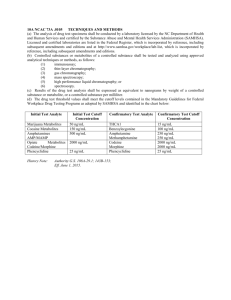
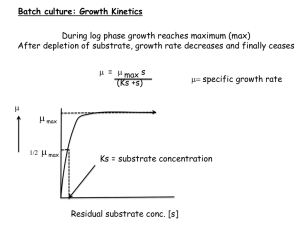
Drug Metabolism Compendium - Guide to Innovation
advertisement

Drug Metabolism GUIDE TO INNOVATION For Research Use Only. Not for use in diagnostic procedures. Drug Metabolism – Analysis with Confidence Pharmaceutical and biopharmaceutical companies have leveraged advancements in basic science perhaps more than any other industry. With the advent of whole genome sequencing, sophisticated analysis of metabolic pathways, and exponential improvements in computer processing, R&D organizations have expanded their drug portfolio focus from small molecules to biotherapeutics. To ensure continued success in drug development, the Industry has been challenged to design and deliver targeted new medicines with higher efficiency at a faster pace than ever before. As a result, better and faster information is required to develop a fundamental understanding of the target compound’s efficiency as a therapeutic and its metabolic profile for potential toxicity. Partnering with customers by understanding their challenges, collaborating on ideas, and ultimately creating cutting-edge solutions that provide new ways to address these new challenges in drug metabolism is our priority. The following compendium includes key solutions for drug metabolism — and, more importantly, describes in detail work done by, and in collaboration with, our customers. Your success is our success, and the AB SCIEX team will partner with you to overcome some of the biggest challenges of drug metabolism analysis, now and into the future. Gary Impey, PhD Director, Pharma/CRO MS Business RUO-MKT-01-1583-A Warrington,UK Redwood Shores & Dublin, CA, U.S.A. Concord, ONT,Canada Paris, France Framingham, MA, U.S.A. Darmstadt, Germany Milan, Italy Tokyo, Japan Seoul, S. Korea New Delhi, India Shanghai, China Singapore Melbourne, Australia AB SCIEX global associates and sites 1,500+ associates worldwide 260+ hold PhDs or other advanced degrees 14 operating sites Global sales and service teams 7 global demo labs For Research Use Only. Not for use in diagnostic procedures. Drug Metabolism – Analysis with Confidence Pharmaceutical and biopharmaceutical companies have leveraged advancements in basic science perhaps more than any other industry. With the advent of whole genome sequencing, sophisticated analysis of metabolic pathways, and exponential improvements in computer processing, R&D organizations have expanded their drug portfolio focus from small molecules to biotherapeutics. To ensure continued success in drug development, the Industry has been challenged to design and deliver targeted new medicines with higher efficiency at a faster pace than ever before. As a result, better and faster information is required to develop a fundamental understanding of the target compound’s efficiency as a therapeutic and its metabolic profile for potential toxicity. Partnering with customers by understanding their challenges, collaborating on ideas, and ultimately creating cutting-edge solutions that provide new ways to address these new challenges in drug metabolism is our priority. The following compendium includes key solutions for drug metabolism — and, more importantly, describes in detail work done by, and in collaboration with, our customers. Your success is our success, and the AB SCIEX team will partner with you to overcome some of the biggest challenges of drug metabolism analysis, now and into the future. Gary Impey, PhD Director, Pharma/CRO MS Business RUO-MKT-01-1583-A Warrington,UK Redwood Shores & Dublin, CA, U.S.A. Concord, ONT,Canada Paris, France Framingham, MA, U.S.A. Darmstadt, Germany Milan, Italy Tokyo, Japan Seoul, S. Korea New Delhi, India Shanghai, China Singapore Melbourne, Australia AB SCIEX global associates and sites 1,500+ associates worldwide 260+ hold PhDs or other advanced degrees 14 operating sites Global sales and service teams 7 global demo labs For Research Use Only. Not for use in diagnostic procedures. Xenobiotic Metabolism Workflows in Drug Discovery and Development Introduction 6-15 Contents Xenobiotic Metabolism Workflows in Drug Discovery and Development RUO-MKT-01-1583-A Technology Spotlight 16-27 Technology Drives High Performance in Biomolecular Mass Spectrometry 16-19 High Resolution Time-of-Flight MS 20-23 PCVG: A Powerful Algorithm for Automating Comprehensive Xenobiotic Metabolite Identification 24-27 Early Discovery 28-43 DiscoveryQuant™ Software 2.0: The Definitive Solution for LC/MS/MS Early-ADME Workflows 28-31 Confirmation of In Vitro Nefazodone Metabolites Using the Superior Fragmentation of the QTRAP® 5500 LC/MS/MS System 32-37 Breakthrough Productivity for ADME Studies Using the AB SCIEX TripleTOF® 5600 System 38-43 44-67 Late Stage Discovery 44-47 Rapid Metabolite Identification Using MetabolitePilot™ Software and the TripleTOF® 5600 System 48-51 In Vivo Metabolic Profiling of Carbamazepine Using the QTRAP® 5500 System and LightSight® Software 2.2 52-57 Comprehensive Detection of Metabolites Using Polarity Switching Data Collection with the QTRAP® 5500 LC/MS/MS System 58-61 Simultaneous Pharmacokinetic Profiling and Automated Metabolite Identification Using the AB SCIEX TripleTOF™ 5600 System and MetabolitePilot™ Software 62-67 Metabolite Identification with the QTRAP® 5500 LC/MS/MS System: Sensitivity, Selectivity, Speed, and Unique Workflows 68-85 Definitive Metabolite ID 68-71 Simultaneous Metabolite Identification and Quantitation with UV Data Integration Using LightSight® Software 2.2 72-77 Differential Mobility Spectrometry for Quantitative and Qualitative Applications in Pharmaceutical Workflows 78-81 Solving Bottlenecks in Metabolite Identification Using TripleTOF® Systems and MetabolitePilot™ Software 82-85 Removing Bottlenecks in Metabolite ID Data Analysis with MetabolitePilot™ Software For Research Use Only. Not for use in diagnostic procedures. Xenobiotic Metabolism Workflows in Drug Discovery and Development Introduction 6-15 Contents Xenobiotic Metabolism Workflows in Drug Discovery and Development RUO-MKT-01-1583-A Technology Spotlight 16-27 Technology Drives High Performance in Biomolecular Mass Spectrometry 16-19 High Resolution Time-of-Flight MS 20-23 PCVG: A Powerful Algorithm for Automating Comprehensive Xenobiotic Metabolite Identification 24-27 Early Discovery 28-43 DiscoveryQuant™ Software 2.0: The Definitive Solution for LC/MS/MS Early-ADME Workflows 28-31 Confirmation of In Vitro Nefazodone Metabolites Using the Superior Fragmentation of the QTRAP® 5500 LC/MS/MS System 32-37 Breakthrough Productivity for ADME Studies Using the AB SCIEX TripleTOF® 5600 System 38-43 44-67 Late Stage Discovery 44-47 Rapid Metabolite Identification Using MetabolitePilot™ Software and the TripleTOF® 5600 System 48-51 In Vivo Metabolic Profiling of Carbamazepine Using the QTRAP® 5500 System and LightSight® Software 2.2 52-57 Comprehensive Detection of Metabolites Using Polarity Switching Data Collection with the QTRAP® 5500 LC/MS/MS System 58-61 Simultaneous Pharmacokinetic Profiling and Automated Metabolite Identification Using the AB SCIEX TripleTOF™ 5600 System and MetabolitePilot™ Software 62-67 Metabolite Identification with the QTRAP® 5500 LC/MS/MS System: Sensitivity, Selectivity, Speed, and Unique Workflows 68-85 Definitive Metabolite ID 68-71 Simultaneous Metabolite Identification and Quantitation with UV Data Integration Using LightSight® Software 2.2 72-77 Differential Mobility Spectrometry for Quantitative and Qualitative Applications in Pharmaceutical Workflows 78-81 Solving Bottlenecks in Metabolite Identification Using TripleTOF® Systems and MetabolitePilot™ Software 82-85 Removing Bottlenecks in Metabolite ID Data Analysis with MetabolitePilot™ Software Xenobiotic Metabolism Workflows in Drug Discovery and Development precursor ions are used to filter unbiased scan data to arrive at relevant peaks related to parent drug material.4-6 (A complete lists of filters types are listed or reviewed in the given references.2,3) Emerging to further expand the tool box for metabolite identification are strategies for real-time filtering of non-targeted data during acquisition that can keep pace with the shorter time scales during ultra-high pressure liquid chromatography (UHPLC) separations and identify drug-related material as it is being acquired.2,3 Laura Baker 1, Suma Ramagiri 2 1 Contract Technical Writer at AB SCIEX, Pittsburgh, PA, 2AB SCIEX, Concord, Canada To stay competitive in the field of modern therapeutics, the pharmaceutical industry must deliver increasingly potent and targeted new medicines at an ever faster pace. The discovery and development process for new drugs is a high-risk venture, where numerous prospective candidates are screened, but only one in five compounds in the development stage will successfully complete clinical research studies to become a marketable drug (Figure 1). Key to this process is assessing a compound’s suitability as a drug, and defining its metabolic profile is essential to ensure that its downstream metabolites are not toxic. Rapid feedback on metabolite composition during early stages of drug development means a more effective screening process, preventing the costly possibility of an unsuitable candidate progressing too far down the development pipeline. Characterization of the body’s metabolic response to a drug has evolved from a late-stage regulatory requirement to an essential and pivotal finding in the discovery stage that must be attained quickly alongside in vitro biological screening results. This shift towards earlier metabolite profiling during the drug discovery process has impacted pharmaceutical discovery workflows, requiring evaluation of drug metabolism properties at every step (Figure 2). In the early discovery stage, metabolic stability and its influence on pharmacological response is explored at a more cursory level relying on in vitro analysis to help steer the selection of lead candidates. A drug must have appropriate bioavailability and efficacy over a desired interval of time to be successful, leading to increased efforts to characterize metabolites early in the discovery stage and to provide critical soft spot analysis for lead optimization. Later stage metabolic profiling may reveal that in vitro metabolites are different from those obtained in vivo; furthermore, in vivo metabolites may vary between animal models.1 Comparison of metabolites from these screens early in the process allows for the selection of appropriate animal models for toxicology screens.2 In the development stage, application for new drug status with the FDA mandates that a high level of metabolic characterization be delivered, including in-depth structural elucidation and metabolite identification.4 Metabolites may clear from the body differently from the parent drug and act by differing enzymatic mechanisms; thus, it is ideal to have detailed pharmacokinetic and absolute quantitative information in hand before clinical studies begin. Appreciating that the safety of patients is compromised if the toxicological effects of a new drug are not well understood, a drug candidate’s metabolic fingerprint can constructively shape the design of a clinical trial.2 The substantial impact that metabolic parameters have on a drug’s continued success through the development pipeline and the effectiveness of providing in-depth metabolic information earlier in this process highlights the increasing need for high-throughput bioanalytical techniques that rapidly deliver comprehensive metabolic profiles. In the past decade, metabolite identification workflows have evolved fundamentally due to breakthrough advances in mass spectrometry technologies.4 This shift has driven the movement from analytical compound-based mass spectrometry workflows, which were often multi-step and multi-injection, towards more generic workflows that focus on extracting metabolite structural information using post-acquisition processing of unbiased, non-targeted mass spectral data (all-in-one approach).4 Traditional Figure 2: Metabolite profiling tasks required for each stage of the drug discovery process. phase I and II metabolite detection often involves two separate injections, including a full scan of the sample and then a second scan to perform MS/MS of pre-selected peaks that correspond to potential parent drug metabolites.2,4 This dual injection approach is very laborious and further consumes valuable sample; however, the primary limiting factor is the processing of the extensive data files generated during non-targeted fragmentation of every component included within the sample.3 MS/MS fragment ion interpretation and assignment of new metabolite structures is very labor-intensive and typically conducted using non-integrated, independent software packages, adding complexity to an already daunting process. Increasing to the challenge, drug studies are frequently carried out in biological fluids or