture Instructions for C-Fern® Investigations Culture Instructions for C-Fern ® Investigations Introduction C -Fern®is an exciting and unique organism that is easy to grow using inexpensive materials and simple growing conditions. To help you get started quickly and easily, follow the Quick Start instructions below. To ensure your success with CFern, additional detailed information and options are included in the subsequent sections of this document. Please also see the C-Fern Web site (http://www.C-Fern.org). All of the INSTRUCTIONS presented here are based on the use of the original C-Fern (RN) cultivar. A new cultivar, C-Fern Express, was released in 2009. C-Fern Express was derived from crosses between two Japanese strains of C. thalictroides and exhibits a shorter life cycle time (<60 days) and smaller sporophyte size. With the exception of sporophyte culture, all methods developed for RN are applicable to C-Fern Express. C-Fern branded materials, supplies, teaching aids and investigations are available from distributors of C-Fern products. Quick Start C-Fern® and C-Fern Express cultures are started from spores. Surface-sterilized spores of the wildtype and mutant stocks are available through distributors in 10-mg units. For general use, 4 mL of sterile water should be added to a vial to make a suspension of spores. Spores are then inoculated into 60- × 15-mm petri dishes containing Basic C-Fern Medium. Medium is available pre-made or in powder form. Instructions for preparing the medium are detailed in this manual. Use a sterile, disposable transfer pipet to inoculate each petri dish with 3 drops of the spore suspension. This will generate up to 36 cultures with a standard density of >300 spores per dish. Spores should be spread evenly over the agar surface using a sterile bent paper clip or microbial spreader. It is very important to maintain cultures under continuous light and at a temperature of 28°C (82°F). These conditions are easily achieved by using a 15-W screw-in fluorescent bulb to illuminate cultures within a C-Fern Growth Pod™. This setup is described in these instructions and on the Web (http://www.C-Fern.org). Under these C-Fern® Web Manual 18 Culture Instructions for C-Fern® Investigations conditions, development will proceed as depicted in Figure 8. If the culture temperature is cooler and/or constant illumination is not provided, development will be slower. For C-Fern Express cultures, follow the same basic culture setup as described for original or standard C-Fern. Please take note of the following differences of C-Fern Express. If cultured at 82 ºF, mature gametophytes can be observed at 10 days from sowing spores. While standard C-Fern gametophytes have clear distinctions between the small males and larger hermaphrodites, CFern Express gametophytes are more variable. The basic distinction between the smaller males (no meristem and containing many antheridia) and larger hermaphrodites (a meristem, archegonia and few antheridia) remains. However, some smaller C-Fern Express gametophytes with several antheridia may initially look like males, but subsequently develop a meristem and become hermaphrodites with several antheridia. Water added to gametophyte cultures at 10 days or later will allow fertilization by swimming sperm. One week later, young sporophytes will be visible at the first leaf stage. Culture Instructions 1. Getting Started Detailed written exercises are available from C-Fern suppliers and contain both Teacher and Student versions. Dried C-Fern spores are long lived and are available from C-Fern suppliers in small plastic vials, either as pre-sterilized 5- or 10-mg units or as unsterilized 40-mg units. The pre-sterilized spores are very convenient and easy to use— just add the specified amount of sterile water and sow! If using unsterilized spores, the sterilization procedure is simple and efficient (refer to Step-by-Step Procedure for Spore Sterilization in the Surface Sterilization of C-Fern Spores Section on page 41). C-Fern® Web Manual 19 Culture Instructions for C-Fern® Investigations 2. Determining Sowing Density and Number of Cultures There are approximately 1,250 C-Fern spores per mg dry weight. Standard sowing density is approximately 300 spores per 60-mm petri dish. Table 1 provides information for sowing spores at various densities. Note: Depending upon technique, there is always some spore loss during sowing procedures. Therefore, the spore numbers given are approximate. Table 1. Spore-sowing densities FOR this density 1 USING 10- or 5mg vials ADD this much water (mL) to vial SOW onto petri dish standard 10 4 3 - 36 300+ standard 5 2 3 - 18 300+ 2/3 10 4 2 1 54 200+ 2/3 5 2 2 1 27 200+ ½ 10 2 3 - 18 150+ ½ 5 4 3 - 36 150+ 1/3 10 4 1 2 108 100+ 1/3 5 2 1 2 54 100+ This many drops of spore suspension Plus these additional drops of water1 MAKE about this many petri dish cultures WITH this number of spores per petri dish Additional water is to allow enough liquid (a total of 3 drops) for spreading spores evenly on the agar surface. 3. Preparing the Culture Medium Four options for preparing Basic C-Fern Medium are given below: A) premade bottled medium (melt and pour) B) & C) powdered medium (mix, autoclave or microwave, and pour) D) preparation and use of stock solutions. A. Using Pre-Made Bottled C-Fern Medium Materials: • Bottled C-Fern Medium (Basic C-Fern Medium is available in 160- and 400-mL bottles. C-Fern Medium is also provided in most kits or kit refills.) • Sterile Petri Dishes, 60 × 15 mm (Petri dishes are also provided in most kits or kit refills.) C-Fern® Web Manual 20 Culture Instructions for C-Fern® Investigations • Hot Water Bath • Clean Area for Pouring Dishes (or a sterile bench or laminar flow hood) Melt and pour medium into dishes in advance to allow sufficient time for medium to cool to room temperature and solidify completely. To speed melting, vigorously shake the bottle to break up the medium before placing the bottle in a hot water bath. To melt the medium, loosen the cap and place the bottle into a hot water bath such that the water level is only just above the level of the medium in the bottle. Do not fully submerge the bottle. A cover on the water bath helps the medium to melt faster. In a boiling water bath (100°C or 212°F), the melting time for medium is about 15 minutes for 160 mL and 45 minutes for 400 mL. Medium should be poured in a clean area free from drafts and traffic. Basic C-Fern Medium lacks a carbohydrate source (sugars), so contamination problems, if any, are usually minimal if directions and precautions are followed. Prepare the area by wiping it down with 70% ethanol, 70% isopropanol, or a damp, clean sponge. Open a sleeve of 60 × 15-mm petri dishes by cutting through the end of the sleeve with scissors. Save the sleeve for storage of prepared dishes. Gently swirl the medium in the bottle to be sure that it is thoroughly mixed and completely melted. Remove the bottle cap, then tilt the lid of a petri dish upward just enough to permit pouring of the medium into the dish. Fill dishes about 3/4 full, i.e., 15 mL for 60 × 15-mm petri dishes; 160 mL of medium should prepare about 10 petri dishes, and 400 mL about 25 dishes. Do not under-fill dishes. Sufficient medium is needed for proper growth and development of C-Fern through the sporophyte stage. Replace the petri dish lid and allow dishes to cool undisturbed. Condensation that may form on petri dish lids during cooling is minimized if dishes are poured and cooled in stacks of 5–10 dishes. Once dishes have cooled and the medium solidified, they may be returned to the plastic sleeves, sealed with tape and stored at room temperature for several weeks prior to use in an investigation. Unused dishes may be stored in sleeves in a refrigerator for several months, but do not freeze them. B. Using Powdered Basic C-Fern Medium Materials: • Powdered Basic C-Fern Medium C-Fern® Web Manual 21 Culture Instructions for C-Fern® Investigations Table 3. Composition of basic C-Fern medium stock solutions and final medium Nutrient Components Stock Solution (g/L) Final Medium (mL Stock/L) Final Medium Composition (mg/L) 100 1 10× Macronutrients NH4NO3 1.25 125 KH2PO4 5.00 500 MgSO4•7H2O 1.20 120 CaCl2•2H2O 0.26 26 5 2 200× Micronutrients MnSO4•H2O 0.0500 0.25 CuSO4•5H2O 0.0740 0.37 ZnSO4•7H2O 0.1040 0.52 H3BO3 0.3720 1.86 (NH4)6Mo7O24•4 H2O 0.0074 0.037 10 3 100× Chelated Iron Solution FeSO4•7H2O 2.78 27.8 Disodium EDTA•2H2O 3.73 37.3 a This formulation is based on a medium described in Klekowski, 1969 (Klekowski. 1969. Botanical Journal of the Linnean Society 62: 361-377). Higher concentrations of macronutrients in the stock solution are unstable and may form precipitates, as will most combinations of macronutrient, micronutrient, and chelated iron stock solutions. C-Fern® Web Manual 22 Culture Instructions for C-Fern® Investigations • • • • • • • • • 1-L Volumetric Flask or Graduated Cylinder Distilled or Deionized Water 2-liter Erlenmeyer Flask(s) (per 1 liter medium) Bacto Agar Magnetic Stir Plate and Magnetic Stir Bar pH Meter 1 M NaOH Solution Autoclave or Microwave Sterile Petri Dishes, 60 × 15 mm To prepare 1 L of Basic C-Fern Medium, open the packet of powdered medium and add it to 800 mL distilled water in a volumetric flask or graduated cylinder. Rinse out the powder remaining in the packet with distilled water and bring the medium to a final volume of 1 L. For agar-solidified medium, transfer the nutrient solution to a 2-L Erlenmeyer flask and add 10 g (1% w/v) of Bacto Agar (Difco Laboratories). Note: Some plant tissue culturegrade agars and agar substitutes can result in inhibited or abnormal growth of gametophytes or sporophytes and should, therefore, be avoided. Adjust the nutrient medium to pH 6.0 using 1 M NaOH. Cover the top of the flask with foil and autoclave the nutrient medium at 120°C/20 psi for 15 minutes, or see the protocol that follows for using a microwave. Some of the powdered nutrients or agar may not completely dissolve even after autoclaving or microwaving. This is not a problem. Dispense medium into petri dishes, i.e., about 15 mL in a 60- × 15-mm dish and 40 mL in a 100- × 15-mm dish. The dishes should be about w full. This ensures an adequate nutrient and water supply through to the young sporophyte stage. One liter of nutrient medium should pour about 55 60- × 15-mm dishes and 20 100- × 15-mm dishes. Allow the dishes to cool, completely undisturbed. Condensation on petri dish lids is minimized if the dishes are poured and cooled in stacks. Once the dishes have cooled and the medium has solidified, they may be returned to the plastic sleeves, sealed with tape, and stored at room temperature prior to use in an investigation. Unused dishes may be stored in sleeves in a refrigerator for several months. Do not freeze. C-Fern® Web Manual 23 Culture Instructions for C-Fern® Investigations C. Using a Microwave to Prepare C-Fern Powdered Medium If an autoclave is not available, a microwave can be used to prepare Basic C-Fern Powdered Medium. Although the lower temperatures and shorter times of microwaving cannot guarantee sterility to the degree that autoclaving does, this method has been used repeatedly to successfully prepare contaminant-free medium. Use caution when heating and handling! Wear safety glasses, use gloves, and do not leave the microwave unit unattended! To prepare 1 L of Basic C-Fern Medium, open the packet of powdered medium and add it to 800 mL of distilled water in a volumetric flask or graduated cylinder. Rinse out the powder remaining in the packet with distilled water and bring the medium to a final volume of 1 L. For agar-solidified medium, transfer the nutrient solution to a 2-L Erlenmeyer flask, then add 10 g (1% w/v) Bacto Agar (Difco Laboratories). Note: Some plant tissue culture grade agars and agar substitutes can result in inhibited or abnormal growth of gametophytes or sporophytes and should therefore be avoided. Adjust the nutrient medium to pH 6.0 using 1 M NaOH. Cover the top of the flask with plastic wrap and process the solution in a microwave unit as specified in Table 2. You may need to compensate for microwave units of different powers by adjusting the suggested times. Plates should be poured 3/4 full. Table 2. Steps for using a microwave to prepare 1 L of medium1 Step Microwave Time (1000-W output unit) State of Solution2 Post-Microwave Procedure3 1 5 minutes hot remove, swirl 2 1 minute boiling remove, swirl 3 15 seconds boiling remove, swirl 4 15 seconds boiling remove, swirl 5 15 seconds boiling swirl to mix and pour petri dishes 1 Be sure to use a vessel at least twice the volume of the medium. 