Organic Chemistry II Problem Set Answer Key
advertisement

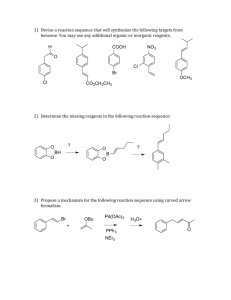
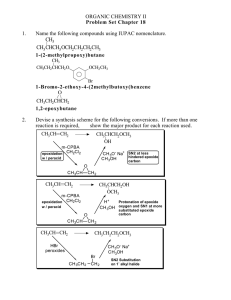
ORG CHEM II Problem Set Chapter 17 ANSWER KEY 1. Name the following compound using IUPAC nomenclature. OH CH3CHCH2CH2CHCH3 5-phenyl-2-hexanol 2. Compound A has the formula, C7H14OH. When it is reacted with POCl3 in pyridine, it forms Compound B, C7H12. Compound B reacts with ozone, followed by zinc in acetic acid, to form Compound C, O O HCCH2CH2CH2CH2CCH3 . Draw the chemical structures for Compounds A & B. CH 3 CH 3 OH O POCl3 2) Zn, H+ A O 1) O 3 B H CH 3 C 3. Over each arrow show the chemical reagents and conditions needed to perform the following reactions. (Only one reaction is implied for each example below.) PCC, CH 2Cl 2 a. CH3CH2CH2CH2OH → CH3CH2CH2CHO SOCl 2 b. CH3CH2CH2CH2OH → CH3CH2CH2CH2Cl CrO 3, H 2SO4 c. CH3CH2CH2CH2OH → CH3CH2CH2CO2H d. CH3CH2OH NaH → CH3CH2O-Na+ 4. Complete the following road map using any needed reagents, containing 4 or less carbons, and any needed conditions. Show the major product for each reaction used. OH CH 3CH 2CH 2CH 2OH OH POCl3 CH 3CH 2CHCH3 Hg(OAc)2 NaBH 4 H2SO 4 H2O CrO3 H2SO 4 OH O 8. What reagent or series of reagents would you need to accomplish the following conversions? A series of reactions should be numbered in sequence. O CH 2CH 3 POCl 3 1) CrO 3, H 2SO 4 2) CH 3CH 2 MgX 3) H3O + CrO 3 H2SO 4 OH 1) POCl 3 2) O3 3) Zn, H+ PBr 3 Br 1) POCl 3 2) OsO 4 3) NaHSO 3 H H OH OH O O 9. Show three different Grignard reactions which would result in the alcohol at right. CO 2CH 3 1) 2eq CH3 MgX 2) H3O + C O H3C O C CH 3 1) CH3 MgX 2) H3O + 1) 2) H3O + CH 3 MgX 10. Show, over the arrows, the reagents and conditions needed for the transformat CH 2Cl CO 2H CO 2H 2eq LiAlH4 CHO SOCl 2 CrO 3 CH 2OH NaBH4 PCC O O MgX 1) H C CHO H3C H S Cl O 2) H3O + NaH CH 2O- Na+ CH 2OTs 11. What reagents are needed for the following transformations? If more than one reaction is necessary, show each set of reagents and conditions over a separate arrow and show the major product for each reaction. PCC a. CH3CH2OH → CH3CHO 1) NaBH 4 2) H 3 O+ b. CH3CHO → CH3CH2OH SOCl 2 c. CH3CH2OH → CH3CH2Cl NaH d. CH3CH2OH → CH3CH2O-Na+ CrO3 , H 2 SO 4 e. CH3CH2OH → CH3CO2H f. CH3CH2OH → CH3CH2CO2H