tissues, so low-level metabolites from clinically-relevant doses of the parent drug must be detected against a backdrop of competing ions that can suppress signals of interest.3 For the pharmaceutical scientist, the key to overcoming this data bottleneck at the discovery stage is to rapidly find, identify, and confirm low-level metabolites in complex matrices using highly sensitive and selective automated processes – and obtain this data from just a single injection. This resource on xenobiotic metabolism workflows explores fast, efficient mass spectrometry techniques that meet the challenge of high-throughput metabolite identification and quantitation during the drug discovery and development process. With the advent of a new generation of high resolution mass spectrometers (HRMS), including Fourier transform mass spectrometry and hybrid triple quadrupole/time-of-flight instruments, non-targeted, unbiased detection of all metabolites, even unpredicted, can be carried out in just one run.2,4 What has emerged as the new standard for metabolite studies is the coupling of high resolution, accurate mass instruments with effective computational strategies for filtering the large data sets from all-in-one fragmentation approaches, easing interpretation and increasing throughput.2-5 Newer workflows emphasize post-acquisition analysis of data files, where metabolite predictions based on known biotransformation activities, intensity cut-offs, neutral loss, mass defects, and INTRODUCTION INTRODUCTION For Research Use Only. Not for use in diagnostic procedures. Herein we present an extensive resource composed of drug metabolite identification and quantitation studies that were performed by our own scientists, customers, and collaborators, which reveal how highly selective and sensitive detection of lowlevel metabolites can be carried out on state-of-the-art AB SCIEX mass spectrometers, even in the presence of the high background noise from biological samples. • • • Section 1: We explore high-throughput ADMET (adsorption, distribution, metabolism, excretion and toxicity) screening workflows that are employed in early drug discovery. Efficiency is essential due to the large number of compounds to be analyzed, and we focus on the automation of screening methods using DiscoveryQuant™ Software, as well as sensitive metabolite profiling using a predictive multiple reaction monitoring (pMRM) approach on a rapidly scanning hybrid triple quadrupole/linear ion trap mass spectrometer, the QTRAP® 6500 System. Non-selective data acquisition analyzed using a real-time multiple mass defect filter (RT-MMDF) is employed on a TripleTOF® 5600 System for high resolution, accurate mass detection and bioanalysis of pharmacokinetic samples. Section 2: Late stage discovery workflows are examined, evaluating technologies that integrate quantitative and qualitative processing in one run at high speeds and high sensitivity. Automated, rapid metabolite identification conducted on the TripleTOF 5600 system with MetabolitePilot™ Software enables high-throughput structure analysis and quantitation. Metabolite data captured using non-targeted SWATH™ Acquisition uncovers unpredicted, low-level metabolites. Workflows on the QTRAP 6500 system with LightSight® Software are dedicated finding very low-level phase I metabolites and reactive phase II metabolite conjugates. Section 3: Definitive metabolite identification during DMPK (drug metabolism and pharmacokinetics) workflows is highlighted, examining how mass accuracy and mass-defect-triggered, informationdependent acquisition (IDA) enable assisted structural characterization of even minor metabolites in complex matrices with the TripleTOF 5600 system and MetabolitePilot Software. Metabolite signal and detection is augmented after removing competing interferences using the orthogonal filtering capacity of SelexION™ Differential Mobility Separation (DMS) Technology for improved selectivity in complex, in vivo samples. LightSight Software integrates external data sources (such as UV quantitation) with mass spectrometry results for improved estimation of metabolite concentration. Figure 1: Drug discovery and development pipeline. 6 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 7 Xenobiotic Metabolism Workflows in Drug Discovery and Development precursor ions are used to filter unbiased scan data to arrive at relevant peaks related to parent drug material.4-6 (A complete lists of filters types are listed or reviewed in the given references.2,3) Emerging to further expand the tool box for metabolite identification are strategies for real-time filtering of non-targeted data during acquisition that can keep pace with the shorter time scales during ultra-high pressure liquid chromatography (UHPLC) separations and identify drug-related material as it is being acquired.2,3 Laura Baker 1, Suma Ramagiri 2 1 Contract Technical Writer at AB SCIEX, Pittsburgh, PA, 2AB SCIEX, Concord, Canada To stay competitive in the field of modern therapeutics, the pharmaceutical industry must deliver increasingly potent and targeted new medicines at an ever faster pace. The discovery and development process for new drugs is a high-risk venture, where numerous prospective candidates are screened, but only one in five compounds in the development stage will successfully complete clinical research studies to become a marketable drug (Figure 1). Key to this process is assessing a compound’s suitability as a drug, and defining its metabolic profile is essential to ensure that its downstream metabolites are not toxic. Rapid feedback on metabolite composition during early stages of drug development means a more effective screening process, preventing the costly possibility of an unsuitable candidate progressing too far down the development pipeline. Characterization of the body’s metabolic response to a drug has evolved from a late-stage regulatory requirement to an essential and pivotal finding in the discovery stage that must be attained quickly alongside in vitro biological screening results. This shift towards earlier metabolite profiling during the drug discovery process has impacted pharmaceutical discovery workflows, requiring evaluation of drug metabolism properties at every step (Figure 2). In the early discovery stage, metabolic stability and its influence on pharmacological response is explored at a more cursory level relying on in vitro analysis to help steer the selection of lead candidates. A drug must have appropriate bioavailability and efficacy over a desired interval of time to be successful, leading to increased efforts to characterize metabolites early in the discovery stage and to provide critical soft spot analysis for lead optimization. Later stage metabolic profiling may reveal that in vitro metabolites are different from those obtained in vivo; furthermore, in vivo metabolites may vary between animal models.1 Comparison of metabolites from these screens early in the process allows for the selection of appropriate animal models for toxicology screens.2 In the development stage, application for new drug status with the FDA mandates that a high level of metabolic characterization be delivered, including in-depth structural elucidation and metabolite identification.4 Metabolites may clear from the body differently from the parent drug and act by differing enzymatic mechanisms; thus, it is ideal to have detailed pharmacokinetic and absolute quantitative information in hand before clinical studies begin. Appreciating that the safety of patients is compromised if the toxicological effects of a new drug are not well understood, a drug candidate’s metabolic fingerprint can constructively shape the design of a clinical trial.2 The substantial impact that metabolic parameters have on a drug’s continued success through the development pipeline and the effectiveness of providing in-depth metabolic information earlier in this process highlights the increasing need for high-throughput bioanalytical techniques that rapidly deliver comprehensive metabolic profiles. In the past decade, metabolite identification workflows have evolved fundamentally due to breakthrough advances in mass spectrometry technologies.4 This shift has driven the movement from analytical compound-based mass spectrometry workflows, which were often multi-step and multi-injection, towards more generic workflows that focus on extracting metabolite structural information using post-acquisition processing of unbiased, non-targeted mass spectral data (all-in-one approach).4 Traditional Figure 2: Metabolite profiling tasks required for each stage of the drug discovery process. phase I and II metabolite detection often involves two separate injections, including a full scan of the sample and then a second scan to perform MS/MS of pre-selected peaks that correspond to potential parent drug metabolites.2,4 This dual injection approach is very laborious and further consumes valuable sample; however, the primary limiting factor is the processing of the extensive data files generated during non-targeted fragmentation of every component included within the sample.3 MS/MS fragment ion interpretation and assignment of new metabolite structures is very labor-intensive and typically conducted using non-integrated, independent software packages, adding complexity to an already daunting process. Increasing to the challenge, drug studies are frequently carried out in biological fluids or tissues, so low-level metabolites from clinically-relevant doses of the parent drug must be detected against a backdrop of competing ions that can suppress signals of interest.3 For the pharmaceutical scientist, the key to overcoming this data bottleneck at the discovery stage is to rapidly find, identify, and confirm low-level metabolites in complex matrices using highly sensitive and selective automated processes – and obtain this data from just a single injection. This resource on xenobiotic metabolism workflows explores fast, efficient mass spectrometry techniques that meet the challenge of high-throughput metabolite identification and quantitation during the drug discovery and development process. With the advent of a new generation of high resolution mass spectrometers (HRMS), including Fourier transform mass spectrometry and hybrid triple quadrupole/time-of-flight instruments, non-targeted, unbiased detection of all metabolites, even unpredicted, can be carried out in just one run.2,4 What has emerged as the new standard for metabolite studies is the coupling of high resolution, accurate mass instruments with effective computational strategies for filtering the large data sets from all-in-one fragmentation approaches, easing interpretation and increasing throughput.2-5 Newer workflows emphasize post-acquisition analysis of data files, where metabolite predictions based on known biotransformation activities, intensity cut-offs, neutral loss, mass defects, and INTRODUCTION INTRODUCTION For Research Use Only. Not for use in diagnostic procedures. Herein we present an extensive resource composed of drug metabolite identification and quantitation studies that were performed by our own scientists, customers, and collaborators, which reveal how highly selective and sensitive detection of lowlevel metabolites can be carried out on state-of-the-art AB SCIEX mass spectrometers, even in the presence of the high background noise from biological samples. • • • Section 1: We explore high-throughput ADMET (adsorption, distribution, metabolism, excretion and toxicity) screening workflows that are employed in early drug discovery. Efficiency is essential due to the large number of compounds to be analyzed, and we focus on the automation of screening methods using DiscoveryQuant™ Software, as well as sensitive metabolite profiling using a predictive multiple reaction monitoring (pMRM) approach on a rapidly scanning hybrid triple quadrupole/linear ion trap mass spectrometer, the QTRAP® 6500 System. Non-selective data acquisition analyzed using a real-time multiple mass defect filter (RT-MMDF) is employed on a TripleTOF® 5600 System for high resolution, accurate mass detection and bioanalysis of pharmacokinetic samples. Section 2: Late stage discovery workflows are examined, evaluating technologies that integrate quantitative and qualitative processing in one run at high speeds and high sensitivity. Automated, rapid metabolite identification conducted on the TripleTOF 5600 system with MetabolitePilot™ Software enables high-throughput structure analysis and quantitation. Metabolite data captured using non-targeted SWATH™ Acquisition uncovers unpredicted, low-level metabolites. Workflows on the QTRAP 6500 system with LightSight® Software are dedicated finding very low-level phase I metabolites and reactive phase II metabolite conjugates. Section 3: Definitive metabolite identification during DMPK (drug metabolism and pharmacokinetics) workflows is highlighted, examining how mass accuracy and mass-defect-triggered, informationdependent acquisition (IDA) enable assisted structural characterization of even minor metabolites in complex matrices with the TripleTOF 5600 system and MetabolitePilot Software. Metabolite signal and detection is augmented after removing competing interferences using the orthogonal filtering capacity of SelexION™ Differential Mobility Separation (DMS) Technology for improved selectivity in complex, in vivo samples. LightSight Software integrates external data sources (such as UV quantitation) with mass spectrometry results for improved estimation of metabolite concentration. Figure 1: Drug discovery and development pipeline. 