2 The solution will boil vigorously in Steps 2–5. The medium must be visually monitored constantly so the power can be reduced or turned off briefly if it starts to boil over. 3 It is important to mix the medium thoroughly by swirling the flask after each step and while pouring the plates. C-Fern® Web Manual 24 Culture Instructions for C-Fern® Investigations D. Preparation of Basic C-Fern Medium From Stock Solutions Materials (Preparation of Stock Solutions): • Macronutrient, Micronutrient, and Fe Salts (refer to Table 3 for list) • Distilled or Deionized Water • 1-L Volumetric Flask(s) or Graduated Cylinder • Microbalance • Magnetic Stir Plate and Magnetic Stir Bar(s) • For Fe Stock Solution: • Hot plate, • 2-L Erlenmeyer Flask(s), • Watch Glass • Storage bottle(s) All components of Basic C-Fern Medium should be prepared using high quality distilled and/or deionized water. Prepare macronutrient stock solution and micronutrient stock solutions separately by dissolving all listed quantities of components (see Table 3) individually, in sequence, into about 800 mL of distilled water; bring to a 1liter final volume. Both macronutrient and micronutrient stock solutions can be autoclaved. Autoclaved stock solutions will keep for over 6 months and should be stored in glass at 4°C. Prepare Chelated Fe-EDTA stock solution by dissolving each component separately in ca. 450 mL of water. On a hot plate, heat the EDTA solution to boiling and then add the hot EDTA solution TO the FeSO4 solution. Cover with a watch glass and boil combined solutions for 1 hour, cool completely, then bring to 1-L volume. Store Chelated Fe-EDTA solution in glass at 4°C. Materials (Preparation of Final Medium): • • • • • • • Macronutrient, Micronutrient, and Fe Stock Solutions 100-mL Graduated Cylinder 10-mL Pipet 1-L Volumetric Flask or Graduated Cylinder Distilled or Deionized Water 2-L Erlenmeyer Flask (per 1 liter of medium) Bacto Agar C-Fern® Web Manual 25 Culture Instructions for C-Fern® Investigations • • • • • • pH Meter Magnetic Stir Plate and Magnetic Stir Bar 1 M NaOH Solution Foil (to cover flask during autoclaving) Autoclave Sterile Petri Dishes, 60 × 15 mm To prepare Basic C-Fern Medium, add the appropriate volume of each of the three stock solutions to about 800 mL of distilled water in a volumetric flask or graduated cylinder and bring to 1-L final volume. For agar-solidified medium, transfer nutrient solution to a 2-L Erlenmeyer flask, add 10 g (1% w/v) agar (Bacto Agar). NOTE: Some plant tissue culture grade agars and agar substitutes can result in inhibited or abnormal growth of gametophytes or sporophytes and should therefore be avoided. Adjust nutrient medium to pH 6.0 using 1 M NaOH. Autoclave nutrient medium at 120°C/20 psi for 15 minutes. Dispense medium to petri dishes. Dishes should be about w full—about 15 mL in a 60- × 15-mm dish and 40 mL in a 100-mm dish. This ensures an adequate nutrient and water supply through to the young sporophyte stage. One liter of nutrient medium should pour about 55 60-mm dishes and 20 100-mm dishes. Allow the dishes to cool, completely undisturbed. Condensation on petri dish lids is minimized if dishes are poured and cooled in stacks. Once the dishes have cooled and the medium solidified, they may be returned to the plastic sleeves, sealed with tape, and stored at room temperature prior to use in an investigation. Unused dishes may be stored in sleeves in a refrigerator for several months. Do not freeze. C-Fern® Web Manual 26 Culture Instructions for C-Fern® Investigations Seven Good Habits for Sowing C-Fern Cultures 1. Work very carefully when sowing spores. Secure the vial in a convenient holder. 2. Suspend spores before every sowing. 3. Immediately sow three uniform drops onto the agar surface. Hold pipet at a constant angle. 4. Do not touch the pipet to the agar surface. 5. In most Investigations, use a sterile spore spreader to distribute spores uniformly over the agar surface. 6. Keep the petri dish lid in place as much as possible. 7. Label dishes with name or initials, the date spores are sown, and the treatment code, if any. 4. Inoculating (Sowing) Cultures Using Presterilized CFern Spores Materials: • Surface Sterilized C-Fern Spores in a Graduated Spore Vial (appropriate pre-sterilized spores are provided in most kits or refills; spores for wildtype and mutant stocks are also available separately.) • Sterile Distilled Water (provided in most kits or refills) • Sterile Transfer Pipet (plastic; provided in most kits or refills) • Sterile Spore Spreader (metal spreader [e.g., a paperclip bent into a T-pin shape] or alternative provided in most kits) • Petri Dishes Containing C-Fern Medium (prepared in advance) Preparing Spores Before opening any spore vial, be sure that all spores are at the bottom of the vial by tapping the bottom of the vial on a hard surface. Transfer the appropriate amount of sterile distilled water to the spore vial using a sterile transfer pipet. Do not return any water to the sterile water bottle. See Table 1 for the amount of water to add and sowing densities. Wet spores C-Fern® Web Manual 27 Culture Instructions for C-Fern® Investigations completely by firmly attaching the cap and inverting the vial two or three times. With the cap on, check the bottom of the vial to be sure that all spores have been suspended. Allow the spores to soak for 15 minutes prior to sowing. Sowing Spores To achieve consistent sowings, spores should be thoroughly suspended between each sowing. Suspend spores gently by drawing the liquid along with the spores in and out of the pipet. To sow, withdraw a small amount of the spore suspension into the pipet and immediately dispense 3 Figure 9. drops—not squirts—onto the agar surface (Figure 9). When sowing, tilt the lid of the petri dish upward just enough to permit access of the pipet tip. Do not touch the agar surface with the tip of the pipet. Resuspend the spores in the vial between each sowing by gently squeezing and releasing the pipet bulb. Spreading Spores To make a sterile spore spreader, bend a paper clip into a “T” shape, as shown in Figure 10. While holding the straight end, wipe the “T” end with the alcohol prep pad and let it air dry. Figure 10. Allow the spreader to rest on the agar surface Figure 11. without pressure and move it gently back and forth across the surface of the agar while rotating the dish slowly with the other hand (Figure 11). The goal is to uniformly distribute spores over the entire surface of the medium. This may require some practice but is worth the effort—evenly spread cultures are easy to observe and work with. C-Fern® Web Manual 28 Culture Instructions for C-Fern® Investigations 5. Maintaining C-Fern Cultures Any number of plant-growing systems can be easily adapted to provide adequate environmental conditions for C-Fern. However, in order to attain the growth and timing of development shown in Figure 1, cultures require correct and controlled temperature and adequate lighting. If your temperature and lighting conditions differ substantially from that indicated here, a test run should be carried out to determine when, under your conditions, specific developmental stages will be present for observation and manipulation. The descriptions of two options, Culture Domes and Growth Pods, follow. A. Culture Domes ® Once cultures are inoculated with spores, place them into Culture Domes consisting of clean plastic greenhouse trays covered with transparent humidity domes. For best results, Culture Domes should be thoroughly clean. Both trays and domes are available from distributors of C-Fern products. Culture Domes serve to reduce the possibility of contamination, variations in temperature and humidity, and permit easy handling of a larger number of dishes. Do not tightly seal the petri dishes, for example with Parafilm, as this can result in poor growth, presumably due to ethylene buildup. B. Growth Pod The C-Fern® Growth Pod™ can replace or complement the standard Culture Dome that was recommended in early versions of the C-Fern Manual. The pod’s reduced space requirement, improved temperature control, and increased portability are highly advantageous, especially in situations where the optimum culture temperature of 28°C (82°F) is difficult to attain using the standard Culture Dome. Growth Pods (without light) are available from distributors of C-Fern products, or you can make them yourself, as follows. The C-Fern Growth Pod is made using insulated, vinyl 6-pack coolers or student lunch boxes that are readily available commercially. When fitted with an interior cardboard box, lined with aluminum foil or tape and covered with a fitted lid of 1/4”clear acrylic, the pod can be filled with six stacks of five petri dishes (60 × 15 mm), enough for a class of 30 or more students. A simple C-Fern® Web Manual 29 Culture Instructions for C-Fern® Investigations lighting fixture consisting of a 6”reflective dome and a switch (small clamp or utility lights work well) and a 15-W screw-in fluorescent bulb is all that is needed. The newer screw-in fluorescent bulbs are long lasting, highly efficient, and feel only warm to the touch, even when left on continuously. The light can be rested directly on the acrylic lid, upon stacks of petri dishes or small blocks on top of the acrylic lid, or it can be suspended over the top of the lid. The height should be adjusted to achieve a 28–30°C (82–86°F) internal temperature. Small, inexpensive digital thermometers are handy to keep track of temperature. Thermometers that have an “outside” temperature probe on the end of a thin Figure 12. C-Fern Growth Pod wire are especially useful. The Growth Pod provides a bright, humid, and warm environment for rapid gametophyte and young sporophyte development. Because of the stacking arrangement of the petri dishes, cultures on the bottom may develop slower than those on the top. Reversing the order of the stacks after 6 or 7 days can minimize this. However, the slight variation in gametophyte size that results from the stacking can be beneficial for observations and use. For local or longdistance transport, the top of the Growth Pod can be zipped closed to reduce temperature and humidity fluctuations. The light is small, portable, and can be plugged in anywhere. C-Fern® Web Manual 30 Culture Instructions for C-Fern® Investigations C. Temperature The optimum temperature for spore germination and gametophytic development is about 28°C (82°F), as measured inside the Culture Dome or Growth Pod. This is somewhat higher than for many other plants. Similar growth and development of gametophytes can be obtained at 26–30°C (79–86°F). However, temperatures lower than this will substantially alter developmental timing; for example, development will take twice as long at 20°C (68°F). It is a good idea to monitor and record the temperature inside your Culture Dome or Growth Pod daily. Control of the temperature inside the Culture Dome or Growth Pod can be achieved by adjusting the distance between the light source and Culture Dome or Growth Pod. Once a suitable temperature is achieved, the height of the lights should remain constant during all phases of culture. A constant temperature within the Culture Dome or Growth Pod reduces the chances of condensation on petri dish lids. If condensation is a problem, cultures may be grown upside down once the sowing water has been absorbed into the agar medium. D. Light Note: Continuous illumination is recommended i. Growth Pod Requirements A screw-in 15-W fluorescent bulb with a simple fixture, such as a clamp or garage light, is long lasting, safe, and easy to use for light and temperature maintenance. See the above description for constructing Growth Pods. ii. Culture Dome Requirements Continuous illumination by two 40-W cool-white fluorescent tubes at a distance of 45 cm from the cultures will accommodate two standard Culture Domes (54 × 27 cm). This will provide about 80 µmoles of photosynthetically active radiation m–2sec–1, depending on the age of the bulbs. Two Culture Domes provide enough space for up to 64 individual 60- × 15-mm petri dishes. Smaller or larger setups can be used to serve individual needs. Temperature inside the Culture Dome is more important than light level, so the distance between the Culture Dome and the light source should be adjusted to obtain a temperature near the optimum. C-Fern® Web Manual 31 Culture Instructions for C-Fern® Investigations iii. Constructing a C-Fern Light Stand With inexpensive and simple materials, you can easily construct a C-Fern light stand. Materials and Tools: • 10' of PVC Pipe (1”internal diameter, ID) • 2 L-Shaped PVC Connectors (1” ID) • 2 T-Shaped PVC Connectors (1” ID) • 4 PVC End Caps (1” ID) • Tape Measure • Hacksaw • Marker (The PVC pipe and fixtures in a variety of sizes are readily available in most self-serve home Figure 13. Construction of C-Fern Light Stand improvement stores.) 1. Mark the pipe to the correct lengths, as follows: • 4 6” light bank “feet” • 2 24” risers • 1 24”or 48” crossbeam 2. Cut the pipe with a hacksaw or PVC pipe cutter. 3. Construct the light bank by securely pushing the pipes into the appropriate Figure 14. Complete C-Fern light bank C-Fern® Web Manual 32 Culture Instructions for C-Fern® Investigations connectors. Adjust the light bank “feet” so they are parallel. Hang the light source from the crossbeam, and you are ready to grow CFern. Do not glue the PVC pipe if you want the light bank to be portable and easy to store. When your light stand is finished, you’ll need a light source. For CFern, use a 2-foot fluorescent fixture (4 F20-W cool-white bulbs) OR a 4-foot fluorescent fixture (2 F40-W cool-white bulbs) OR 2 or more dome reflector fixtures with screw-in 15-W fluorescent bulbs. Light systems suitable for C-Fern are available from distributors of C-Fern products. Grow your C-Fern cultures inside a Culture Dome or Growth Pod under continuous light and at a temperature of about 28°C (82°F). Adjust temperature inside the Culture Dome or Growth Pod by varying the distance between it and the lights. 6. Observing Cultures Observations of germinating C-Fern spores, developing gametophytes, and swimming sperm can be conveniently made at low magnification (10×–40+×) using a stereomicroscope. Illumination from below, i.e., with transmitted light, is best. Because the stage of a microscope with transmitted illumination may become quite warm after extended use, students should be encouraged to turn the base light off and to remove the culture plates from the stage when they are not being observed. If cultures are to be observed with the petri dish lids in place, condensation may form on the inside of the lid and obstruct observation. If spare lids from clean unused dishes are available, these can replace the fogged lids. If spare lids are not available, it is possible to remove the condensation by carefully wiping the lids with a clean, preferably sterile, tissue. If the lids are not kept in place during observation, gametophytes may begin to show signs of drying after being open for several minutes. In this case, it is important to remind students to place the lids back on the cultures when the cultures are not being observed. Observations can also be made using a compound microscope (40×–400×) by making wet mounts of some of the gametophytes. Select and remove gametophytes from the culture medium with a sterile probe or toothpick. C-Fern® Web Manual 33 Culture Instructions for C-Fern® Investigations 7. Sporophyte Culture A. Standard C-Fern (RN cultivar) Sporophytes Although most student investigations using C-Fern concentrate on gametophyte development and sexual reproduction through the early sporophyte stage, 14–21 days following sowing (DFS), extended observations of sporophyte development, including the production of the next generation of haploid spores, are often desirable. Under adequate conditions, sporophytes with mature spores can be grown within 90 DFS. Because of its tropical nature, a large-scale culture of C-Fern sporophytes requires a warm and humid greenhouse environment. However, culture of individual sporophytes can be easily accomplished in a terrarium or other suitable vessel. The following instructions are provided for growing individual sporophytes in 2-L clear plastic beverage bottles. 1. One to 3 weeks after water has been added to the gametophyte culture to facilitate fertilization, young sporophytes should be transferred individually to separate 60- × 15-mm petri dishes containing Basic C-Fern Medium. Transfer can be made with a clean toothpick or probe so that the root end of the sporophyte is imbedded in the agar and the first leaf is above the agar. Use care to avoid contamination. 2. Place the sporophyte cultures in the Culture Dome or Growth Pod under the same lighting and temperature conditions that were used for gametophyte cultures. Sporophytes should remain under these conditions for 2–4 weeks. Usually, it is not necessary to add water, but if the culture begins to get dry, use a pipet to add sterile distilled water as needed. 3. When sporophytes have produced several roots and leaves, they can be transferred to a bottle “terrarium.” If the sporophyte is small enough, both it and the soil mix (see step 4) can be pushed through the mouth of the bottle and adjusted/planted with a long narrow stick, etc. Otherwise, make a circular cut 2 inches below the neck. Cut only w of the distance around the bottle so that a “hinge” is left. The top can now be lifted to provide a larger opening for planting. 4. Place 2 cups of pre-moistened (damp but not wet) ProMix®Potting Soil in the bottom of the bottle. Add 4–6 mediumsized beads of Osmocote® 14-14-14 Fertilizer and mix them into the C-Fern® Web Manual 34 Culture Instructions for C-Fern® Investigations top 2-inch of potting mix. Make a shallow concave cavity (2"deep × 1"diameter) in the center of the bottle. 5. Carefully remove a sporophyte from its petri dish by lifting the agar out of the dish, roots and all. A flat knife or thin spatula works well for this. Remove as much agar as possible without damaging the roots and place the sporophyte (roots down!) in the cavity made in step 4. Gently cover the roots with potting mix and lightly press down on the surface around the sporophyte to give it a firm footing in the potting mix. 6. Add enough distilled water to thoroughly moisten the potting mix and help displace any air spaces between the roots and soil mix. The mix should be moist but not soaked, although excess water will not typically harm C-Fern. Subsequent watering can usually be made as needed, usually on a weekly basis. Use distilled or bottled drinking water, if available. Note: Constant moist conditions will result in mostly vegetative growth. If fertile leaves with spores are desired, a moderate level of water stress will typically cause a change from vegetative to reproductive leaf production. This can be accomplished by allowing the soil to dry substantially between waterings. Place the terrarium under the same lighting conditions used for the gametophyte cultures. Warm temperatures and constant 24-hour illumination work best; cold window sills are not recommended. Cooler temperatures and less light will slow development but still allow growth. Other than watering, as described in step 6, little care is needed. Consult the C-Fern® Web Manual section for additional information on the greenhouse culture and manipulation of sporophytes. B. C-Fern Express Sporophytes C-Fern Express is a new strain of C-Fern® that has been developed to enhance student investigations through to the fertile sporophyte stage of the life cycle. Derived from two Japanese varieties of Ceratopteris thalictroides, C-Fern Express exhibits very rapid sporophyte development. It can be substituted for the RN cultivar in investigations of the fern life cycle. Culture requirements for gametophytes are essentially the same as the standard C-Fern strains, with some slight developmental differences in gametophyte morphology. The following guidelines for sporophyte culture will C-Fern® Web Manual 35 Culture Instructions for C-Fern® Investigations allow studies of the entire life cycle, from spore to spore, within a 60-day period. C-Fern Express sporophytes can be grown to maturity within the same size petri dishes that are used for gametophyte culture. Dishes should be 2/3 full with Basic C-Fern Medium. At 1-2 weeks of age, use a sterile toothpick to transfer young sporophytes to fresh dishes (4 per dish). Maintain the cultures in a Growth Pod or similar container under the same conditions for gametophytes. However, cooler temperatures as low as 75 ºF helps to accelerate maturation, while limiting growth. As the medium dries over the next several weeks, add a small amount of water to keep things moist. Avoid adding excess water that cannot be absorbed by the agar. Monitor the sporophytes weekly and note developmental changes in the leaves. Nutrient and space limitations speed up the change from vegetative leaves (round to oval shaped) to fertile leaves (becoming more finely dissected with narrow lobes). After a few weeks, the edges of the fertile leaves will roll inward. Subsequently, several round sporangia will be observed within and along the length of the rolled edges (use a 10X hand lens or microscope). The processes of spore formation from meiotic division and spore maturation will occur over the next few weeks, until the sporangia and spores are brown. Maturity is marked by a distinct brown line on the underside of the leaf. At this time, spores can be harvested by removing the leaves and drying them out in a clean dry petri dish. Under the conditions described, total time from first starting the cultures to harvesting mature spores is typically 60 days. See the web pages (www.c-fern.org) for images. 8. Additional Information For Educators A. Hints for Large Classes The 40-mg vial of unsterilized bulk spores is a convenient size for larger classes. To sterilize a single batch or multiple batches of this quantity of spores, it is advisable to sterilize each 40-mg quantity using the standard techniques described in the Culture Instructions. After sterilization has been completed, transfer the sterile spore suspension to a sterile test tube of larger capacity (e.g., =20 mL). Then, for each 40-mg quantity, bring the liquid volume up to 16 mL with sterile distilled water. Dispensing 3 drops per dish from a C-Fern® Web Manual 36 Culture Instructions for C-Fern® Investigations sterile transfer pipet will generate approximately 140 small petri plates. Be sure to keep the spores suspended by repeatedly drawing them in and out of the pipet during the inoculation procedure. All of the spore suspension can be sown at one time. However, if laboratories are to last for more than 2 days, it may be advisable to sow only a portion (perhaps one-half) initially. This can be conveniently done by separating the sterilized spore suspension into two equal volumes. One volume can be kept in the dark for 1–3 days, as is done in the DarkStart procedure. In this way, cultures of approximately the same age can be scheduled for an entire week of laboratories. This can even be a convenient way of bridging schedule difficulties caused by weekends or vacation days. B. Independent Student Research Projects C-Fern is an excellent tool for use in independent student research. Projects can be completely independent of the normal classroom activities or may be integrated with group exercises. The principal goals of independent research are to stimulate students' interest in the subject by encouraging them to formulate questions and subsequently to design appropriate experiments that can provide answers to those questions. This directly involves students in the process of science. Perhaps the most important task of the teacher in this situation is to give helpful but minimal advice and guidance in order to avoid a level of frustration that can dampen students’ initial enthusiasm or willingness to undertake a project. The individual investigations contained within the C-Fern®®Manual can serve as a starting point to generate questions to pursue. In particular, some of the open-ended questions that are presented at the end of the exercises may be useful. Some of these, along with others, follow. Another source is the bibliography contained in Part C of the C-Fern Manual, which gives references for a number of separate investigations that involve a variety of experimental approaches. Use of mutagenesis and selection procedures, determinations of the effects of various growth hormones, comparative tests of the responses of wild type and various mutants to environmental stresses and herbicides, and many other examples are contained within the cited references. Emphasize to the students that many other questions, likely the most interesting ones, haven’t even been asked yet! C-Fern® Web Manual 37 Culture Instructions for C-Fern® Investigations To cultivate independence in students, it is perhaps best to provide them with only the minimal materials that are needed for C-Fern culture. These materials could consist of vials of unsterile spores of the wild type or particular mutants, as well as the basic materials for preparing nutrient medium. In this way, students gain more familiarity with the organism by performing necessary tasks that are often completed by the teacher or others prior to students’ involvement with structured exercises. Very good questions often arise from these types of basic manipulations of the organism. Environmental parameters, such as temperature and light, can be conveniently varied by altering the distance between the Culture Dome or Growth Pod and the light source, and/or by utilizing various types of shading material to decrease the amount of incident light on the cultures. Manipulation of other parameters, such as medium composition, pH, sowing density, population composition, light quality, day length, etc., can also be accomplished easily. C. Questions for Discussion • • • • • • • • • • • • • • • How far do sperm swim? How do sperm move? How do sperm find the archegonium? Why are archegonia restricted to the central region of the hermaphrodite? Can sperm respond chemotactically to artificial (chemical) signals? How do sperm reach the egg after finding the archegonium? How do gametophytes limit fertilization to 1 and rarely 2 events? What happens to gametophytes after a fertilization event? What happens to meristem activity after a fertilization event? What type of signal elicited by a fertilization event is responsible for stopping meristem activity? What are the patterns of cell division and expansion that yield the heart-shape of the hermaphrodite? What are the effects of gravity on developing gametophytes? How long will unfertilized gametophytes live? Other than using a bioassay, what approach could you use to show that a chemical substance was responsible for controlling development of sexual types in C-Fern? Do all gametophytes secrete the pheromone? C-Fern® Web Manual 38 Culture Instructions for C-Fern® Investigations • When are gametophytes sensitive to the pheromone? • How could you chemically characterize the active agent? • How could you design a bioassay to detect other agents that control development? • What might be the advantage of controlling the sex ratio in the cultures? • What other factors might be important in controlling the sex ratio in cultures? • How could one determine if there was a genetic difference that controlled sexual type in gametophytes? • How could you test the relative fitness of different genotypes, e.g., wild type versus polka dot? • Can you think of an advantage to having more males at higher densities? How could you test your hypothesis? • How do higher densities result in more males within a population? • What might be the advantage of having fewer males at lower densities? • How do changing proportions of males influence the kinds of mating that can occur? • •What factors limit sporophyte growth at high densities? • Can sporophytes form without fertilization? • Can gametophytes develop directly from sporophyte tissue? • What role does the meristem play in maintaining the hermaphroditic gametophyte? • Can microsurgery on gametophytes demonstrate alternate paths of development? • Can sporophytes develop normally if the young embryo within the archegonium is cut away from the gametophyte? • What effect on development is caused by adding sucrose to the medium? • What factors are important in sporophyte development? • Can sporophyte development be speeded up? • What is the difference between vegetative and fertile leaves on sporophytes? • Do rhizoids respond to the direction of gravity? • Do gametophytes show a phototropic response? • Do sporophytes show a phototropic response? • What wavelengths of light are necessary to induce germination? C-Fern® Web Manual 39 Culture Instructions for C-Fern® Investigations • Does gametophyte growth and development respond to different wavelengths of light? • Which phase, gametophyte or sporophyte, is more important in limiting the distribution of C-Fern? • Under what conditions is vegetative development more important than sexual development? • Do rhizoids have a specific function? • If a meristem is surgically removed, will a new one form? What factors could influence this? • Is distance between gametophytes important during their sexual development? • Do sperm swim best in distilled water or in some other medium? • Following meiosis, how long does