6 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 7 Title “Software automation tools for increased throughput metabolic soft-spot identification in early drug discovery” Article Highlights • • • “Comparison of information-dependent acquisition, SWATH™ Acquisition, and MSAll Techniques in • metabolite identification study employing ultrahigh-performance liquid chromatography— quadrupole time-of-flight mass spectrometry” • • • “Identification of urinary metabolites of imperatorin with a single run on an LC/Triple TOF system based on multiple mass defect filter data acquisition and multiple data mining techniques” • • • • “Identification of metabolites of deoxyschizandrin in rats by UPLC-Q-TOF-MS/MS based on multiple mass defect filter data acquisition and multiple data processing techniques” • • • • “Standardized workflows for increasing efficiency and productivity in discovery stage bioanalysis” • • • Citation Application of software automation tools for rapidly identifying metabolites using mass defect filtering and HRMS instruments Review of MetabolitePilot™ Software and Mass-MetaSite™ Software workflows for finding peaks, identifying metabolites, and elucidating structures Focus was on accuracy and throughput for the localization of the primary soft spots on firstgeneration metabolites of proprietary compounds Zelesky V, Schneider, R, Janiszewski J, Zamora I, Ferguson J, Troutman M. Bioanalysis. 2013; 5(10): 1165-1179. Evaluation of methods for acquiring MS/MS data using an HRMS instrument for metabolite ID in microsomes and urine Comparison of IDA, SWATH™ Acquisition, and MSAll data acquisition methodologies using eight non-proprietary compounds that produced a diverse array of metabolites SWATH Acquisition and MSAll methods surpassed IDA hit rates, triggering MS/MS for all metabolites, while IDA methods produced superior MS/MS data for facilitation of structural assignment. MS/MS spectra quality is highly dependent on the Q1 selection window width (and thus the acquisition method). Zhu Z, Chen Y, Subramanian R. Analytical Chemistry. 2014; 86(2):1202-9. A generic, single-injection approach for detecting in vivo metabolites, including low-level, of an herbal Chinese medicine using HRMS instrumentation and a single injection protocol Data acquisition was conducted using real-time multiple mass defect filters (MMDF) combined with dynamic background subtraction to identify urinary metabolites. Low-level metabolites were identified from high background and excess endogenous components by combining HRMS data mining techniques (XIC, MDF, PIF, and NLF). Structures for 44 phase I and 7 phase II metabolites were reported using this powerful, integrated approach. Qiao S, Shi X, Shi R, Liu M, Liu T, Zhang K, Wang Q, Yao M, Zhang L. Anal Bioanal Chem. 2013; 405: 6721-6738. Development of a novel and efficient strategy for screening the in vivo metabolites of an herbal Chinese medicine using HRMS instrumentation and a single injection protocol Filters such as MMDF and DBS were applied on-line during data acquisition, and multiple post data-acquisition filters (XIC, MDF, PIF, and NLF) were combined to obtain 51 phase I and II metabolites in rat urine and bile. MetabolitePilot Software identified metabolites, and structure elucidation was enabled by accurate mass information, biotransformation knowledge, and fragmentation patterns. A unique, Clog P value was assigned to compounds to distinguish between multiple isomers, which was based on varying retention times for isomers. Liu M, Zhao S, Wang Z, Wang Y, Liu T, Song L, Wang C, Wang H, Tu P. Journal of Chromatography B. 2014; 949-950:115-126. Discussion of standardized discovery LC-MS workflows for high-throughput, small-molecule bioanalysis and the efficiency gains after implementation Bioanalytical processes (compound tuning, LC method development, analytical acceptance criteria, automated sample preparation, sample analysis platforms, data processing, and data reporting) were harmonized across multiple research sites. Reducing time and resources on routine bioanalysis has allowed for more challenging studies and development of future research. Bateman KP, Cohen L, Emary B, Pucci V. Bioanalysis. 2013; 5(14): 1783-1794. “Bioactivation of sitaxentan in liver microsomes, hepatocytes, and expressed human P450s with characterization of the glutathione conjugate by liquid chromatography tandem mass spectrometry” • Identification of a reactive metabolite and its structure for an endothelin-A receptor antagonist withdrawn for idiosyncratic drug toxicity • Characterization of an in vitro GSH-conjugate in liver microsomes from wide array of mammals using hybrid triple quadrupole/time-of-flight mass spectrometry full scan data and product ion spectra • Reactive metabolite inhibition of a specific P450 isoform was demonstrated through competitive and time-dependent assays and proposed as a mechanism for drug toxicity. Erve JCL, Gauby S, Maynard, Jr. JW, Svensson MA, Tonn G, Quinn KP. Chemical Research in Toxicology. 2013; 26: 926-936. “Drug metabolite profiling and identification by high-resolution mass spectrometry” • Overview of HRMS acquisition methods (both targeted and non-targeted) and data mining techniques (mass defect, product ion, isotope pattern filters, and background subtraction) • Review of single HRMS platforms with the capacity for multiple metabolite ID tasks • Future developments for metabolite ID on HRMS instruments Zhu, M, Zhang, H, Humphreys WG. J Biol Chem. 2011; 286 (29): 25419-25425. *These articles were reprinted with permission in the first 30 copies of this resource. Each section and experiment featured in this resource includes an overview of the key challenges, benefits, and features of the bioanalytical technique presented. In this way, the mass spectrometric techniques and metabolic data analysis can be put into context with other bioanalytical tools and help highlight the many advantages that LC/MS/MS offers to all stages of drug discovery and development. For further reference, Table 1 showcases the current literature, reviewing the trends and novel techniques that are behind the high-throughput innovations for detecting and identifying metabolites. Presented below are some of the key challenges and benefits of metabolite identification, alongside the innovative analytical instruments that AB SCIEX features for accelerating workflows, meeting the demands for speed, efficiency, and depth of data that are vital to intelligent and effective drug optimization. Key challenges of metabolite identification, structural assignment, and bioanalysis Analyzing very small amounts of drug-related material obscured by cellular components presents a unique challenge to a process that requires increasing levels of productivity to attain the high throughput necessary for the efficient screening of thousands of compounds (Table 2). The interfering signals from the complex matrices that harbor drug metabolites can prolong data processing times or impede the collection of adequate MS/MS information. Structural assignment is often a painstaking, manual process, relying on a biotransformation scientist’s expertise to unravel the data. Capturing information on all metabolites, particularly low-level, in the early stages, is of particular importance to the selection of viable drug candidates. Missing a potentially toxic compound during the screening process due to incomplete data collection may cause valuable resources to be directed towards an unsuitable candidate that may later fail during clinical trials. (An example of a drug with a boxed warning, pulled from the market due to a previously-undetectable reactive metabolite, is given.7) Testing is often completed at low, therapeutically-relevant concentrations, which hinders accurate concentration determination and requires the use of highly sensitive instrumentation. In many cases, data is obtained using multiple, non-integrated platforms making it difficult to transfer data to different applications but also making it problematic to share data with other groups. Limitations on sample availability strongly impact workflow design, and even a single re-injection to re-evaluate a peak or a missed compound requires expensive and time-consuming re-experimenation. To tackle these collective challenges, advanced LC/MS/MS techniques have been developed that are responsive to industry standards for productivity levels requisite for the successful screening of viable compounds. INTRODUCTION INTRODUCTION For Research Use Only. Not for use in diagnostic procedures. Key benefits of LC/MS/MS workflows for metabolite profiling AB SCIEX is dedicated to overcoming drug discovery bottlenecks and automating the metabolic profiling process by developing high-throughput, intelligent workflows that fuel drug discovery. Improvements to instrument capacity, data processing, and software design have propelled metabolite workflows forward, attaining the efficiency and productivity essential for accommodating large sample batches and the screening of thousands of compounds. (See Table 3 for an overview of the benefits of specific AB SCIEX technologies.) Crucial to quicker sample run times are fast scanning speeds and the mass spectrometer’s capacity to collect data in time frames compatible with UHPLC sample elution. Data collection on orbital trapping instruments is hampered by the slower scanning speeds needed to achieve high resolution, but both the TripleTOF 5600 system and the QTRAP 6500 system have the speed and power for rapid MS and MS/MS analysis. The TripleTOF 5600 system maintains approximately 30K resolution regardless of the analysis speed and provides accurate mass data for the unambiguous assignment of elemental composition, as well as the sensitivity and linearity of a triple quadruple instrument for excellent quantitative performance. Prior to the development of hybrid machines, separate MS instruments were employed for accurate quantitation and qualitative discovery of metabolites. The high sensitivity of the QTRAP 6500 system permits metabolic profiling at physiologically relevant dosing and enables the discovery of Table 1: Selected citations for further reading on metabolism workflows in drug discovery and development High Res XICs counts SWATH™ Acquisition 25 Da CID retention time TOF counts Q1 m/z Figure 3: A representation of SWATH™ Acquisition on the TripleTOF ® 5600 System. 8 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 9 Title “Software automation tools for increased throughput metabolic soft-spot identification in early drug discovery” Article Highlights • • • “Comparison of information-dependent acquisition, SWATH™ Acquisition, and MS All Techniques in • metabolite identification study employing ultrahigh-performance liquid chromatography— quadrupole time-of-flight mass spectrometry” • • • “Identification of urinary metabolites of imperatorin with a single run on an LC/Triple TOF system based on multiple mass defect filter data acquisition and multiple data mining techniques” • • • • “Identification of metabolites of deoxyschizandrin in rats by UPLC-Q-TOF-MS/MS based on multiple mass defect filter data acquisition and multiple data processing techniques” • • • • “Standardized workflows for increasing efficiency and productivity in discovery stage bioanalysis” • • • Citation Application of software automation tools for rapidly identifying metabolites using mass defect filtering and HRMS instruments Review of MetabolitePilot™ Software and Mass-MetaSite™ Software workflows for finding peaks, identifying metabolites, and elucidating structures Focus was on accuracy and throughput for the localization of the primary soft spots on firstgeneration metabolites of proprietary compounds Zelesky V, Schneider, R, Janiszewski J, Zamora I, Ferguson J, Troutman M. Bioanalysis. 2013; 5(10): 1165-1179. Evaluation of methods for acquiring MS/MS data using an HRMS instrument for metabolite ID in microsomes and urine Comparison of IDA, SWATH™ Acquisition, and MSAll data acquisition methodologies using eight non-proprietary compounds that produced a diverse array of metabolites SWATH Acquisition and MSAll methods surpassed IDA hit rates, triggering MS/MS for all metabolites, while IDA methods produced superior MS/MS data for facilitation of structural assignment. MS/MS spectra quality is highly dependent on the Q1 selection window width (and thus the acquisition method). Zhu Z, Chen Y, Subramanian R. Analytical Chemistry. 