it take a spore to mature? • How long can sperm swim? What environmental factors are important in determining this? • What happens when C-Fern cultures are grown upside down or on their side? Why? C-Fern® Web Manual 40 Additional Culture Methods and Techniques Additional Culture Methods and Techniques Surface Sterilization of C-Fern Spores The procedure outlined on the next page describes how to surface sterilize spores that are either collected directly from sporophytes or obtained in bulk, unsterilized lots. Before you begin, Make sure the area is clean and free from drafts and traffic. •Wipe down the area with 70% ethanol, 70% isopropanol, or a damp clean sponge. Materials: • Bulk Unsterilized C-Fern Spores (For information on wild-type and mutant spores, refer to the Sport Reports - Descriptions of C-Fern Mutants section and distributors of C-Fern products.) • Sterile Tube With Conical Bottom • Sterile Transfer Pipets • Sterile Distilled Water • Plain Commercial Laundry Bleach or Equivalent (5.25% sodium hypochlorite solution) • Waste Container • Timer NOTE: For handling larger amounts of spores (15–250 mg) it may be more convenient to use 12- to 15-mL conical centrifuge tubes. Other materials, such as conical microfuge tubes and automatic pipets, may also be successfully used for spore sterilization, but should be tested first. DarkStart Method In some cases, it is desirable to decrease the length of time required to obtain gametophyte material at a particular developmental stage, or give greater control over the timing of particular developmental stages. Because the 10–12 days required to obtain sexually mature gametophytes under standard conditions includes approximately 2 days of imbibition (absorption of water) for dry spores, it is possible to “jump start” cultures by adding water to the vial and immediately placing the sealed vial into a foil bag or covering it with two or more layers of foil. Complete darkness will C-Fern® Web Manual 41 Additional Culture Methods and Techniques Step-by-Step Procedure for Spore Sterilization 1. WEIGH SPORES. Weigh out the spores onto glassine weigh paper and transfer them to a sterile conical tube. Using 4 mL of water, 10-mg spores will sow about 35 petri dishes at a density of 300+ spores per dish. 2. PRESOAK SPORES. Cover the spores with 1–2 mL of distilled water. The spores may be soaked just long enough to become wet, i.e., about 5 minutes, or for up to 24 hours. To hasten the wetting of spores, tightly seal the vial and invert it 2–3 times. 3. REMOVE PRESOAK WATER. Insert the sterile pipet into a conical tube and suspend the spores by bubbling a small amount of air into the water (see Figure 4). While air is slowly bubbling out of the pipet, gently but securely seat the pipet onto the base of the conical tube. Sometimes it helps to gently rotate the pipet tip to seat it properly. Squeeze the bulb to force additional air out of the pipet. When the bulb is released, water should enter the pipet, and the spores should collect around the outside of the base of the pipet tip, provided the pipet is securely seated on the tube bottom (see Figure 4). If you cannot remove the liquid without bringing the spores along, try another pipet. Remember that timing is critical when the sterilizing solution is in the tube! Practice this technique with the presoak water prior to sterilizing the spores. With practice, you should be able to remove the liquid, free of spores, in about 10 seconds or less. 4. SURFACE STERILIZE SPORES. To sterilize, suspend spores in 1–2 mL of 0.875% sodium hypochlorite. Prepare 0.875% sodium hypochlorite by mixing 1 part commercial bleach (5.25%) to 5 parts distilled water. Rinse down the lip and sides of the vial with bleach solution. To ensure that the spore mass becomes evenly suspended, bubble air through a clean, sterile pipet. Surface sterilize the spores for 3 minutes. Remove the bleach solution with a clean, sterile pipet using the technique described in step 3. 5. RINSE SPORES. To rinse the spores, add about 2 mL of sterile distilled water. The pipet used to add the rinse water may be used repeatedly as long as care is taken to prevent contamination with foreign spores. Remove the rinse water with a clean, sterile pipet. Repeat the rinse step 1 or 2 more times. The pipet used to remove the sterile distilled water may be left in the tube for use in sowing spores. 6. SOWING AND SPREADING SPORES.For 10 mg of spores, add 4 mL of sterile distilled water and proceed to the sowing and spreading of spores as outlined in Section 4, Inoculating (Sowing) Cultures Using Presterilized C-Fern Spores. Adjust for the density of spores per dish and the number of dishes sown as needed (see Table 1). C-Fern® Web Manual 42 Additional Culture Methods and Techniques Figure 15. Liquid removal from C-Fern spore suspension.A. Spores soaking in liquid in vialB. Pipet enters liquid while air is expelled. Bubbling before “seating” pipet on bottom prevents spores from entering pipet. Expelling air should be completed while pipet is seated on bottom - enough to subsequently achieve complete aspiration of liquid.C-E. When pipet is seated on bottom, Bulb is released to initiate aspiration. Aspiration of liquid occurs while spores remain behind. F. Spores remain in bottom of vial. C-Fern® Web Manual 43 Additional Culture Methods and Techniques prevent spore germination in most genotypes (the dark germinator stock, dkg is the exception) while still allowing normal imbibition. After 3–7 days in darkness, when spores are sown and placed under lights, germination will occur about 2 days earlier than with the standard light sowing technique. DarkStart can be used to pre-start a lab so that students sow fully imbibed spores and can more easily observe and/or measure all of the early stages of development. For example, spores “DarkStarted” on a Friday, Monday, or Tuesday could be sown and exposed to light the following Friday to ensure initial germination by Monday and complete germination by Wednesday. DarkStart also increases synchrony of germination in cultures and reduces the frequency of male gametophytes. Dry Sowing of Presterilized C-Fern Spores As an alternative to using a liquid suspension of spores for inoculating petri dishes, spores can be sown with a sterile cotton swab. Gently touch the tip of the swab to dry spores in the bottom of the vial. Disperse onto the culture medium by gently tapping the stem of the swab with a finger. Although this method does not produce a uniform distribution or density, it can be useful to quickly establish a few cultures without using the entire vial of spores. Liquid Culture of C-Fern Gametophytes For liquid culture of gametophytes, use the same conditions of temperature and light as with the agar-solidified culture medium. Inoculate surface-sterilized spores into sterile medium without agar at a rate of 3–10 mg per 100 mL and cap the flask securely with foil. In general, the flask size should be about twice the volume of medium used, e.g., 500 mL of medium in a 1-L Erlenmeyer flask. To reduce adhesion of spores and of very young gametophytes to the walls of the culture flask, allow cultures to develop without shaking for 7 days after inoculation. After 7 days, place cultures on a shaker at 50–100 rpm, depending on the volume of solution. If the cultures are not shaken, they will release sperm and rapidly fertilize as soon as gametophytes become sexually mature. Mutagenesis of C-Fern Spores Large numbers of C-Fern spores are readily mutagenized by chemicals (e.g., EMS) or X-irradiation as a means to enhance mutation frequency for screening or selection protocols. For C-Fern® Web Manual 44 Additional Culture Methods and Techniques environmental- or chemical-based selections or screenings, the response of unmutagenized C-Fern should be thoroughly tested. Complete dose response curves should be used to identify the appropriate selection conditions that permit clear discrimination of a mutant from the wild-type response. To identify visible mutations (e.g. pigmentation, morphology), a thorough understanding of the variation in gametophytic development is necessary. With lethal or near-lethal selection conditions, e.g., with the herbicide paraquat, mutagenized spores may be sown at a density of 10 mg spores (or greater) per large petri dish (100 × 20 mm). With nonlethal selection conditions, sow at a rate of 5 mg spores (or less) per large petri dish to permit clear observation of individual gametophytes. 1. Ethyl Methane Sulfonate (EMS) Mutagenesis To initiate ethyl methane sulfonate mutagenesis, you will need the following: C-Fern Spores 200 mM EMS (ethyl methane sulfonate) To prepare 1 mL of 200 mM EMS solution: 21.7 µL EMS (MW=124.2, d=1.1452, 1 M=108.5 mL/L) 978.3 µL 100 mM Phosphate Buffer, pH 7.00 To make 200 mL of 100 mM Phosphate Buffer, pH 7.00: 39 mL 200 mM NaH2PO4•H2O (2.76 g/100 mL) 61 mL 200 mM Na2HPO4 (2.84 g/100 mL) 100 mL Distilled Water 0.4M Sodium Thiosulfate (Na2S2O3) Sterile Conical Tubes or Vials (e.g., glass centrifuge tubes with a uniform,conical inside bottom—either autoclaved or foil covered and dry sterilized at 120°C for 2 hr; plastic, disposable centrifuge tubes; or plastic, conical vials) Sterile Pipets (e.g., sterile plastic pipets or autoclaved glass pipets with sterile, rubber bulbs) Other Supplies for Surface Sterilization of Spores (refer to the section on Surface Sterilization of C-Fern Spores in the Culture Instructions) Protective Clothing and Materials Fume Hood C-Fern® Web Manual 45 Additional Culture Methods and Techniques Caution: EMS is a potent mutagen and suspected carcinogen! Use proper protective clothing, eye protection, gloves, and containment tray. Carry out all mutagenesis procedures in a properly functioning and safe fume hood according to your local institutional safety requirements and regulations. Sodium thiosulfate has been used to neutralize EMS spills, treatment solutions, and contaminated glassware after mutagenesis procedures. Use an excess volume of 0.4 M sodium thiosulfate to assist in neutralization of EMS. Dispose of unneutralized and neutralized EMS waste in a manner consistent with appropriate institutional safety regulations and requirements. Procedures 1. Weigh out C-Fern spores to be mutagenized and transfer them to a conical centrifuge tube. Treat up to 250 mg of spores per tube. 2. Prepare EMS-Buffer solution fresh. Mix appropriate amounts of buffer and EMS, and vortex very thoroughly and carefully. Note: Because EMS is difficult to dissolve in buffer, it is very important to vortex the mixture exhaustively. If EMS solution is properly mixed, spores should mostly sink to the bottom after wetting and settling. If spores are mostly floating, mixing was not adequate. 3. Add EMS-Buffer solution to completely cover spores. Treat spores for 36 hr at room temperature. 4. Remove EMS solution and wash spores three times with about 2 mL sterile distilled water, using the technique described in the Surface Sterilization of C-Fern Spores section. Neutralize all liquid waste and glassware surfaces with 0.4 M sodium thiosulfate solution. 5. Surface sterilize spores according to procedures in the Surface Sterilization of C-Fern Spores section. Inoculate mutagenized spores according to directions below. 2. X-Ray Mutagenesis To initiate X-ray mutagenesis, you will need the following: Materials: • C-Fern spores • Sterile Conical Tubes or Vials (e.g., glass centrifuge tubes with a uniform,conical inside bottom—either autoclaved or foil covered and dry sterilized at 120°C for 2 hr; plastic, disposable centrifuge tubes; or plastic, conical vials) C-Fern® Web Manual 46 Additional Culture Methods and Techniques • Sterile Pipets (e.g., autoclaved, glass, disposable Pasteur pipets with sterile, rubber bulbs; sterile plastic pipets) • Sterile Distilled Water • Sterile Glass Petri Dish (100 × 20 mm) • Plastic Sandwich Bag or Parafilm • X-Ray Machine Procedures 1. Weigh out C-Fern spores to be mutagenized and transfer them to a conical centrifuge tube. Up to 250 mg of spores may be treated per tube. Add sterile distilled water and soak spores for 24 hr prior to sterilization and irradiation. 2. Surface sterilize spores according to Surface Sterilization of CFern Spores in the Culture Instructions. Using a minimal amount of sterile water, transfer surface-sterilized spores to a sterile glass petri dish. Seal dish with Parafilm or place dish inside a plastic sandwich bag. 3. Irradiate spores with 25,000 Roentgens or about 555 Roentgens min–1for 45 min. After irradiation, sow spores as usual. After these X-irradiation conditions, spore germination at 10 days following sowing should be about 80% of the control. A screen with 10–5 M fluorodeoxyuridine gives a mutant frequency of about 2 × 10–5 . Controlled Fertilization: Selfing and Crossing Techniques 1. Self-Fertilization Individual hermaphroditic gametophytes have both archegonia and antheridia and can readily self-fertilize under the appropriate conditions. To insure selffertilization and eliminate the possibility of cross-fertilization, individual spores or sexually immature gametophytes should be transferred to petri dishes (isolated) and cultured until sexually mature, i.e. 10–12 days following sowing. Use a stereomicroscope and sterile dissecting needle for the transfers. To accomplish self-fertilization, add 0.1–0.5 mL sterile distilled water directly on top of the sexually mature hermaphrodite. Within a few minutes, if antheridia are fully-developed, spermatozoids should be observed swimming and swarming around the area of the notch meristem where mature archegonia C-Fern® Web Manual 47 Additional Culture Methods and Techniques are found. If gametophytes are floating in the water, they can be submerged with a sterile dissecting needle to ensure that the archegonia are covered with water. After 3–4 days in culture, swollen archegonia containing young embryonic sporophytes will be observable—typically one, sometimes two, per gametophyte. If some isolate cultures do not contain embryos, they can be rewatered. Root and leaf formation will be evident within 1 week after embryos are observed. Maintain cultures until the sporophytes are large enough to be transferred to the greenhouse or a terrarium (e.g., bottle culture). 