2014; 86(2):1202-9. A generic, single-injection approach for detecting in vivo metabolites, including low-level, of an herbal Chinese medicine using HRMS instrumentation and a single injection protocol Data acquisition was conducted using real-time multiple mass defect filters (MMDF) combined with dynamic background subtraction to identify urinary metabolites. Low-level metabolites were identified from high background and excess endogenous components by combining HRMS data mining techniques (XIC, MDF, PIF, and NLF). Structures for 44 phase I and 7 phase II metabolites were reported using this powerful, integrated approach. Qiao S, Shi X, Shi R, Liu M, Liu T, Zhang K, Wang Q, Yao M, Zhang L. Anal Bioanal Chem . 2013; 405: 6721-6738. Development of a novel and efficient strategy for screening the in vivo metabolites of an herbal Chinese medicine using HRMS instrumentation and a single injection protocol Filters such as MMDF and DBS were applied on-line during data acquisition, and multiple post data-acquisition filters (XIC, MDF, PIF, and NLF) were combined to obtain 51 phase I and II metabolites in rat urine and bile. MetabolitePilot Software identified metabolites, and structure elucidation was enabled by accurate mass information, biotransformation knowledge, and fragmentation patterns. A unique, Clog P value was assigned to compounds to distinguish between multiple isomers, which was based on varying retention times for isomers. Liu M, Zhao S, Wang Z, Wang Y, Liu T, Song L, Wang C, Wang H, Tu P. Journal of Chromatography B. 2014; 949-950:115-126. Discussion of standardized discovery LC-MS workflows for high-throughput, small-molecule bioanalysis and the efficiency gains after implementation Bioanalytical processes (compound tuning, LC method development, analytical acceptance criteria, automated sample preparation, sample analysis platforms, data processing, and data reporting) were harmonized across multiple research sites. Reducing time and resources on routine bioanalysis has allowed for more challenging studies and development of future research. Bateman KP, Cohen L, Emary B, Pucci V. Bioanalysis. 2013; 5(14): 1783-1794. “Bioactivation of sitaxentan in liver microsomes, hepatocytes, and expressed human P450s with characterization of the glutathione conjugate by liquid chromatography tandem mass spectrometry” • Identification of a reactive metabolite and its structure for an endothelin-A receptor antagonist withdrawn for idiosyncratic drug toxicity • Characterization of an in vitro GSH-conjugate in liver microsomes from wide array of mammals using hybrid triple quadrupole/time-of-flight mass spectrometry full scan data and product ion spectra • Reactive metabolite inhibition of a specific P450 isoform was demonstrated through competitive and time-dependent assays and proposed as a mechanism for drug toxicity. Erve JCL, Gauby S, Maynard, Jr. JW, Svensson MA, Tonn G, Quinn KP. Chemical Research in Toxicology. 2013; 26: 926-936. “Drug metabolite profiling and identification by high-resolution mass spectrometry” • Zhu, M, Zhang, H, Humphreys WG. J Biol Chem . 2011; 286 (29): 25419-25425. • • Overview of HRMS acquisition methods (both targeted and non-targeted) and data mining techniques (mass defect, product ion, isotope pattern filters, and background subtraction) Review of single HRMS platforms with the capacity for multiple metabolite ID tasks Future developments for metabolite ID on HRMS instruments *These articles were reprinted with permission in the first 30 copies of this resource. Each section and experiment featured in this resource includes an overview of the key challenges, benefits, and features of the bioanalytical technique presented. In this way, the mass spectrometric techniques and metabolic data analysis can be put into context with other bioanalytical tools and help highlight the many advantages that LC/MS/MS offers to all stages of drug discovery and development. For further reference, Table 1 showcases the current literature, reviewing the trends and novel techniques that are behind the high-throughput innovations for detecting and identifying metabolites. Presented below are some of the key challenges and benefits of metabolite identification, alongside the innovative analytical instruments that AB SCIEX features for accelerating workflows, meeting the demands for speed, efficiency, and depth of data that are vital to intelligent and effective drug optimization. Key challenges of metabolite identification, structural assignment, and bioanalysis Analyzing very small amounts of drug-related material obscured by cellular components presents a unique challenge to a process that requires increasing levels of productivity to attain the high throughput necessary for the efficient screening of thousands of compounds (Table 2). The interfering signals from the complex matrices that harbor drug metabolites can prolong data processing times or impede the collection of adequate MS/MS information. Structural assignment is often a painstaking, manual process, relying on a biotransformation scientist’s expertise to unravel the data. Capturing information on all metabolites, particularly low-level, in the early stages, is of particular importance to the selection of viable drug candidates. Missing a potentially toxic compound during the screening process due to incomplete data collection may cause valuable resources to be directed towards an unsuitable candidate that may later fail during clinical trials. (An example of a drug with a boxed warning, pulled from the market due to a previously-undetectable reactive metabolite, is given.7) Testing is often completed at low, therapeutically-relevant concentrations, which hinders accurate concentration determination and requires the use of highly sensitive instrumentation. In many cases, data is obtained using multiple, non-integrated platforms making it difficult to transfer data to different applications but also making it problematic to share data with other groups. Limitations on sample availability strongly impact workflow design, and even a single re-injection to re-evaluate a peak or a missed compound requires expensive and time-consuming re-experimenation. To tackle these collective challenges, advanced LC/MS/MS techniques have been developed that are responsive to industry standards for productivity levels requisite for the successful screening of viable compounds. INTRODUCTION INTRODUCTION For Research Use Only. Not for use in diagnostic procedures. Key benefits of LC/MS/MS workflows for metabolite profiling AB SCIEX is dedicated to overcoming drug discovery bottlenecks and automating the metabolic profiling process by developing high-throughput, intelligent workflows that fuel drug discovery. Improvements to instrument capacity, data processing, and software design have propelled metabolite workflows forward, attaining the efficiency and productivity essential for accommodating large sample batches and the screening of thousands of compounds. (See Table 3 for an overview of the benefits of specific AB SCIEX technologies.) Crucial to quicker sample run times are fast scanning speeds and the mass spectrometer’s capacity to collect data in time frames compatible with UHPLC sample elution. Data collection on orbital trapping instruments is hampered by the slower scanning speeds needed to achieve high resolution, but both the TripleTOF 5600 system and the QTRAP 6500 system have the speed and power for rapid MS and MS/MS analysis. The TripleTOF 5600 system maintains approximately 30K resolution regardless of the analysis speed and provides accurate mass data for the unambiguous assignment of elemental composition, as well as the sensitivity and linearity of a triple quadruple instrument for excellent quantitative performance. Prior to the development of hybrid machines, separate MS instruments were employed for accurate quantitation and qualitative discovery of metabolites. The high sensitivity of the QTRAP 6500 system permits metabolic profiling at physiologically relevant dosing and enables the discovery of Table 1: Selected citations for further reading on metabolism workflows in drug discovery and development High Res XICs counts SWATH® Acquisition 25 Da CID retention time TOF counts Q1 m/z Figure 3: A representation of SWATH™ Acquisition on the TripleTOF ® 5600 System. 8 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 9 • Overlooking low-level drug metabolites omitted from MS survey scans conducted on complex biological matrices such as bile, plasma, and tissue extracts • Repeat sample injections due to inadequate collection of metabolite information during initial studies leads to costly re-experimentation and re-analysis. • Non-definitive metabolite identification due to inadequate acquisition of MS/MS information on minor or unpredicted metabolites • Multiple, non-integrated software platforms complicate data processing, slowing metabolite identification and structure elucidation. • Co-elution of isomeric or isobaric metabolites is one the major bottlenecks during definitive metabolite identification process. This may result in long chromatographic run times or even expensive column chemistry separating them before identification. Table 2: Summary of key challenges of metabolite identification hard-to-detect, low-level metabolites from just a single run. The linear ion trap scan speeds on the QTRAP 6500 system at rates of 20,000 Da/sec enhance information-dependent acquisition (IDA) coverage as well, allowing for more MS/MS scans on drugrelated peaks. Positive/negative polarization switching on both machines eliminates the need for separate runs for detecting phase II metabolites that, unlike phase I metabolites are often only detected in negative ion mode, allowing for the consolidation of run times and further gains in workflow efficiency. Improvements to DiscoveryQuant™ Software have automated many aspects of method development and sample processing. The integrated, easy-to-use software provides automated methodbuilding templates, auto sampler support, and batch processing for fast optimization of sample acquisition without requiring extensive operator input for every compound. Multiple aspects of data collection are consolidated into one software package, so that method building, data acquisition, and processing can occur on one platform, streamlining data review and saving time. Data processing platforms (MetabolitePilot and LightSight Software) have accelerated metabolic profiling with a more automated approach to interpreting fragment spectra, speeding up metabolite identification, structural elucidation, and metabolic site assignment.6 Correlation of metabolites across multiple samples is possible with batch modes, providing a way to pinpoint lot-to-lot anomalies or conduct time course studies. Sharing this data amongst multiple groups and providing rapid feedback to medicinal chemists is assisted through software linkages to laboratory information management software (LIMS) and global databases, easing data transfer between neighboring laboratories or far-flung research centers.10 Additional software improvements have enhanced the relevancy of peaks identified as drug-related material with the development of novel algorithms that can effectively mine large data sets in a meaningful way and ease fragment interpretation. Data sets obtained through all-in-one approaches, such as SWATH Acquisition (Figure 3), form a comprehensive safety net, capturing both predicted and unpredicted metabolites, and allow for retroactive re-evaluation of the data in the event of an overlooked metabolite. But the enormity of the collection of all MS/MS data for a particular sample raises concerns on 10 RUO-MKT-01-1583-A Real-time multiple mass defect (RT-MMDF) algorithm on the TripleTOF® 5600 System • Increased productivity by simultaneously capturing both qualitative and quantitative data via: – Single injection workflows that capture both TOF MS and TOF MS/MS – Increased MS/MS triggering efficiency on a UPLC time scale (2-3 sec. peak width) • More accurate identification and confirmation of analytes in complex, in vivo samples (e.g., plasma with PEGs, bile samples, tissue samples) SWATH™ Acquisition for data-independent, all-in-one MS/MS fragmentation • Comprehensive quantitative and qualitative analysis in a single injection captures MS/MS information for both predicted and unpredicted metabolites, creating the ultimate safety net • Informative, more complete MS/MS spectra for better metabolite structure prediction and site-modification identification including: – All-inclusive MS/MS for low-level metabolite/catabolite ID – Retention of isotope pattern in MS/MS for each fragment (14C/SIL ADC metabolism studies) – Spectra are less complex than traditional DIA techniques and display more drug-related peaks. AB SCIEX TripleTOF® 5600 System Accurate mass at the speed and sensitivity of a triple quadrupole: • Fast MS and MS/MS acquisition speeds compatible with fast chromatography • Resolution over 30,000 • External mass accuracy ~1 ppm • 4 orders of linear dynamic range AB SCIEX QTRAP® 5500 System A single platform for drug metabolism and bioanalytical quantification workflows • Targeted and non-targeted workflows • Increased sensitivity • Increased speed • Full quantitative capabilities MetabolitePilot™ Software Accurate mass data processing and interrogation: • Generate cleaner, more relevant data with multiple mass defect filtering (MMDF) • Store and retrieve critical information in the compound library and results database • Quickly process multiple sample sets in batches • Increase confidence in your results with intelligent scoring and easy-to-visualize color-coding • Predict formulae with a high level of chemical intelligence LightSight® Software Exploit the full functionality of QTRAP® Technology and processing strategies for multiple metabolite ID workflows • pMRM – high-sensitivity, targeted approach for really low-level detection and confirmation • PI/NL (+/-ve polarity switching) – structure-based filtering approach ideal for reactive metabolite screening • Multiple ion monitoring and Q3 single MS strategies for a non-targeted approach INTRODUCTION INTRODUCTION For Research Use Only. Not for use in diagnostic procedures. • High-resolution quantification reduces the potential for interferences, yet maintains the sensitivity and dynamic range of leading triple quads. – Selective, MRM-style quantitation using product ion mass and summation of product ions • Easy method development and retrospective data mining – Requirement for sample-specific method development is eliminated. – Creation of a digital archive of all analytes enables post-acquisition investigation without additional injections. SelexION™ Technology for metabolite identification workflows • Capable of separating isobaric metabolites or isobaric co-administered drugs – Reduced chromatographic run times accelerate productivity – Elimination of tedious LC method development and expensive columns reduce cost of analysis – Selective and specific quantification provide accurate results for PK profiling and clearance rates. – Enhanced structure elucidation capabilities achieved with more relevant, less complex MS/MS spectra • Elimination of background noise and co-eluting interference – Increased sensitivity lowers the LLOQ and boosts the S/N ratio – Better peak integration facilitates improvement to data quality benchmarks (%CV, accuracy, dynamic range, LLOQ) Table 3: Key benefits of pivotal AB SCIEX metabolite screening workflows how to isolate drug-related material that is overshadowed by background noise. SWATH Acquisition employs an algorithm called principal components variable grouping (PCVG) analysis to filter unbiased data sets, highlighting peaks of interest while removing obfuscating background or chemical noise. The resulting spectra, overall, are less complex after PCVG-filtering and display more relevant drug-related peaks, which represent an inclusive record of all metabolites and their changes over time.2,3 In that vein, the algorithm for real-time multiple mass defect filtering (RT-MMDF) performs a similar feat, but is employed during data-dependent acquisition. (Examples of RT-MMDF applied to metabolite discovery are provided.8,9) During UHPLC/ MS acquisition, the RT-MMDF filter identifies peaks related to the parent drug, selectively triggering MS/MS scans to produce a trimmer data set that accentuates metabolite information without further post-acquisition processing (Figure 4). Both of these innovative approaches – PCVG-filtering and RT-MMDF – continue to effectively redefine and re-invigorate the solutions needed for the complex challenges of therapeutic drug development. DRUG METABOLISM Table 4: Simple, clear metabolism workflows for every stage of drug discovery. The TripleTOF™ 5600 System and the MetabolitePilot™ Software deliver high sensitivity quantification and new qualitative capabilities on a single platform. The QTRAP® 5500 and LightSight® Software use predictive MRM and multiple precursor ion and/or neutral loss survey scans in a single analysis, including polarity switching. Key features of AB SCIEX instrumentation for metabolite profiling As a global front-runner in the development of innovative technology, AB SCIEX continues to provide technological solutions that are transforming the drug pipeline. Each step of drug development has a unique set of benchmarks that must be met prior to advancement (Figure 2). Early in the discovery phase, rapid structural identification and in vitro metabolic stability results are used to screen thousands of compounds and drive the decision-making process surrounding compound optimization. Development phase requirements for integrated qual/quant workflows, quantitation of low-level reactive metabolites, and high-level structure elucidation must meet the compliance and validation standards of regulatory agencies and clinical safety. Mindful of the need for high productivity, AB SCIEX has advanced high-throughput workflows and remarkable, high-performance instruments that streamline the journey from the lab to the clinic for the development of modern, new therapeutics. Discussed below are the workflows and instruments that have set the industry’s standards for competitive operation (summarized in Table 4). 1) High-resolution mass spectrometry quant/qual workflows: TripleTOF 5600 system and MetabolitePilot Software The superb quantitative capacity of a triple quadrupole and the high-performance accurate mass analyzer of a high-resolution www.absciex.com www.absciex.com time-of-flight mass spectrometer are united into one innovative, hybrid instrument, the TripleTOF 5600 system – that performs both quantitative and qualitative analyses with just one method. Throughout every stage of the drug discovery process, it is essential to find, identify, and confirm metabolites as quickly as possible, and the workflows designed for the TripleTOF system enable the fast, accurate metabolic profiling necessary for both ADMET and DMPK studies. The collection of information-rich data using a specialized quant/qual workflow (Figure 5) and the selective extraction of drug-related information during data acquisition produce highly-relevant structural information for detection and characterization of metabolites in the same run. The TripleTOF system has the capacity to collect both MS and MS/MS scan data simultaneously; whilst survey scans generate TOF-MS spectra, a real-time filter based on mass defects (RT-MMDF) triggers MS/MS acquisition of peaks similar to the parent drug, rather than on unrelated background noise, so that only highly relevant information is exhibited in the resulting spectra. Another feature of the hybrid instrumentation is the combination of high resolution and high detector speed that can maintain sufficiently high resolution at low m/z, thereby including even more fragments in the equation for unambiguous structural assignment. (For a full listing of TripleTOF system features, see Table 5.) Altogether, intelligent data acquisition, simultaneous quant/qual analyses, and high scan speeds packaged in one hybrid instrument can support these powerful metabolite workflows. DRUG METABOLISM 11 • Overlooking low-level drug metabolites omitted from MS survey scans conducted on complex biological matrices such as bile, plasma, and tissue extracts • Repeat sample injections due to inadequate collection of metabolite information during initial studies leads to costly re-experimentation and re-analysis. • Non-definitive metabolite identification due to inadequate acquisition of MS/MS information on minor or unpredicted metabolites • Multiple, non-integrated software platforms complicate data processing, slowing metabolite identification and structure elucidation. • Co-elution of isomeric or isobaric metabolites is one the major bottlenecks during definitive metabolite identification process. This may result in long chromatographic run times or even expensive column chemistry separating them before identification. Table 2: Summary of key challenges of metabolite identification hard-to-detect, low-level metabolites from just a single run. The linear ion trap scan speeds on the QTRAP 6500 system at rates of 20,000 Da/sec enhance information-dependent acquisition (IDA) coverage as well, allowing for more MS/MS scans on drugrelated peaks. Positive/negative polarization switching on both machines eliminates the need for separate runs for detecting phase II metabolites that, unlike phase I metabolites are often only detected in negative ion mode, allowing for the consolidation of run times and further gains in workflow efficiency. Improvements to DiscoveryQuant™ Software have automated many aspects of method development and sample processing. The integrated, easy-to-use software provides automated methodbuilding templates, auto sampler support, and batch processing for fast optimization of sample acquisition without requiring extensive operator input for every compound. Multiple aspects of data collection are consolidated into one software package, so that method building, data acquisition, and processing can occur on one platform, streamlining data review and saving time. Data processing platforms (MetabolitePilot and LightSight Software) have accelerated metabolic profiling with a more automated approach to interpreting fragment spectra, speeding up metabolite identification, structural elucidation, and metabolic site assignment.6 Correlation of metabolites across multiple samples is possible with batch modes, providing a way to pinpoint lot-to-lot anomalies or conduct time course studies. Sharing this data amongst multiple groups and providing rapid feedback to medicinal chemists is assisted through software linkages to laboratory information management software (LIMS) and global databases, easing data transfer between neighboring laboratories or far-flung research centers.10 Additional software improvements have enhanced the relevancy of peaks identified as drug-related material with the development of novel algorithms that can effectively mine large data sets in a meaningful way and ease fragment interpretation. Data sets obtained through all-in-one approaches, such as SWATH Acquisition (Figure 3), form a comprehensive safety net, capturing both predicted and unpredicted metabolites, and allow for retroactive re-evaluation of the data in the event of an overlooked metabolite. But the enormity of the collection of all MS/MS data for a particular sample raises concerns on 10 RUO-MKT-01-1583-A Real-time multiple mass defect (RT-MMDF) algorithm on the TripleTOF® 5600 System • Increased productivity by simultaneously capturing both qualitative and quantitative data via: – Single injection workflows that capture both TOF MS and TOF MS/MS – Increased MS/MS triggering efficiency on a UPLC time scale (2-3 sec. peak width) • More accurate identification and confirmation of analytes in complex, in vivo samples (e.g., plasma with PEGs, bile samples, tissue samples) SWATH™ Acquisition for data-independent, all-in-one MS/MS fragmentation • Comprehensive quantitative and qualitative analysis in a single injection captures MS/MS information for both predicted and unpredicted metabolites, creating the ultimate safety net • Informative, more complete MS/MS spectra for better metabolite structure prediction and site-modification identification including: – All-inclusive MS/MS for low-level metabolite/catabolite ID – Retention of isotope pattern in MS/MS for each fragment (14C/SIL ADC metabolism studies) – Spectra are less complex than traditional DIA techniques and display more drug-related peaks. AB SCIEX TripleTOF® 5600 System Accurate mass at the speed and sensitivity of a triple quadrupole: • Fast MS and MS/MS acquisition speeds compatible with fast chromatography • Resolution over 30,000 • External mass accuracy ~1 ppm • 4 orders of linear dynamic range AB SCIEX QTRAP® 5500 System A single platform for drug metabolism and bioanalytical quantification workflows • Targeted and non-targeted workflows • Increased sensitivity • Increased speed • Full quantitative capabilities MetabolitePilot™ Software Accurate mass data processing and interrogation: • Generate cleaner, more relevant data with multiple mass defect filtering (MMDF) • Store and retrieve critical information in the compound library and results database • Quickly process multiple sample sets in batches • Increase confidence in your results with intelligent scoring and easy-to-visualize color-coding • Predict formulae with a high level of chemical intelligence LightSight® Software Exploit the full functionality of QTRAP® Technology and processing strategies for