2. Cross-Fertilization Accomplishing cross-fertilizations or hybridizations between specific stocks is easy if you follow procedures carefully and take care with regard to gametophyte age and developmental stage. Cultures to be used as male stocks should be 10–18 days DFS (under standard culture conditions). At this age, the male stock should contain well-defined tongue-shaped (spatulate) males with numerous antheridia. The female stock should be 8–12 DFS (under standard culture conditions) and contain heart-shaped (cordate) gametophytes with one to three archegonia and few, if any, mature antheridia. There is a very brief stage of development at which there are 1–2 mature archegonia and no mature antheridia, so the cordate gametophytes are effectively female. Careful attention to gametophytic development age ensures high spermatozoid number and activity for the male parent and 1–2 receptive archegonia for the female with a minimal chance of selfing. In addition, all manipulations should be carried out in a reasonably rapid and efficient way. Set up females for crosses first. (Removing cultures from a warm culture room to a cooler area can result in premature release of sperm and establishment of archegonial receptivity.) Set up isolate cultures of the female stock by transferring individuals at the appropriate developmental stage to separate petri dishes. Use a stereomicroscope and sterile dissecting needle for the transfers. Place the gametophytes on the agar surface with their archegonia facing up. It is also a good idea not to transfer any females that are in contact with males on the stock dish. Once all females are transferred, add 1–2 pipets of sterile distilled water to cover the gametophytes on the male stock culture dish. With a stereomicroscope, monitor sperm release. Maximum sperm C-Fern® Web Manual 48 Additional Culture Methods and Techniques activity should be evident within 5 min. With a sterile transfer pipet, carefully collect the sperm suspension and place 1–2 drops of it on each female to be crossed. (Avoid picking up gametophytes and other debris.) Use a microscope to check all attempted crosses. If female gametophytes are floating in the water, they can be submerged with a sterile dissecting needle to ensure that the archegonia are covered with water. Remove any males that were carried in with the sperm suspension. Sperm should be seen swarming around receptive archegonia. It is a good practice to discard any attempted crosses that do not show immediate swarming of sperm since these may subsequently self-fertilize. At 3–4 days following the crossing attempts, check the females for successful fertilizations as evidenced by obvious swollen regions (embryos) at the base of the archegonia. Discard all females that do not carry embryos. If the gametophytes, techniques, and conditions used for crosses are ideal, 90–95% of crossing attempts can be successful. Maintain cultures until the sporophytes are large enough to be transferred to the greenhouse or a terrarium (e.g., bottle culture). Crosses must be confirmed by segregation from the F1sporophyte or by use of a marker stock. Preparation of Semipermanent Slides for Observation and Analysis of C-Fern Gametophytes Semipermanent slides of gametophytes suitable for later observations or archival documentation can be prepared by mounting gametophytes in a Hoyer’s/Acetocarmine mixture. Hoyer’s medium clears and preserves tissue; acetocarmine is one of the few stains stable in the presence of Hoyer’s medium. To prepare the slides, you will need the following: Materials: • • • • • • Hoyer’s Medium 0.5% Acetocarmine Stain Plastic Dropper Bottle Slides Coverslips Source of Iron Oxide or Rust (e.g., a rusty nail) C-Fern® Web Manual 49 Additional Culture Methods and Techniques Prepare mounting mixture fresh by mixing 4 parts Hoyer’s medium and 1 part acetocarmine stain (1 mL of this mixture should be sufficient for 7–8 slides, using 22- × 22-mm coverslips). For the acetocarmine to stain properly, add a small amount of iron oxide to the Hoyer’s/acetocarmine mixture. Do this by dipping an old rusty dissecting tool (e.g., forceps) or a rusty, iron nail into the mixture until a sufficient amount of iron oxide has leached off. Experiment some to determine the correct amount of iron—too little gives very weak staining, but too much yields excessive precipitation and a strongly colored background. Place 4–10 drops of the Hoyer’s/acetocarmine mixture onto a slide. (For best results, be sure slides are very clean.) Transfer gametophytes from the culture dish to the mixture. Submerge gametophytes in the mixture. Put coverslip in place. If the gametophytes are large or convoluted, it may be difficult to avoid trapping some air bubbles. Place completed slides on a tray to dry undisturbed for several days. Full clearing and staining of gametophytes may take more than several days. Retain the remaining mixture to add to those slides that dry out excessively. Slides prepared with a sufficient amount of Hoyer’s/acetocarmine can keep for well over 10 years. To prepare acetocarmine, you will need the following: • 45% Glacial Acetic Acid 100 mL • Carmine 0.5 g In a fume hood, boil acetic acid and carmine gently for 5 min in a beaker covered with a watch glass. Shake the mixture occasionally as it cools to room temperature. Filter cooled solution through #2 Whatman paper. Filtration takes a long time, and several changes of paper are usually necessary. Store acetocarmine stain in brown glass at room temperature. To prepare Hoyer’s Medium, you will need the following: • • • • Distilled Water 50 mL Gum Arabic (acacia) 30 g Choral Hydrate 200 g Glycerin 16 mL Dissolve gum arabic completely in the distilled water. Then, and only then, add and completely dissolve the choral hydrate. Then add glycerin and mix well. Medium may be diluted as needed with small amounts of distilled water. Before using Hoyer’s medium, let it C-Fern® Web Manual 50 Additional Culture Methods and Techniques stand undisturbed for several days in order to clarify. Store mixture at 4°C. Caution: Choral hydrate is used as a hypnotic and sedative. Abuse may lead to habituation or addiction. Some individuals are extremely sensitive to the vapors of Hoyer’s medium and develop headaches or other symptoms. Greenhouse Culture of C-Fern Sporophytes To set up greenhouse cultures of C-Fern sporophytes, you will need the following: Greenhouse Containers, Cubes, or Pots Commercial Potting Soil, or Mixture: Pro-Mix Potting Soil (15-9705) Potting Mixture (2 parts peat moss, 1 part vermiculite, and 1 part perlite). Adjust to about pH 6.5 with 100 g agricultural lime per 10 gal of potting mixture. Newly transplanted sporophytes should be covered with a clean culture dome or inverted petri dish lid for 3–5 days. 1. Transplantation Young sporophytes grown from agar culture can be transplanted to the greenhouse when several well-developed roots and about 10 leaves are evident. The mineral nutrient medium used for gametophytes will not support long-term sporophytic growth. Subculturing very young sporophytes into fresh nutrient medium may be necessary to generate sporophytes that are large enough for successful transfer to the greenhouse. For initial transplanting, transfer sporophytes to small potting containers (cubes, etc.) that contain moist Pro-Mix Potting Soil or a potting mixture. ProMix®gives excellent results when used as is. Other potting mixtures should be adjusted to pH 6.5 as indicated above. The potting containers can be placed in a greenhouse tray containing 1–2 inches of water. When roots begin to emerge from the holes in the cube, the sporophyte should be transplanted to a 6to-8-in pot. Soil should be kept moist-wet at all times; an automated watering system helps. At all stages of growth, Ceratopteris sporophytes require warm temperatures and high humidity; it’s a tropical fern! C-Fern® Web Manual 51 Additional Culture Methods and Techniques 2. Fertilization A low rate of fertilization is required for Ceratopteris and most other ferns. We achieve good growth and fertile frond production with PETERS General Purpose Fertilizer (20-20-20 N-P-K). A solution of 9.6 g/10 gallons should be applied once weekly by saturating the pots or other containers. A regular watering schedule should also be maintained so that the pots do not dry out. Cloning of C-Fern Sporophytes Ceratopteris produces buds on the margins of fronds. These can be readily cultured to maintain specific stocks for spore production or for use as experimental tissue. Larger buds that have developed into small plantlets can be pinched from the mother plant and transferred to small potting cubes containing ProMix. Maintain the buds on a standard greenhouse mist bench, or alternatively, keep them moist and covered. Once the root system is well developed (usually about 14 days), the buds can be directly transplanted to potting mix as described previously. Smaller buds (<1 cm) can be surface sterilized and temporarily cultured on agar medium until they reach adequate size for greenhouse transplantation, as described below. To clone sporophytes from buds, you will need the following: • • • • • • • Sterile 50-mL Plastic Centrifuge Tubes Sterile Pipets Sterile Distilled Water Sterile Forceps Household Bleach (5.25% sodium hypochlorite) Waste Container Container for Floating Buds (e.g., cup with plastic sandwich bag cover) • Culture Dishes with Basic C-Fern Medium Procedures Collect and Prepare Buds. Collect smaller marginal frond buds from greenhouse plants. Buds should be no larger than about 1 cm and most of the leaf tissue from the mother plant should be trimmed into the shape of a small triangle. In addition, leaf tissue from the bud should be removed. As buds are collected, float them in water in a container covered with a plastic sandwich bag. C-Fern® Web Manual 52 Additional Culture Methods and Techniques Sterilize and Prepare Buds. Prepare sterilization solution (1 part household bleach to 4 parts distilled water). Carefully transfer buds to the bottom of a sterile 50-mL centrifuge tube—do not touch sides of tubes. Do not sterilize more than about 50 buds at one time. With a sterile pipet add sufficient sterilizing solution to completely cover buds. Shake tube for 1 min to thoroughly wet buds with solution. Carefully decant sterilizing solution to the waste container; use the sterile cap of the centrifuge tube to retain buds. Rinse and decant buds three times with about 25 mL of sterile distilled water. Culture Buds. Wash buds into a petri dish using sterile distilled water. Plant buds to the medium with forceps. Push the root end of the bud slightly into the agar to establish good contact. If small buds are cloned, be sure that the triangle of mother sporophyte tissue is parallel to the agar surface. Once young sporophytes have a well-developed root system and 10 leaves, they can be transplanted to the greenhouse or a terrarium (e.g., bottle culture). Collection of C-Fern Spores Sporophytes produce a heteromorphic sequence of increasingly filiform fronds characterized by an inrolled margin or false indusium that covers developing sporangia. When this inrolled margin becomes brown (brown-stripe stage), the spores have developed sufficiently for harvest. To harvest spores, you will need the following: • Scissors, • Razor Blades, or Scalpel Blades • 70% Alcohol, or Bunsen Burner or Alcohol Lamp (to sterilize utensils) • Collection Vessels (e.g., glassine envelopes, petri dishes, plastic tubs) • Sieves—U.S.A. Standard Testing Sieves No. 100 and 170 (with openings of 150 and 90 µm, respectively) • Collection Pan • Storage Vessels (clean and dry—e.g., glass jars, screw-cap plastic tubes) • 25-mL Glass Graduated • Cylinder • Forceps Procedures C-Fern® Web Manual 53 Additional Culture Methods and Techniques For harvesting spores from individual plants, clip the mature fertile fronds and place (stuff!) them into a labeled glassine envelope. Various sizes of glassine envelopes can be obtained from photographic supply houses. The envelopes should be well sealed to prevent leakage of spores around the seams. Other collection vessels such as large plastic petri dishes may also be used. Between collections, utensils should be sterilized with alcohol or flame to prevent cross contamination of genotypes. For harvesting mass collections of spores, as from many clones of the same genotype, clip mature fertile fronds and place them in a large plastic tub. It is best to use a tub that can be easily sterilized, such as by autoclaving, to prevent cross-contamination of genotypes. Dry material at about room temperature for about 48 hr or longer, depending on humidity. When the material is sufficiently dry, it may be stored indefinitely at room temperature; i.e., for at least several years. Refrigeration is not recommended. Small quantities of spores can be decanted directly from glassine envelopes while retaining sporophytic material. Gentle agitation can help release additional spores. For larger collections, spores may be processed by sieving through (in order) No. 40 and No. 60 sieves into a collection pan. These sieves have 425- and 250-µm openings, respectively. Small batches of dried fertile fronds are placed onto a No. 40 sieve and lightly ground with the base of a 25mL graduated cylinder. This grinding action should release nearly all of the spores by breaking up the fertile frond material without completely pulverizing it. The spores should fall through the sieves to the collection pan while the fertile frond material is retained. An additional pass through a No. 100 sieve (150-µm opening) may help purify spores that are heavily contaminated with pulverized fertile frond material. Store dry spores at room temperature in the dark in screw-cap containers. C-Fern® Web Manual 54 Sport Reports - Descriptions of C-Fern Mutants Sport Reports - Descriptions of C-Fern Mutants The term sport is sometimes used to refer to unusual forms of plants or animals that typically arise by mutation. This section of the manual includes a short descriptions, suggestions for culture and uses and references for a variety of C-Fern mutant lines. Mutant: polka dot; gene symbol, cp This is a very striking visual mutant that exhibits a distinct green polka-dot appearance in cells of both gametophytes and homozygous sporophytes when viewed with a low-power microscope. In sporophytes, older leaves of individuals homozygous for this mutation have an attractive silver-green appearance. Although there is a slight reduction in growth and a decrease in spore viability in spores produced from homozygotes, gametophytes carrying the polka-dot mutant and homozygous polka-dot sporophytes grow nearly as well as the wild type. This recessive trait has been observed in several independent selections that have been generated using both X-rays and the chemical mutagen EMS. A study of the trait, using transmission electron microscopy, was not successful in discerning the structural basis of the phenotype. The phenotype is associated with a clumping of chloroplasts and other organelles around the nucleus. It may involve some disruption of the cytoskeleton. The pleiotropic effect of reduced spore viability is associated with somewhat fragile spore walls. Different degrees or strengths of the clumping phenotype are positively associated with increased spore wall weakness. Vaughn, K. C., L. G. Hickok, T. R. Warne, and A. C. Farrow. 1990. Structural analysis and inheritance of a clumped-chloroplast mutant in the fern Ceratopteris. Journal of Heredity 81:146–151. Mutant: pale; gene symbols, pal1 Ghost-like pal1 gametophytes appear to have decreasing amounts of chlorophyll as they enlarge. Nonetheless, they grow to sexual maturity within 2 weeks and can self- or cross-fertilize. At 1–2 weeks, pal1 gametophytes are slightly smaller than wild type, and their pale phenotype makes them clearly recognizable in a segregating population (use of transmitted light is best). This is a C-Fern® Web Manual 55 Sport Reports - Descriptions of C-Fern Mutants recessive mutation in sporophytes, and leaves of pal1/pal1 homozygotes show a yellow green phenotype when viewed with reflected light (from the side or top). These attractive sporophytes are quite viable, but slower growing than wild type. This EMSinduced mutation is available only in an F1 hybrid stock. The F spores can be used very effectively as a substitute for polka dot (cp) in basic Mendelian genetic labs. An additional pale mutant (pal2) is available in combination with pal1. These mutants are non-allelic and unlinked. Segregation from the F11results in a 1:3 (green:pale) gametophyte segregation ratio. Random fertilizations produce an F2 sporophyte ratio of 9:7 (green:pale). Mutant: dark germinator; gene symbol, dkg1 This is a fascinating mutation that shows a reversal in the light requirement for germination of C-Fern spores. In wild type, light (red) is necessary for spore germination. However, in spores containing the dkg1 mutation, germination occurs readily in the complete absence of light. Use of this mutant allows investigations of gametophyte growth and development to be conducted in the dark or by using specific wavelengths of light without the requirement to expose spores to red or white light to initiate germination. Another interesting aspect of this mutation is that germination in white light is substantially reduced relative to its germination in the dark. Cooke, T., L. Hickok, W. J. Vanderwoude, J. Banks, and R. Scott. 1993. Photobiological characterization of a spore germination mutant with reversed photoregulation in the fern Ceratopteris richardii. Photochemical Photobiology 57:1032–1041. Cooke, T. J., L. G. Hickok, and M. Sugai. 1995. The fern Ceratopteris richardii as a lower plant model system for studying the genetic regulation of plant photomorphogenesis. International Journal of Plant Science 156:367–373. Mutant: non-etiolated; gene symbol, det30 If C-Fern gametophytes are given an initial light exposure (1–2 days under standard conditions) to initiate germination and then placed in the dark, their growth form will be dramatically altered. The alteration in some ways resembles the etiolation response that is well known for higher plants, in which plants subjected to dark conditions grow substantially longer than those in light. In darkgrown C-Fern gametophytes, some of the basal cells undergo C-Fern® Web Manual 56 Sport Reports - Descriptions of C-Fern Mutants extreme elongation, which results in strap-shaped structures consisting of a few highly elongated cells just above the spore coat and a pad of smaller cells at the tip of the gametophyte. In gametophytes containing the det30 mutation, which was induced by X-rays, this elongation response is reduced. In contrast to flowering plants, both C-Fern wild type and det30 gametophytes grown in the dark still have the capacity to synthesize chlorophyll, although they are typically a very light green. Cooke, T. J., R. H. Racusen, L. G. Hickok, and T. R. Warne. 1987. The photocontrol of spore germination in the fern Ceratopteris richardii. Plant and Cell Physiology 28:753–759. Cooke, T. J., L. G. Hickok, and M. Sugai. 1995. The fern Ceratopteris richardii as a lower plant model system for studying the genetic regulation of plant photomorphogenesis. International Journal of Plant Science 156:367–373. Murata, T., A. Kadota, and M. Wada. 1997. Effects of blue light on cell elongation and microtubule orientation in dark-grown gametophytes of Ceratopteris richardii. Plant Cell Physiology 38:201–209. Mutant: day-night responder; gene symbol, dnr1 Gametophytes and homozygous sporophytes containing this mutation accumulate massive amounts of starch in their plastids when grown under constant light conditions. This can be easily observed by viewing the plastids under a compound microscope; staining with I2KI can enhance observations. The massive starch grains give the plastids a very lumpy appearance. The accumulation of starch suggests that under constant light conditions the cells are unable to use photosynthate effectively and, as a result, gametophyte growth is severely impaired. This is a conditional mutation in that near-normal growth and depletion of the abnormal starch accumulation occur when gametophytes are grown under day-night conditions. Sporophytes grow well under normal greenhouse day-night conditions. This X-ray induced mutation is recessive in sporophytes. Mutant: abscisic acid tolerant; gene symbol, abr48 This mutation confers tolerance to the typical effects of abscisic acid (ABA) on gametophytes. Wild-type gametophytes cultured in the presence of ABA exhibit multiple effects involving decreased C-Fern® Web Manual 57 Sport Reports - Descriptions of C-Fern Mutants growth rate and altered development. The number of antheridia on hermaphroditic wild-type gametophytes is reduced and the number of rhizoids is increased. In some wild-type hermaphrodites, the presence of ABA can cause the marginal notch meristem to become more centrally located. This results in an interesting pattern of growth that resembles a tube sock, with the tip of the toe representing the position of the meristem. The number of male gametophytes is also reduced. These effects, which can be induced in the wild type at a concentration of 5.0–50.0 mM ABA, are not evident in the mutant. Hickok, L. G. l985. Abscisic acid-resistant mutants in the fern Ceratopteris: Characterization and genetic analysis. Canadian Journal of Botany 63:1582–1585. Warne, T. R., and L. G. Hickok. 1991. Control of sexual development of Ceratopteris richardii: antheridiogen and abscisic acid. Botanical Gazette 152:148–153. Mutant: maleless; gene symbol, her1 The her1 mutation, which is one of many mutations that affect sexual differentiation in C-Fern, was induced by X-irradiation of spores. The effects of this mutation can be seen clearly in populations of gametophytes. Populations of wild-type C-Fern gametophytes show two sexual types, males and hermaphrodites. In contrast, this maleless mutant does not contain male gametophytes in populations. This mutation renders gametophytes insensitive to the presence of the male-inducing pheromone, antheridiogen (ACe). On the other hand, development of hermaphrodite gametophytes occurs quite normally and appears no different from the wild type. It is also possible to see the effects of the mutation by using staggered sowings, over time, of spores on the same petri dish. For direct testing, it is possible to obtain a crude source of antheridiogen by growing multispore cultures of gametophytes for 2–3 weeks on basic C-Fern medium and then extracting the liquid from the medium. Extraction can be readily done by taking the older cultures, scraping most of the gametophyte material away, and then freezing the remaining agar. After freezing, the agar can be thawed to effectively separate the solid portion from the liquid. The solution can then be filtered and used as a portion of the liquid to formulate new antheridiogensupplemented C-Fern medium, which can be referred to as CFM+A. If spores of the wild type are sown on CFM+50% ACe, most C-Fern® Web Manual 58 Sport Reports - Descriptions of C-Fern Mutants of the gametophytes in the culture will be male. In contrast, if spores carrying the maleless her1 mutation are sown on such medium, all of the gametophytes will be hermaphrodites. Banks, J. A., L. Hickok, and M. A. Webb. 1993. The programming of sexual phenotype in the homosporous fern, Ceratopteris richardii. International Journal of Plant Science 154:522–534. Banks, J. A. 1997. Sex determination in the fern Ceratopteris. Trends in Plant Science 2:175–179. Warne, T. R., L. G. Hickok, and R. J. Scott. 1988. Characterization and genetic analysis of antheridiogen insensitive mutants in Ceratopteris richardii. Botanical Journal of the Linnean Society 96:371–379. Mutant: highly male; gene symbol, him1 Initial characterization of this EMS-induced mutation suggests that it is highly sensitive to the pheromone, ACe. Typically, C-Fern gametophytes always develop as hermaphrodites when cultured as isolates. In contrast, him1 types can spontaneously develop as males in isolate culture, and there are correspondingly higher numbers of males in multispore cultures. In addition, him1 hermaphrodites tend to have much higher numbers of antheridia, especially as they age. The higher numbers of males and antheridia combine to produce cultures that have the capacity to produce many sperm that can be observed in large masses. Banks, J. A., L. Hickok, and M. A. Webb. 1993. The programming of sexual phenotype in the homosporous fern, Ceratopteris richardii. International Journal of Plant Science 154:522–534 Banks, J. A. 1997. Sex determination in the fern Ceratopteris. Trends in Plant Science 2:175–179. Warne, T. R., L. G. Hickok, and R. J. Scott. 1988. Characterization and genetic analysis of antheridiogen insensitive mutants in Ceratopteris richardii. Botanical Journal of the Linnean Society 96:371–379. Mutant: salt tolerant; gene symbol, stl2 This mutation, which was induced with X-rays, shows an interesting collection of co-segregating traits. It was selected for tolerance to salt (NaCl), and gametophytes carrying the mutation show a high level of tolerance to this agent. Sporophytes also show NaCl C-Fern® Web Manual 59 Sport Reports - Descriptions of C-Fern Mutants tolerance, but at a lower level and in a semi-dominant fashion. In addition, stl2 confers tolerance to magnesium salts, but sensitivity to moderate levels of potassium in the medium. Sensitivity of the wild type to sodium salts is associated with the level of calcium in the medium. In the presence of 150 mM NaCl, the calcium level in basic C-Fern medium results in very poor growth and necrosis of the wild type with little apparent effect on the mutant. At higher calcium levels commonly found in other plant nutrient formulations, the wild type and stl2 show similar responses at 150 mM NaCl. This mutation can be used to show a simple salt tolerance response and also can be used to demonstrate the complexities of mineral and nutrient interactions in the environment. Vogelien, D. L., L. G. Hickok, and T. R. Warne. 1996. Differential effects of Na, Mg2+, K+, Ca2+and osmotic stress on the wild type and NaCl-tolerant mutants, stl2, of Ceratopteris richardii. Plant, Cell and Environment 19:17–23. Warne, T. R., L. G. Hickok, T. B. Kinraide, and D. L. Vogelien. 1996. High salinity tolerance of the stl2 mutation of Ceratopteris richardii is associated with enhanced K+ influx and loss. Plant, Cell and Environment 19:24–32. Mutant: FUDR tolerant; gene symbol, fdr1 This mutation, which was induced by X-rays, confers tolerance to 2'-deoxy-5fluorouridine (FUDR). This substance is a pyrimidine nucleoside analog and is highly toxic to many organisms. As a nucleotide analog, FUDR interferes with normal metabolism and nucleic acid biosynthesis. This mutation confers a very clear tolerance to FUDR and when gametophytes carrying it are grown in medium containing 10 mM FUDR, wild-type gametophytes are killed shortly after germinating while mutant gametophytes grow quite normally. Wu, K., and J. King. 1994. Biochemical and genetic characterization of 5fluro-2'-deoxyuridine-resistant mutants of Arabidopsis thaliana. Planta 194:117–122. Mutant: sleepy sperm; gene symbol, zzz1 Wild-type C-Fern sperm, when released from the antheridium, are encased in a thin-walled vesicle. Several seconds after release, the sperm break free of the vesicle and quickly swim away. In contrast, most zzz1 sperm remain in the vesicles after release and some C-Fern® Web Manual 60 Sport Reports - Descriptions of C-Fern Mutants eventually break free only after several minutes. The free sperm typically move only slightly or swim very slowly to moderately, with some showing faster movement. The more normal types apparently allow zzz1 gametophytes to self fertilize, despite the abnormalities in most zzz1 sperm. This EMS-generated mutant is excellent for observations at both low and high (>50) magnifications. Duckett, J. G., E. J. Klekowski, and L. G. Hickok. 