multiple metabolite ID workflows • pMRM – high-sensitivity, targeted approach for really low-level detection and confirmation • PI/NL (+/-ve polarity switching) – structure-based filtering approach ideal for reactive metabolite screening • Multiple ion monitoring and Q3 single MS strategies for a non-targeted approach INTRODUCTION INTRODUCTION For Research Use Only. Not for use in diagnostic procedures. • High-resolution quantification reduces the potential for interferences, yet maintains the sensitivity and dynamic range of leading triple quads. – Selective, MRM-style quantitation using product ion mass and summation of product ions • Easy method development and retrospective data mining – Requirement for sample-specific method development is eliminated. – Creation of a digital archive of all analytes enables post-acquisition investigation without additional injections. SelexION™ Technology for metabolite identification workflows • Capable of separating isobaric metabolites or isobaric co-administered drugs – Reduced chromatographic run times accelerate productivity – Elimination of tedious LC method development and expensive columns reduce cost of analysis – Selective and specific quantification provide accurate results for PK profiling and clearance rates. – Enhanced structure elucidation capabilities achieved with more relevant, less complex MS/MS spectra • Elimination of background noise and co-eluting interference – Increased sensitivity lowers the LLOQ and boosts the S/N ratio – Better peak integration facilitates improvement to data quality benchmarks (%CV, accuracy, dynamic range, LLOQ) Table 3: Key benefits of pivotal AB SCIEX metabolite screening workflows how to isolate drug-related material that is overshadowed by background noise. SWATH Acquisition employs an algorithm called principal components variable grouping (PCVG) analysis to filter unbiased data sets, highlighting peaks of interest while removing obfuscating background or chemical noise. The resulting spectra, overall, are less complex after PCVG-filtering and display more relevant drug-related peaks, which represent an inclusive record of all metabolites and their changes over time.2,3 In that vein, the algorithm for real-time multiple mass defect filtering (RT-MMDF) performs a similar feat, but is employed during data-dependent acquisition. (Examples of RT-MMDF applied to metabolite discovery are provided.8,9) During UHPLC/ MS acquisition, the RT-MMDF filter identifies peaks related to the parent drug, selectively triggering MS/MS scans to produce a trimmer data set that accentuates metabolite information without further post-acquisition processing (Figure 4). Both of these innovative approaches – PCVG-filtering and RT-MMDF – continue to effectively redefine and re-invigorate the solutions needed for the complex challenges of therapeutic drug development. DRUG METABOLISM Table 4: Simple, clear metabolism workflows for every stage of drug discovery. The TripleTOF™ 5600 System and the MetabolitePilot™ Software deliver high sensitivity quantification and new qualitative capabilities on a single platform. The QTRAP® 5500 and LightSight® Software use predictive MRM and multiple precursor ion and/or neutral loss survey scans in a single analysis, including polarity switching. Key features of AB SCIEX instrumentation for metabolite profiling As a global front-runner in the development of innovative technology, AB SCIEX continues to provide technological solutions that are transforming the drug pipeline. Each step of drug development has a unique set of benchmarks that must be met prior to advancement (Figure 2). Early in the discovery phase, rapid structural identification and in vitro metabolic stability results are used to screen thousands of compounds and drive the decision-making process surrounding compound optimization. Development phase requirements for integrated qual/quant workflows, quantitation of low-level reactive metabolites, and high-level structure elucidation must meet the compliance and validation standards of regulatory agencies and clinical safety. Mindful of the need for high productivity, AB SCIEX has advanced high-throughput workflows and remarkable, high-performance instruments that streamline the journey from the lab to the clinic for the development of modern, new therapeutics. Discussed below are the workflows and instruments that have set the industry’s standards for competitive operation (summarized in Table 4). 1) High-resolution mass spectrometry quant/qual workflows: TripleTOF 5600 system and MetabolitePilot Software The superb quantitative capacity of a triple quadrupole and the high-performance accurate mass analyzer of a high-resolution www.absciex.com www.absciex.com time-of-flight mass spectrometer are united into one innovative, hybrid instrument, the TripleTOF 5600 system – that performs both quantitative and qualitative analyses with just one method. Throughout every stage of the drug discovery process, it is essential to find, identify, and confirm metabolites as quickly as possible, and the workflows designed for the TripleTOF system enable the fast, accurate metabolic profiling necessary for both ADMET and DMPK studies. The collection of information-rich data using a specialized quant/qual workflow (Figure 5) and the selective extraction of drug-related information during data acquisition produce highly-relevant structural information for detection and characterization of metabolites in the same run. The TripleTOF system has the capacity to collect both MS and MS/MS scan data simultaneously; whilst survey scans generate TOF-MS spectra, a real-time filter based on mass defects (RT-MMDF) triggers MS/MS acquisition of peaks similar to the parent drug, rather than on unrelated background noise, so that only highly relevant information is exhibited in the resulting spectra. Another feature of the hybrid instrumentation is the combination of high resolution and high detector speed that can maintain sufficiently high resolution at low m/z, thereby including even more fragments in the equation for unambiguous structural assignment. (For a full listing of TripleTOF system features, see Table 5.) Altogether, intelligent data acquisition, simultaneous quant/qual analyses, and high scan speeds packaged in one hybrid instrument can support these powerful metabolite workflows. DRUG METABOLISM 11 2) All-in-one, data-acquisition workflows for comprehensive metabolite profiling: SWATH Acquisition and the TripleTOF system The concept of a real-time algorithm for multiple mass defect filtering • • • • • • Regarded as separate from data-processing algorithms Eliminates MS/MS-triggering on background noise Determines which ion(s) are significantly changing with time Selects the best ion(s) to target for MS/MS acquisition Applied during UPLC/MS acquisition Exists as part of information-dependent data acquisition (IDA) logic Table 5: Key features of TripleTOF® System workflows for metabolite profiling Coupled with MetabolitePilot Software, the TripleTOF system can alleviate the data analysis bottlenecks traditionally associated with metabolite discovery by supplying one integrated platform for all metabolite profiling tasks, including metabolite identification, confirmation of metabolic site, time-course tracking, and inter-species metabolite comparisons. Offering one software package simplifies and automates the interpretation of complex, unwieldy data sets for the fast deconstruction of metabolite information that is crucial to compound optimization. The multifaceted quant/qual workflow has the flexibility and versatility to deliver metabolite information needed for each stage of drug development: • • • Early drug discovery: High-throughput quant/qual assays can quickly analyze microsomal clearance data, matching fragments to proposed structures to determine critical soft spot information, as well as quantitating parent drug concentrations to establish metabolic stability — all using a generic acquisition methodology in a single run. Late stage discovery: The wide-ranging approaches for data filtration (e.g., neutral loss, product ions, mass defect, isotope patterns) provide multiple methods for the discovery of expected and unexpected metabolites, even low-level, and phase II reactive metabolites. Accurate mass, automated structure-driven processing, and weighted scoring of structures speeds confidence in determining the site of metabolism. Automated correlation of peak areas with analog data delivers easier relative quantitation of metabolite and parent peaks for time course studies. Development stage: Processing of accurate mass and high resolution data, low range mass accuracy, and isotope patterns accelerate fragment assignment for definitive structural elucidation and correlation. Integrated processing of pharmacokinetic batch data using MultiQuant Software reveals metabolite concentrations and kinetic profiles relative to the parent, relying on the fast scanning speeds and the high sensitivity of the TripleTOF system to maintain resolution and to detect minor, low-level metabolites. SWATH Acquisition takes its name from the narrow precursor mass range (or swath) of ions selected in Q1 for advancement to Q2 for collision-induced fragmentation (Figure 3). Relying on the fast scan speed of the TripleTOF 5600 system to sequentially process a broad aggregate of narrow mass ranges in a small amount of time, SWATH Acquisition generates a comprehensive map of MS/MS spectra for every ion at every time point. Effectively simplified using PCVG-filtering and other post-acquisition data mining tools, these complex SWATH Acquisition data sets are transformed into spectra comprised of peaks relevant to the parent drug. In this way, one of the primary challenges of metabolite profiling is overcome – the all-inclusive detection of metabolites, including unpredicted and trace-level, is accomplished just one injection without the need for specialized, sample-specific methods. Using data-independent methods, such as SWATH Acquisition, provides accessible results for more straightforward metabolite structure elucidation and identification. A layer of selectivity is established when using successive narrow mass windows to record MS/MS of the chosen ions within, converting complex MSe spectra to simplified datasets displaying relevant, drug-related peaks. The applicability of SWATH Acquisition data to high-resolution quantitation is extended when data is collected using a TripleTOF 5600 system. Using MRM-style methods to gather single product ion peak areas or to sum multiple product ions, metabolite quantitation can be accomplished with the same sensitivity and the dynamic ranges as those achieved on leading triple quadrupole mass spectrometers. High-resolution acquisition lends additional weight to the data quality by improving the completeness of MS/MS data sets for structural elucidation and permitting narrower mass windows to be used for peak selection, removing potentially confounding interferences from the quantitation process. The impact that the SWATH Acquisition workflow has on throughput and productivity during drug discovery and development is unparalleled. Access to an all-encompassing digital archive of complete MS/MS quantitative and qualitative information for every peak at every time point in a sample is valuable at every stage for increasing productivity, throughput, and data quality. (For a review of the additional features of SWATH Acquisition, see Table 6.) Unique qualitative features 1. 2. 3. RUO-MKT-01-1583-A Another hybrid instrument, the QTRAP 6500 system, a unique combination of triple quadrupole and linear ion trap, delivers the enhanced speed and sensitivity needed for rapidly detecting the most number of metabolites using the minimum number of injections – an efficient approach that complements accurate mass HRMS (see Table 4). During in vivo testing, drugs and metabolites need to be detected at physiologically relevant concentrations, which require an exceedingly sensitive approach. Additionally, early discovery of toxic, reactive metabolites is critical for the selection of appropriate candidates during drug development. Building on the significant gains in sensitivity and fast scanning speeds resulting from modern ion trap innovations, pMRM methods find and confirm the presence of ultra-low level in vivo and in vitro metabolites. These highly sensitive, targeted experiments are based on the parent drug fragmentation pattern and possible biotransformations to yield significantly more metabolites, even from complex biological matrices. Reactive metabolite screening experiments are also enabled by the high scan speeds of the QTRAP system. Dual scan surveys (precursor ion and neutral loss) can be conducted in one fast IDA cycle; and when combined with positive/negative polarity switching, these are the first workflows that can detect metabolites of varying polarities in the same run with sufficient sensitivity and accuracy for the highest level of confidence. In just a single injection, these highly sensitive workflows, pMRM and the dual scan IDA methods, provide the reassurance that an all-encompassing list of even the most minor of metabolites has been generated. Unique quant/qual characteristics 1. SelexION™ Technology drives quantitative workflows, but has the added benefit of boosting qualitative techniques. 2. Other mobility techniques are qual focused, not quant. a. SelexION technology’s pre-ion-source location maintains precursor selection and fragmentation. b. Ion mobility spectrometry (IMS) occurs in the collision cell and provides no selectivity before Q1. c. FAIMS, another ion-mobility-based spectrometry, displays reduced robustness and reliability. 3. Chemical modifiers gives the flexibility to try various options for better separation. 4. SelexION technology is compatible with QTRAP and TripleTOF systems. a. MRM-style application for quantitation (continuous flux of ions) b. Full-scan QTOF (pulsed technique) 5. Easy switch between DMS on and off modes a. Allows ions to be transmitted in transparent mode with voltages turned off b. Easily installed in a few minutes without breaking vacuum or using any tools Table 7: Key features of SelexION™ Technology for metabolite identification. • • Early stage discovery: In combination with pMRM methods, polarity switching and fast scanning can quickly and sensitively detect low-level, transient metabolites, both predicted and unpredicted, in biological samples. The detection of minor, but potentially highly toxic, reactive metabolites by identifying glutathione-conjugates is very important for early safety assessments. Building these compound specific acquisition methods, acquiring the data, and then processing it can be consolidated on just one platform – LightSight Software. Late stage discovery: Highly sensitive pMRM scans used during first-pass, in vitro and in vivo metabolic screening can quickly and efficiently profile metabolites at clinically relevant concentrations, while simultaneously generating quantitative data for metabolic stability and pharmacokinetic studies. The high sensitivity of the linear ion trap detects more trace metabolites and their fragments, which in turn provide more information for structural assignments. Unique quantitative features Less complex MS/MS spectrum than traditional DIA techniques like MSe Retention of isotope pattern for each fragment due to wider Q1 selection • Supports 14C/SIL ADC metabolism studies Capture of 100% MS/MS for comprehensive identification of minor metabolites/catabolites 1. Selective MS/MS quantification realized using MRM-style methods for measurement of single product ions or for summing multiple product ions. 2. Multicomponent quantification is feasible with this single-acquisition method. • Quantification of total mAb, conjugated and free small molecule Table 6: Key features of SWATH™ Acquisition on the TripleTOF® System for metabolite ID. 12 3) Cutting-edge workflows for detecting low-level metabolites: QTRAP 6500 system and LightSight Software INTRODUCTION INTRODUCTION For Research Use Only. Not for use in diagnostic procedures. DRUG METABOLISM Figure 4: An illustration of multiple mass defect ranges that were calculated in MetabolitePilot™ Software. www.absciex.com www.absciex.com DRUG METABOLISM 13 2) All-in-one, data-acquisition workflows for comprehensive metabolite profiling: SWATH Acquisition and the TripleTOF system The concept of a real-time algorithm for multiple mass defect filtering • • • • • • Regarded as separate from data-processing algorithms Eliminates MS/MS-triggering on background noise Determines which ion(s) are significantly changing with time Selects the best ion(s) to target for MS/MS acquisition Applied during UPLC/MS acquisition Exists as part of information-dependent data acquisition (IDA) logic Table 5: Key features of TripleTOF® System workflows for metabolite profiling Coupled with MetabolitePilot Software, the TripleTOF system can alleviate the data analysis bottlenecks traditionally associated with metabolite discovery by supplying one integrated platform for all metabolite profiling tasks, including metabolite identification, confirmation of metabolic site, time-course tracking, and inter-species metabolite comparisons. Offering one software package simplifies and automates the interpretation of complex, unwieldy data sets for the fast deconstruction of metabolite information that is crucial to compound optimization. The multifaceted quant/qual workflow has the flexibility and versatility to deliver metabolite information needed for each stage of drug development: • • • Early drug discovery: High-throughput quant/qual assays can quickly analyze microsomal clearance data, matching fragments to proposed structures to determine critical soft spot information, as well as quantitating parent drug concentrations to establish metabolic stability — all using a generic acquisition methodology in a single run. Late stage discovery: The wide-ranging approaches for data filtration (e.g., neutral loss, product ions, mass defect, isotope patterns) provide multiple methods for the discovery of expected and unexpected metabolites, even low-level, and phase II reactive metabolites. Accurate mass, automated structure-driven processing, and weighted scoring of structures speeds confidence in determining the site of metabolism. Automated correlation of peak areas with analog data delivers easier relative quantitation of metabolite and parent peaks for time course studies. Development stage: Processing of accurate mass and high resolution data, low range mass accuracy, and isotope patterns accelerate fragment assignment for definitive structural elucidation and correlation. Integrated processing of pharmacokinetic batch data using MultiQuant Software reveals metabolite concentrations and kinetic profiles relative to the parent, relying on the fast scanning speeds and the high sensitivity of the TripleTOF system to maintain resolution and to detect minor, low-level metabolites. SWATH Acquisition takes its name from the narrow precursor mass range (or swath) of ions selected in Q1 for advancement to Q2 for collision-induced fragmentation (Figure 3). Relying on the fast scan speed of the TripleTOF 5600 system to sequentially process a broad aggregate of narrow mass ranges in a small amount of time, SWATH Acquisition generates a comprehensive map of MS/MS spectra for every ion at every time point. Effectively simplified using PCVG-filtering and other post-acquisition data mining tools, these complex SWATH Acquisition data sets are transformed into spectra comprised of peaks relevant to the parent drug. In this way, one of the primary challenges of metabolite profiling is overcome – the all-inclusive detection of metabolites, including unpredicted and trace-level, is accomplished just one injection without the need for specialized, sample-specific methods. Using data-independent methods, such as SWATH Acquisition, provides accessible results for more straightforward metabolite structure elucidation and identification. A layer of selectivity is established when using successive narrow mass windows to record MS/MS of the chosen ions within, converting complex MSe spectra to simplified datasets displaying relevant, drug-related peaks. The applicability of SWATH Acquisition data to high-resolution quantitation is extended when data is collected using a TripleTOF 5600 system. Using MRM-style methods to gather single product ion peak areas or to sum multiple product ions, metabolite quantitation can be accomplished with the same sensitivity and the dynamic ranges as those achieved on leading triple quadrupole mass spectrometers. High-resolution acquisition lends additional weight to the data quality by improving the completeness of MS/MS data sets for structural elucidation and permitting narrower mass windows to be used for peak selection, removing potentially confounding interferences from the quantitation process. The impact that the SWATH Acquisition workflow has on throughput and productivity during drug discovery and development is unparalleled. Access to an all-encompassing digital archive of complete MS/MS quantitative and qualitative information for every peak at every time point in a sample is valuable at every stage for increasing productivity, throughput, and data quality. (For a review of the additional features of SWATH Acquisition, see Table 6.) Unique qualitative features 1. 2. 3. RUO-MKT-01-1583-A Another hybrid instrument, the QTRAP 6500 system, a unique combination of triple quadrupole and linear ion trap, delivers the enhanced speed and sensitivity needed for rapidly detecting the most number of metabolites using the minimum number of injections – an efficient approach that complements accurate mass HRMS (see Table 4). During in vivo testing, drugs and metabolites need to be detected at physiologically relevant concentrations, which require an exceedingly sensitive approach. Additionally, early discovery of toxic, reactive metabolites is critical for the selection of appropriate candidates during drug development. Building on the significant gains in sensitivity and fast scanning speeds resulting from modern ion trap innovations, pMRM methods find and confirm the presence of ultra-low level in vivo and in vitro metabolites. These highly sensitive, targeted experiments are based on the parent drug fragmentation pattern and possible biotransformations to yield significantly more metabolites, even from complex biological matrices. Reactive metabolite screening experiments are also enabled by the high scan speeds of the QTRAP system. Dual scan surveys (precursor ion and neutral loss) can be conducted in one fast IDA cycle; and when combined with positive/negative polarity switching, these are the first workflows that can detect metabolites of varying polarities in the same run with sufficient sensitivity and accuracy for the highest level of confidence. In just a single injection, these highly sensitive workflows, pMRM and the dual scan IDA methods, provide the reassurance that an all-encompassing list of even the most minor of metabolites has been generated. Unique quant/qual characteristics 1. SelexION™ Technology drives quantitative workflows, but has the added benefit of boosting qualitative techniques. 2. Other mobility techniques are qual focused, not quant. a. SelexION technology’s pre-ion-source location maintains precursor selection and fragmentation. b. Ion mobility spectrometry (IMS) occurs in the collision cell and provides no selectivity before Q1. c. FAIMS, another ion-mobility-based spectrometry, displays reduced robustness and reliability. 3. Chemical modifiers gives the flexibility to try various options for better separation. 4. SelexION technology is compatible with QTRAP and TripleTOF systems. a. MRM-style application for quantitation (continuous flux of ions) b. Full-scan QTOF (pulsed technique) 5. Easy switch between DMS on and off modes a. Allows ions to be transmitted in transparent mode with voltages turned off b. Easily installed in a few minutes without breaking vacuum or using any tools Table 7: Key features of SelexION™ Technology for metabolite identification. • • Early stage discovery: In combination with pMRM methods, polarity switching and fast scanning can quickly and sensitively detect low-level, transient metabolites, both predicted and unpredicted, in biological samples. The detection of minor, but potentially highly toxic, reactive metabolites by identifying glutathione-conjugates is very important for early safety assessments. Building these compound specific acquisition methods, acquiring the data, and then processing it can be consolidated on just one platform – LightSight Software. Late stage discovery: Highly sensitive pMRM scans used during first-pass, in vitro and in vivo metabolic screening can quickly and efficiently profile metabolites at clinically relevant concentrations, while simultaneously generating quantitative data for metabolic stability and pharmacokinetic studies. The high sensitivity of the linear ion trap detects more trace metabolites and their fragments, which in turn provide more information for structural assignments. Unique quantitative features Less complex MS/MS spectrum than traditional DIA techniques like MSe Retention of isotope pattern for each fragment due to wider Q1 selection • Supports 14C/SIL ADC metabolism studies Capture of 100% MS/MS for comprehensive identification of minor metabolites/catabolites 1. Selective MS/MS quantification realized using MRM-style methods for measurement of single product ions or for summing multiple product ions. 2. Multicomponent quantification is feasible with this single-acquisition method. • Quantification of total mAb, conjugated and free small molecule Table 6: Key features of SWATH™ Acquisition on the TripleTOF® System for metabolite ID. 12 3) Cutting-edge workflows for detecting low-level metabolites: QTRAP 6500 system and LightSight Software INTRODUCTION INTRODUCTION For Research Use Only. Not for use in diagnostic procedures. DRUG METABOLISM Figure 4: An illustration of multiple mass defect ranges that were calculated in MetabolitePilot™ Software. www.absciex.com www.absciex.com DRUG METABOLISM 13 For Research Use Only. Not for use in diagnostic procedures. INTRODUCTION Figure 5: A diagram outlining quant/qual workflows conducted on the TripleTOF® 5600 System. • Development stage: LightSight Software integrates UV or radiolabel quantitation data with MS and MS/MS relative peak areas into one report alongside structure elucidation to provide the relative abundances of metabolites without a reference standard for pharmacokinetic analysis. The sensitivity and speed improvements of an MS/MS workflow are suitable for micro-dosing studies, which permit insight into human pharmacokinetic studies prior to clinical research studies. 