1979. Ultrastructural studies of mutant spermatozoids in ferns. I. The mature nonmotile spermatozoid of mutation 230X in Ceratopteris thalictroides (L.) Brongn. Gamete Research 2:317–343. Mutant: slow-mo sperm; gene symbol, slo1 Like wild type, slo1 sperm are released from the antheridium in thin-walled vesicles and then, shortly after release, break free and swim away. However, most slo1 sperm swim very slowly and can be easily observed, even under high (>50×) magnification. A few moderately fast-swimming types allow for selffertilization of slo1 gametophytes. This mutation is EMS generated. Duckett, J. G., E. J. Klekowski, and L. G. Hickok. 1979. Ultrastructural studies of mutant spermatozoids in ferns. I. The mature nonmotile spermatozoid of mutation 230X in Ceratopteris thalictroides (L.) Brongn. Gamete Research 2:317–343. Mutant: bubbles; gene symbol, bub1 This EMS-induced mutant appears normal until day 10, but then begins to show massive swelling of a few to many cells in the gametophyte. Some cells become so large that they actually burst. Expression is variable but easy to observe. Self-fertile. Some irregularity can also be seen in sporophytes that show variable levels of penetrance and expressivity. Mutant Hunt Mix This mixture of wild type and a variety of mutant stocks (many uncharacterized) is an excellent way to introduce students to the wide variety of EMS-induced visible mutant types that can be observed in C-Fern gametophytes. Mutants such as albino, pale, polka dot, early gametophyte lethals, and a variety of other types are present in a combined frequency of at least 10%. Some are very easily observed, while others require more careful observations. Using isolation and selfing or crossing techniques, C-Fern® Web Manual 61 Sport Reports - Descriptions of C-Fern Mutants students can discover, confirm, and characterize mutant phenotypes and determine dominance/recessiveness. C-Fern® Web Manual 62 Glossary of Selected Terms Glossary of Selected Terms abscisic acid—a plant hormone; it reduces growth and sensitivity to antheridiogen in C-Fern gametophytes. allele—one of the alternative forms of a gene that occurs at the same locus. alternation of generations—life cycle alternating between gametophytes and sporophyte. antheridiogen—in ferns, a chemical substance (pheromone) secreted by gametophytes that causes immature gametophytes to develop as males. antheridium (a)—a male sexual organ that produces sperm (spermatozoids); the sperm-producing sex organ of seedless plants as well as some fungi and algae. archegonial neck—the nipple-shaped external portion of an archegonium below the base of which the egg is located. archegonium (a)—a female sexual organ that contains a single egg; the egg-producing sex organ of seedless plants, such as ferns and mosses. buffer—substance that can stabilize the pH of a solution by neutralizing the effects of acids or bases. cell—the fundamental structural unit of living organisms. cell wall—the exterior portion of the plant cell that surrounds the protoplast. chemoattractant—a chemical that can attract motile cells such as sperm. chlorophyll—the green pigment in plant cells used in photosynthesis. chloroplast—a chlorophyll-containing organelle (i.e., plastid) in plants and algae. circinate vernation—the specialized pattern of leaf development that occurs in ferns, in which the leaf unrolls from the “fiddlehead.” cultivar—a type of plant produced by artificial means through cultivation, not existing naturally. C-Fern® Web Manual 63 Glossary of Selected Terms clone—an exact copy of an individual. cytoplasm—the protoplasm of a cell, not including the nucleus. diploid—having two sets of chromosomes. dominant—in genetics, a character state (or the allele that controls it) that is expressed in the phenotype even if another allele for the locus is present. dormant—inactive, not growing. egg cell—the female gamete. embryo—the developing diploid sporophyte resulting from division of the zygote. etiolation—a growth response to the absence of light in which certain cells become elongated; in higher plants chlorophyll synthesis is also terminated. fern—a seedless and flowerless plant that has two life-cycle stages —a vascular sporophyte with roots, stems, and fronds (leaves), which produces spores by meiosis and an independent, nonvascular, microscopic gametophyte stage that produces male and female gametes. fertilization—the union of nuclei from female and male gametes. foot—the portion of the fern embryo that is embedded in the gametophyte. frond—a term used to denote the leaf in ferns. gamete—a haploid reproductive or sex cell that can fuse with another gamete to form a zygote. gametophyte—the microscopic haploid phase of the fern life cycle that produces gametes. gene—a unit of heredity carried on the chromosome. genotype—the genetic (allelic) makeup of an individual. germination—the beginning of growth or development of a spore or seed. haploid—having one set of chromosomes. hermaphrodite—a gametophyte containing a meristem and both antheridia and archegonia. C-Fern® Web Manual 64 Glossary of Selected Terms hermaphroditic—an organism with both male and female reproductive organs. heterosporous—producing two types of spores as a result of meiotic division. heterozygous—having two different alleles at the same locus. homosporous—producing one type of spore as a result of meiotic division. homozygous—having two identical alleles at the same locus. hybrid—a combination of two different types. imbibition—absorption of fluid by physical means. incompatibility—in plants, a situation in which a particular genetic combination is not viable. leaf—the major photosynthetic organ of vascular plants. locus—the location of a gene on a chromosome. male—a gametophyte lacking a meristem and containing numerous antheridia. meiosis—a process of cell division in sexually reproducing organisms that results in reduction of chromosome number (by 2) in the products. meristem—an area of active cell division that gives rise to other cells and tissues. mineral nutrient—an inorganic substance necessary for normal growth. mutant—a genetic variant. neck canal cells—a row of cells present in the neck of an unopened archegonium. pH—a measurement of the acidity or relative concentration of hydrogen ions in a solution (pH=-log[H+]). A neutral solution is pH 7, an acidic solution less than 7, and an alkaline or basic solution greater than 7. phenotype—the appearance of an organism, which results from the interaction of its genotype and the environment. pheromone—a substance produced by one individual of a species that affects the development of other individuals in a population. phloem—the food-conducting tissue of a vascular plant. C-Fern® Web Manual 65 Glossary of Selected Terms photosynthesis—a biochemical process in green plants by which (typically) carbohydrates are synthesized from carbon dioxide and water using light as an energy source and releasing oxygen as a by-product. pigment—any of a number of organic molecules that absorb and reflect light of particular wavelengths. polyploid—having more than double the basic chromosome number. prothallus (also prothallium)—a structure produced from a developing spore that bears sex organs;sometimes used interchangeably with “gametophyte.” pteridophyte—a term commonly referring to ferns and fern allies such as horsetails and club mosses. qualitative data—data expressed as a description of an inherent or distinguishing characteristic, property, or trait. quantitative data—data expressed as number, measurement, or quantity. recessive—in genetics, a character state (or the allele that conrols it) that is expressed in the phenotype only if another identical allele for the locus is present. A recessive allele can be masked by a dominant allele. rhizoid—a clear, thread-like cell visible in germinating spores and at the base of gametophytes. root—the vascular organ of sporophytes that is typically associated with the substrate. root cap—the very tip portion of a root that is composed of a loose aggregation of friable cells. sample (sampling)—in statistics, a subset which is analyzed to be representative of the whole population. seed plant—a plant that bears seeds. segregation—in genetics, the separation during meiosis of a pair of alleles at a given locus, resulting ing ametes containing only a single allele per locus. sexual reproduction—reproduction based on the reciprocal processes of meiosis and fertilization. shoot—the arial portion of a plant distinct from the root. C-Fern® Web Manual 66 Glossary of Selected Terms species—a group of interbreeding individuals with similar characteristics. sperm (spermatozoid)—in ferns, a motile male gamete. spore—single-celled haploid (1n) product of meiosis that germinates and develops into a gametophyte. spore mother cell—a diploid (2n) cell that undergoes meiosis to produce haploid (1n) spores. sporophyte—the macroscopic diploid vascular stage of the fern life cycle that produces spores. strain—a group of plants distinguished by a particular trait. syngamy—the fusion of two gametes. variation—a deviation from the norm or standard type. wet mount—a preparation of tissues or cells placed in water on a slide and covered with a coverslip to allow viewing under a compound microscope. wild type—the phenotype selected as a standard for comparison with other phenotypes; often the predominant phenotype among individuals of species. xylem—the water-conducting tissue of a vascular plant. zygote—the diploid cell produced by the fusion of gametes. C-Fern® Web Manual 67 Bibliography Bibliography Augé, R. M., L. G. Hickok, and A. Stodola. 1989. Psychrometric pressure volume analysis of osmoregulation in roots, shoots, and whole sporophytes of salinized Ceratopteris. Plant Physiology 91: 322–330. Banks, J. 1997. Sex determination in the fern Ceratopteris. Trends in Plant Science 2: 175–180. Banks, J. 1997. The TRANSFORMER genes of the fern Ceratopteris simultaneously promote meristem and archegonia development and repress antheridia development in the developing gametophyte. Genetics 147: 1885–1897. Banks, J. A. 1994. Sex-determining genes in the homosporous fern Ceratopteris. Development 120: 1949–1958. Banks, J. A., L. Hickok, and M. A. Webb. 1993. The programming of sexual phenotype in the homosporous fern Ceratopteris richardii. International Journal of Plant Science 154: 522–534. Benedict, R. C. 1909. The genus Ceratopteris: a preliminary revision. Bulletin of the Torrey Botanical Club 36: 463–479. Benedict, R. C. 1948. Fern miscellany: Brooklyn. American Fern Journal 38: 148–154. Carroll, E. W., O. J. Schwarz, and L. G. Hickok. 1988. Biochemical studies of paraquat-tolerant mutants of the fern Ceratopteris richardii. Plant Physiology 87: 651–654. Chasan, R. 1992. Ceratopteris: a model plant for the 90s. The Plant Cell 2: 113–115. Chiang, S.-H. T., and Y.-L. Chiang. 1967. Influence of shading and gibberellic acid on growth and sex of the prothalli of Ceratopteris thalictroides. Taiwania 13: 179–193. Chiang, Y.-L. 1970. Macro- and microscopic structure of the root of Ceratopteris pteridoides (Hook.) Hieron. Taiwania 15: 31–49. Chiang, Y.-L. 1976. On the sex of the prothalli of Ceratopteris pteridoides (Hook.) Hieron. Taiwania 21: 134–137. C-Fern® Web Manual 68 Bibliography Chun, P. T., and L. G. Hickok. 1992. Inheritance of two mutations conferring glyphosate tolerance in the fern Ceratopteris richardii. Canadian Journal of Botany 70: 1097–1099. Cooke, T. J., L. G. Hickok, and M. Sugai. 1995. The fern Ceratopteris richardii as a lower plant model system for studying the genetic regulation of plant photomorphogenesis. International Journal of Plant Science 156: 367–373. Cooke, T. J., L. G. Hickok, W. J. V. D. Woude, J. A. Banks, and R. J. Scott. 1993. Photobiological characterization of a spore germination mutant dkg1 with reversed photoregulation in the fern Ceratopteris richardii. Photochemistry and Photobiology 57: 1032– 1041. Cooke, T. J., R. H. Racusen, L. G. Hickok, and T. R. Warne. 1987. The photocontrol of spore germination in the fern Ceratopteris richardii. Plant Cell Physiology 28: 753–759. Copeland, E. B. 1942. Edible ferns. American Fern Journal 32: 121–126. Dettmann, M. E., and H. T. Clifford. 1991. Spore morphology of Anemia, Mohria, and Ceratopteris (Filicales). American Journal of Botany 78: 303–325. DeYoung, B., T. Weber, B. Hass, and J. Banks. 1997. Generating tetraploid sporophytes and their use in analyzing mutations affecting gametophyte development in the fern Ceratopteris. Genetics 147: 809–814. Donselaar, J. V. 1969. On the distribution and ecology of Ceratopteris in Surinam. American Fern Journal 59: 3–8. Duckett, J. G., E. J. Klekowski, and L. G. Hickok. 1979. Ultrastructural studies of mutant spermatozoids in ferns. I. The mature nonmotile spermatozoid of mutation 230X in Ceratopteris thalictroides (L.) Brongn. Gamete Research 2: 317–343. Eberle, J., and J. Banks. 1996. Genetic interactions among sexdetermining genes in the fern Ceratopteris. Genetics 142. Eberle, J., J. Nemacheck, C.-K. Wen, M. Hasebe, and J. A. Banks. 1995. Ceratopteris: a model system for studying sex-determining mechanisms in plants. International Journal of Plant Science 156: 359–366. C-Fern® Web Manual 69 Bibliography Edwards, Erin S., and Stanley Roux. 1998. Influence of gravity and light on the developmental polarity of Ceratopteris richardii fern spores. Planta 205:553–560. Endress, A. G. 1974. Spore germination of Ceratopteris thalictroides (L.) Brongn. Annals of Botany 38: 877–881. Ford, S. O. 1902. The anatomy of Ceratopteris thalictroides (L.) Brongn. 16: 95–121. Gottlieb, J. E. 1963. Control of marginal leaf meristem growth in Ceratopteris. American Journal of Botany (Abstract) 50: 614. Hannen, H. H. 1968. The introduction and establishment of Ceratopteris in Texas. American Fern Journal 58: 122. Hickok, L. G. 1976. Cytological relationships between three diploid species of the fern genus Ceratopteris. Canadian Journal of Botany 55: 1660–1667. Hickok, L. G. 1977. An apomictic mutant for sticky chromosomes in the fern Ceratopteris. Canadian Journal of Botany 55: 2186–2195. Hickok, L. G. 1977. The cytology and derivation of a temperaturesensitive meiotic mutant in the fern Ceratopteris. American Journal of Botany 64: 552–563. Hickok, L. G. 1978. Homoeologous chromosome pairing and restricted segregation in the fern Ceratopteris. American Journal of Botany 65: 516–521. Hickok, L. G. 1978. Homoeologous chromosome pairing: frequency difference in inbred and intraspecific hybrid polyploid ferns. Science 202: 982–984. Hickok, L. G. 1979. Apogamy and somatic restitution in the fern Ceratopteris. American Journal of Botany 66: 1074–1078. Hickok, L. G. 1979. A cytological study of intraspecific variation in Ceratopteris thalictroides. Canadian Journal of Botany 57: 1694– 1700. Hickok, L. G. 1983. Abscisic acid blocks antheridiogen-induced antheridium formation in gametophytes of the fern Ceratopteris. Canadian Journal of Botany 61: 888–892. Hickok, L. G. 1985. Abscisic acid resistant mutants in the fern Ceratopteris: characterization and genetic analysis. Canadian Journal of Botany 63: 1582–1585. C-Fern® Web Manual 70 Bibliography Hickok, L. G. 1987. Applications of in vitro selection systems: whole plant selection using the haploid phase of the fern Ceratopteris. Pages 53–65 in H. M. LeBaron, R. O. Mumma, R. C. Honeycutt, and J. H. Duesing, eds. Biotechnology in Agricultural Chemistry. American Chemical Society. Hickok, L. G., S. Baxter Duncan, and T. Warne. 2002. C-Fern and the living Punnett square. Carolina Tips 65: 1–3. Hickok, L. G., and R. M. Kiriluk. 1984. Effects of auxins on gametophyte development and sexual differentiation in the fern Ceratopteris thalictroides (L.) Brongn. Botanical Gazette 145: 37– 42. Hickok, L. G., and E. J. Klekowski. 1973. Abnormal reductional and nonreductional meiosis in Ceratopteris: alternatives to homozygosity and hybrid sterility in homosporous ferns. American Journal of Botany 60: 1010–1022. Hickok, L. G., and E. J. Klekowski. 1974. Inchoate speciation in Ceratopteris: An analysis of the synthesized hybrid C. richardii × C. pteridoides. Evolution 28: 439–446. Hickok, L. G., and O. J. Schwarz. 1986. Paraquat tolerant mutants in Ceratopteris: genetic characterization and reselection for enhanced tolerance. Plant Science 47: 153–158. Hickok, L. G., and O. J. Schwarz. 1989. Genetic characterization of a mutation that enhances paraquat tolerance in the fern Ceratopteris richardii. Theoretical and Applied Genetics 77: 200– 204. Hickok, L. G., D. L. Vogelien, and T. R. Warne. 1991. Selection of a mutation conferring high NaCl tolerance to gametophytes of Ceratopteris. Theoretical and Applied Genetics 81: 293–300. Hickok, L. G., and T. R. Warne. 1998. C-Fern Manual. Carolina Biological Supply Company. Burlington, NC. Hickok, L. G., T. R. Warne, S. L. Baxter and C. T. Melear. 1998. Sex and the C-Fern: Not just another life cycle. BioScience 48: 1031–1037. Hickok, L. G., T. R. Warne, and R. S. Fribourg. 1995. The biology of the fern Ceratopteris and its use as a model system. International Journal of Plant Science 156: 332–345. C-Fern® Web Manual 71 Bibliography Hickok, L. G., T. R. Warne, and M. K. Slocum. 1987. Ceratopteris richardii: Applications for experimental plant biology. American Journal of Botany 74: 1304–1316. Hill, Jeffery P. 2001. Meristern development at the sporophyll pinna apex in Ceratopteris richardii. International Journal of Plant Science. 162:235–247. Hoffman, J. C., and K. C. Vaughn. 1995. Using the developing spermatogenous cells of Ceratopteris to unlock the mysteries of the plant cytoskeleton. International Journal of Plant Science 156: 346– 358. Hou, Gui-chaun, and Jeffery P. Hill. 2002. Heteroblastic root development in Ceratopteris richardii (Parkeriaceae). International Journal of Plant Science. 163:341–351. Howe, M. D. 1931. Origin of leaf, and adventitious and secondary roots of Ceratopteris thalictroides. Botanical Gazette 62: 326–329. Huang, T. C. 1977. Ceratopteris taiwanensis Huang sp. nov. Botanical Bulletin of the Academica Sinica 18: 78–81. Javalgekar, S. R. 1960. Sporogenesis and prothallial development in Ceratopteris thalictroides. Botanical Gazette 122: 45–50. Klekowski, E. J., Jr. 1969. Reproductive biology of the Pteridophyta. III. A study of the Blechnaceae. Botanical Journal of the Linnean Society 62: 361–377. Klekowski, E. J., Jr. 1970. Reproductive biology of the Pteridophyta. IV. An experimental study of mating systems in Ceratopteris thalictroides (L.) Brongn. Botanical Journal of the Linnean Society 63: 153–169. Klekowski, E. J., Jr. , and L. G. Hickok. 1974. Nonhomologous chromosome pairing in the fern Ceratopteris. American Journal of Botany 61: 422–432. Lloyd, R. M. 1973. Systematics of the genus Ceratopteris (Parkeriaceae). I. Sexual and vegetative reproduction in Hawaiian Ceratopteris thalictroides. American Fern Journal 63: 12–18. Lloyd, R. M. 1974. Systematics of the genus Ceratopteris Brongn. (Parkeriaceae) II. Taxonomy. Brittonia 26: 139–160. Lloyd, R. 1993. Parkeriaceae—the water fern family. Pages 119– 121 in Editorial Committee, ed. Flora of North America North of Mexico. Oxford University Press, New York. C-Fern® Web Manual 72 Bibliography Lloyd, R. M., and T. R. Warne. 1978. The absence of genetic load in a morphologically variable sexual species, Ceratopteris thalictroides (Parkeriaceae). Systematic Botany 3: 20–36. Loyal, D. S., and H. K. Chopra. 1977. In vitro life-cycle, regeneration and apospory in Ceratopteris pteridoides (Hook.) Hieron. Current Science 46: 89–91. Masuyama, S. 1992. Clinal variation of frond morphology and its adaptive implication in the fern Ceratopteris thalictroides in Japan. Plant Species Biology 7: 87–96. Masuyama, S. 1996. Progenesis as an adaptive strategy in the annual fern Ceratopteris thalictroides in Japan. Plant Species Biology 11: 225–232. Masuyama, S., and Y. Watano. 1994. Hybrid sterility between two isozymic types of the fern Ceratopteris thalictroides in Japan. Journal of Plant Research 107: 269–274. McGrath, J. M., L. G. Hickok, and E. Pichersky. 1994. Assessment of gene copy number in the homosporous fern Ceratopteris thalictroides and C. richardii (Parkeriaceae) by restriction fragment length polymorphisms. Plant Systematics and Evolution 189: 203– 210. McGrath, J. M., L. G. Hickok, and E. Pichersky. 1994. Restriction fragments length polymorphisms distinguish among accessions of Ceratopteris thalictroides and C. richardii (Parkeriaceae). Plant Systematics and Evolution 189: 193–202. Meekes, H. 1986. Inhibition and recovery of cell wall formation in root hairs of Ceratopteris thalictroides. Journal of Experimental Botany 37: 1201–1210. Murata, Takashi, Akeo Kadota, and Masamitsu Wada. 1997. Effects of blue light on cell elongation and microtubule orientation in dark-grown gametophytes of Ceratopteris richardii. Plant Cell Physiology. 38:201–209. National Committee on Science Education Standards and Assessment; and National Research Council. 1996. National Science Education Standards. National Academies Press. 272 p. (http://www.nap.edu/html/nses/) Pal, N., and S. Pal. 1962. Studies on morphology and affinity of the Parkeriaceae. I. Morphological observations of Ceratopteris thalictroides. Botanical Gazette 123: 132–143. C-Fern® Web Manual 73 Bibliography Pal, N., and S. Pal. 1963. Studies on morphology and affinity of the Parkeriaceae. II. Sporogenesis, development of the gametophyte, and cytology of Ceratopteris thalictroides. Botanical Gazette 125: 405–412. Pal, N., and S. Pal. 1969. Studies in the morphology and affinity of the Parkeriaceae. III. A discussion of the systematic position of the family and specific delimitation in the genus Ceratopteris. Bulletin of the Botanical Society of Bengal 23: 17–26. Renzaglia, K. S., and T. R. Warne. 1995. Ceratopteris: an ideal model system for teaching plant biology. International Journal of Plant Science 156: 385–392. Renzaglia, K. S., T. R. Warne, and L. G. Hickok. 1995. Plant development and the fern life cycle using Ceratopteris richardii. American Biology Teacher 57: 438–442. Schedlbauer, M. D. 1974. Biological specificity of the antheridogen from Ceratopteris thalictroides (L.) Brongn. Planta 116: 39–43. Schedlbauer, M. D. 1976. Fern gametophyte development: Controls of dimorphism in Ceratopteris thalictroides. American Journal of Botany 63: 1080–1087. Schedlbauer, M. D. 1976. Specificity of the antheridogen from Ceratopteris thalictroides (L.) Brongn. Plant Physiology 57: 666– 669. Schedlbauer, M. D., C. F. Cave, and P. R. Bell. 1973. The incorporation of DL[3-14C]cysteine during spermatogenesis in Ceratopteris thalictroides. 12: 765–779. Schedlbauer, M. D., and E. J. Klekowski. 1972. Antheridogen activity in the fern Ceratopteris thalictroides (L.) Brongn. Botanical Journal of the Linnean Society 65: 399–413. Schilling, E. E. 1987. Lack of variability in Ceratopteris flavonoids. Phytochemical Bulletin 18: 1–4. Scott, R. J., and L. G. Hickok. 1987. Genetic analysis of antheridiogen sensitivity in Ceratopteris richardii. American Journal of Botany 74: 1872–1877. Scott, R. J., and L. G. Hickok. 1991. Inheritance and characterization of a dark-germinating, light-inhibited mutant in the fern Ceratopteris richardii. Canadian Journal of Botany 69: 2616– 2619. C-Fern® Web Manual 74 Bibliography Singh, M. R. 1990. Characterization of a hydroxyproline tolerant mutant in the fern Ceratopteris. Botany. The University of Tennessee, Knoxville. Stein, D. B. 1971. Gibberellin-induced fertility in the fern Ceratopteris thalictroides (L.) Brongn. Plant Physiology 48: 416– 418. Tryon, R. 1990. Pteridaceae in K. Kramer and P. Green, eds. The families and genera of vascular plants. Vol 2. Pteridophytes and Gymnosperms. Springer Verlag, New York. Tryon, R., and A. Tryon. 1982. Ferns and Allied Plants with Special Reference to Tropical America. Springer Verlag, New York. Vaughan, K. C., L. G. Hickok, T. R. Warne, and A. C. Farrow. 1990. Structural analysis and inheritance of a clumped-chloroplast mutant in the fern Ceratopteris. Journal of Heredity 81: 146–151. Vogelien, D. L. 1993. Physiological characterization of NaCltolerant mutants from Ceratopteris richardii. University of Tennessee, Knoxville. Vogelien, D. L., L. G. Hickok, R. M. Augé, A. J. Stodola, and D. Hendrix. 1993. Solute analysis and water relations of gametophyte mutants tolerant to NaCl in the fern Ceratopteris richardii. Plant, Cell and Environment 16: 959–966. +Vogelien, D. L., L. G. Hickok, and T. R. Warne. 1996. Differential effects of Na, Mg2+, K+, Ca2+and osmotic stress on the wild type and NaCltolerant mutants, stl1 and stl2, of Ceratopteris richardii. Plant, Cell and Environment 19: 17–23. Warne, T. R., and L. G. Hickok. 1987. (2-Chloroethyl)phosphonic acid promotes germination of immature spores of Ceratopteris richardii Brongn. Plant Physiology 83: 723–725. Warne, T. R., and L. G. Hickok. 1987. Single gene mutants tolerant to NaCl in the fern Ceratopteris richardii: characterization and genetic analysis. Plant Science 52: 49–55. Warne, T. R., and L. G. Hickok. 1989. Evidence for a gibberellinbiosynthetic origin for the antheridiogen of Ceratopteris. Plant Physiology 89: 535–538. Warne, T. R., and L. G. Hickok. 1991. Control of sexual development in gametophytes of Ceratopteris richardii: antheridiogen and abscisic acid. Botanical Gazette 152: 148–153. C-Fern® Web Manual 75 Bibliography Warne, T. R., L. G. Hickok, T. B. Kinraide, and D. L. Vogelien. 1996. High salinity tolerance conferred by the stl2 mutation of Ceratopteris richardii is associated with high K+ influx and efflux. Plant, Cell and Environment 19: 24–32. Warne, T. R., L. G. Hickok, and R. J. Scott. 1988. Characterization and genetic analysis of antheridiogen-insensitive mutants in the fern Ceratopteris. Botanical Journal of the Linnean Society 96: 371–379. Warne, T. R., and R. M. Lloyd. 1981. Inbreeding and homozygosity in the fern Ceratopteris pteridoides (Hooker) Hieronymus (Parkeriaceae). Botanical Journal of the Linnean Society 83: 1–13. Warne, T. R., and R. M. Lloyd. 1987. Gametophytic density and sex expression in Ceratopteris (Brongn.). Canadian Journal of Botany 65: 362–365. Warne, T. R., D. L. Vogelien, and L. G. Hickok. 1995. Analysis of genetically and physiologically complex traits using Ceratopteris: a case history of salt tolerance. International Journal of Plant Science 156: 374–384. Watano, Y., and S. Masuyama. 1991. Inbreeding in natural populations of the annual polyploid fern Ceratopteris thalictroides (Parkeriaceae). Systematic Botany 16: 705–714. Watano, Y., and S. Masuyama. 1994. Genetic differentiation in populations of the polymorphic fern Ceratopteris thalictroides in Japan. Journal of Plant Research 107: 139–146. Whalen, M. D. 1979 . Allozyme variation and evolution in Solanum section Androceras. Systematic Botany. 4: 203–222. Yabe, Y., and K. Yasui. 1913. On the life-history of Ceratopteris thalictroides Brongn. Botanical Magazine 27: 233–245. C-Fern® Web Manual 76 User Service User Service For more details on commercially available C-Fern educational materials and supplies, please contact one of the C-Fern distributors. Carolina Biological Supply Company 2700 York Road Burlington, NC 27215 Toll-Free: 800-227-1150 Email: customer_service@carolina.com http://www.carolina.com WARD'S Natural Science Establishment, LLC 5100 West Henrietta Road P.O. Box 92912 Rochester, NY 14692-9012 Toll-Free: 800-962-2660 Phone: 585-359-2502 Fax: 585-334-6174 Email: customer_service@wardsci.com http://www.wardsci.com For questions regarding C-Fern culture, techniques and applications for teaching and research to offer comments and suggestions, please contact: http://www.C-Fern.org. The web site is a source for additional information on using C-Fern in teaching and research and also includes research ideas, protocols, an image gallery, and an extensive bibliography. C-Fern® Web Manual 77