4) Eliminating isobaric background and separating isomeric metabolites: SelexION technology workflows Separation of isomeric metabolites and co-eluting, isobaric interferences without the need for longer chromatographic run times and expensive columns is especially challenging during early ADME and definitive metabolite identification workflows, where unwanted, overlapping background can significantly affect the signal-to-noise ratio of quantitative experiments. SelexION technology is an orthogonal, gas-phase separation technique that filters hard-to-separate metabolites that have same molecular weight but different spatial orientation (e.g., the positional isomers of oxidized metabolites, demethylation products, and glucuronide metabolites) and removes co-eluting contaminants that arise during high-throughput UHPLC workflows. Mounted at the entrance to the mass spectrometer, SelexION technology is positioned for the maximum impact on quantitative workflows, while still enabling precursor selection and fragmentation requisite for qualitative analysis during preclinical toxicological safety and efficacy testing. One of the major bottlenecks during definitive metabolite identification during the drug development stage is the separation of isomeric metabolites from complex, in vivo matrices; SelexION technology optimizes the selectivity of these workflows, while at the same time improving the signal for an enhanced signal-to-noise reading. (See Table 7, for additional SelexION technology features.) SelexION technology, compatible with both TripleTOF and QTRAP systems, helps alleviate the separation challenges of a high-throughput environment, with the versatility and ease expected for enhancing productivity. 14 RUO-MKT-01-1583-A INTRODUCTION Perspectives for the future Innovation has expedited the process of screening for drug candidates by achieving higher levels of productivity and automation, anticipating that increased efficiency will allow for more resources to be directed towards new scientific research. The improvements to HRMS performance – higher sensitivity along with higher resolution and mass accuracy at faster scanning speeds – and advances to rapid data acquisition methods have significantly improved the feasibility of drug metabolism studies in complex cellular backgrounds. By using data mining techniques for discriminating drug-related material from background, it is now possible to obtain high quality MS/MS spectra needed for good structural determination without the need to customize the acquisition of each, specific peak, making HRMS-based workflows the technique of choice for high-throughput drug discovery environments. To keep pace with the changing demands of drug discovery and development, AB SCIEX offers innovative high-performance instruments and workhorse software applications that allow for multifaceted metabolite identification tasks to be consolidated into one workflow (Figure 6). Quant/qual workflows conducted on the TripleTOF 5600 system and the QTRAP 6500 system provide the sensitivity and selectivity to detect trace-levels of metabolites in one injection. RT-MMDF algorithms add efficiency to the process of discriminating drug-related material from background noise, ensuring that a comprehensive collection of MS/MS fragments is obtained for structural assignment without sample re-injection. SWATH Acquisition also relies on a transformative algorithm to extract relevant fragment information from data acquired using a compound-independent approach – to ensure that no drug-related material is overlooked. Together, this technology leads the way for the finding, identifying, and confirming metabolites as quickly as possible, without compromising throughput. Even as the extraction of relevant drug-related peaks become increasingly efficient and accessible due to highly-selective filtering techniques and HRMS instrument enhancements, the true bottleneck in metabolite profiling remains in the fast and effective assignment of structure in the discovery stage, a process that is now very manual and hands-on, requiring expert interpretation and input. Progress in this area has been encouraging. Structure databases and software packages have begun to incorporate accurate mass HRMS and MS/MS data information to support automated metabolite structure predictions and localization of the site of metabolism from product ion spectra.4,6 MetabolitePilot Software, for example, includes functionality that assists in (but does not fully automate) the assignment of metabolite structures. As breakthroughs in automation of structural assignment are achieved, hands-on review of the data will ultimately still be needed by experienced scientists, but the number of compounds analyzed will grow exponentially, ultimately unleashing drug development pathways DRUG METABOLISM www.absciex.com Figure 6: Complementary workflows and complete product portfolios available to streamline drug metabolism workflows and to address key challenges. from this long-standing barrier to throughput. Looking to the future, structure elucidation will emerge as a fully automated solution, integrated with intelligently-filtered HRSM data and an integrated cheminformatics approach that predicts structures based on known biotransformation pathways, so that finding, identifying, and confirming metabolites is accelerated to a whole new level of productivity. Chemistry. 2014; 86(2):1202-9. 4 Pahler A, Brink A. “Software aided approaches to structure-based metabolite identification in drug discovery and development.” Drug Discovery Today: Technologies. 2013; 10 (1): 207-217. 5 Zhu, M, Zhang, H, Humphreys WG. “Drug metabolite profiling and identification by highresolution mass spectrometry.”J Biol Chem. 2011; 286 (29): 25419-25425. 6 Zelesky V, Schneider, R, Janiszewski J, Zamora I, Ferguson J, Troutman M. “Software automation tools for increased throughput metabolic soft-spot identification in early drug discovery.” Bioanalysis. 2013; 5(10): 1165-1179. 7 Erve JCL, Gauby S, Maynard, Jr. JW, Svensson MA, Tonn G, Quinn KP. “Bioactivation of sitaxentan in liver microsomes, hepatocytes, and expressed human P450s with characterization of the glutathione conjugate by liquid chromatography tandem mass spectrometry.” Chemical Research in Toxicology. 2013; 26: 926-936. 8 Qiao S, Shi X, Shi R, Liu M, Liu T, Zhang K, Wang Q, Yao M, Zhang L. “Identification of urinary metabolites of imperatorin with a single run on an LC/Triple TOF system based on multiple mass defect filter data acquisition and multiple data mining techniques.” Anal Bioanal Chem. 2013; 405: 6721-6738. 9 Liu M, Zhao S, Wang Z, Wang Y, Liu T, Song L, Wang C, Wang H, Tu P. “Identification of metabolites of deoxyschizandrin in rats by UPLC-Q-TOF-MS/MS based on multiple mass defect filter data acquisition and multiple data processing techniques.” Journal of Chromatography B. 2014; 949-950:115-126. 10 Bateman KP, Cohen L, Emary B, Pucci V. “Standardized workflows for increasing efficiency and productivity in discovery stage bioanalysis.” Bioanalysis. 2013; 5(14): 1783-1794. Abbreviations used MS/MS, tandem mass spectrometry; UHPLC, ultra-high pressure liquid chromatography; HRMS, high-resolution mass spectrometry; ADMET, adsorption, distribution, metabolism, excretion and toxicity; pMRM, predictive multiple reaction monitoring; RT-MMDF, real-time multiple mass defect filter; IDA, information dependent acquisition; DMPK, drug metabolism and pharmacokinetics; DMS, differential mobility spectroscopy; and PCVG, principal components variable grouping. References 1 Lin JH, Lu AYH. “Role of Pharmacokinetics and Metabolism in Drug Discovery and Development.” Pharmacological Reviews. 1997; 49(4): 403-449. 2 Ma S, Chowdhury SK. “Data acquisition and data mining techniques for metabolite identification using LC coupled to high-resolution MS.” Bioanalysis. 2013; 5(10): 1285-1297. 3 Zhu Z, Chen Y, Subramanian R. “Comparison of information-dependent acquisition, SWATH™ acquisition, and MSAll techniques in metabolite identification study employing ultrahighperformance liquid chromatography—quadrupole time-of-flight mass spectrometry.” Analytical www.absciex.com DRUG METABOLISM 15 For Research Use Only. Not for use in diagnostic procedures. INTRODUCTION Figure 5: A diagram outlining quant/qual workflows conducted on the TripleTOF® 5600 System. • Development stage: LightSight Software integrates UV or radiolabel quantitation data with MS and MS/MS relative peak areas into one report alongside structure elucidation to provide the relative abundances of metabolites without a reference standard for pharmacokinetic analysis. The sensitivity and speed improvements of an MS/MS workflow are suitable for micro-dosing studies, which permit insight into human pharmacokinetic studies prior to clinical research studies. 4) Eliminating isobaric background and separating isomeric metabolites: SelexION technology workflows Separation of isomeric metabolites and co-eluting, isobaric interferences without the need for longer chromatographic run times and expensive columns is especially challenging during early ADME and definitive metabolite identification workflows, where unwanted, overlapping background can significantly affect the signal-to-noise ratio of quantitative experiments. SelexION technology is an orthogonal, gas-phase separation technique that filters hard-to-separate metabolites that have same molecular weight but different spatial orientation (e.g., the positional isomers of oxidized metabolites, demethylation products, and glucuronide metabolites) and removes co-eluting contaminants that arise during high-throughput UHPLC workflows. Mounted at the entrance to the mass spectrometer, SelexION technology is positioned for the maximum impact on quantitative workflows, while still enabling precursor selection and fragmentation requisite for qualitative analysis during preclinical toxicological safety and efficacy testing. One of the major bottlenecks during definitive metabolite identification during the drug development stage is the separation of isomeric metabolites from complex, in vivo matrices; SelexION technology optimizes the selectivity of these workflows, while at the same time improving the signal for an enhanced signal-to-noise reading. (See Table 7, for additional SelexION technology features.) SelexION technology, compatible with both TripleTOF and QTRAP systems, helps alleviate the separation challenges of a high-throughput environment, with the versatility and ease expected for enhancing productivity. 14 RUO-MKT-01-1583-A INTRODUCTION Perspectives for the future Innovation has expedited the process of screening for drug candidates by achieving higher levels of productivity and automation, anticipating that increased efficiency will allow for more resources to be directed towards new scientific research. The improvements to HRMS performance – higher sensitivity along with higher resolution and mass accuracy at faster scanning speeds – and advances to rapid data acquisition methods have significantly improved the feasibility of drug metabolism studies in complex cellular backgrounds. By using data mining techniques for discriminating drug-related material from background, it is now possible to obtain high quality MS/MS spectra needed for good structural determination without the need to customize the acquisition of each, specific peak, making HRMS-based workflows the technique of choice for high-throughput drug discovery environments. To keep pace with the changing demands of drug discovery and development, AB SCIEX offers innovative high-performance instruments and workhorse software applications that allow for multifaceted metabolite identification tasks to be consolidated into one workflow (Figure 6). Quant/qual workflows conducted on the TripleTOF 5600 system and the QTRAP 6500 system provide the sensitivity and selectivity to detect trace-levels of metabolites in one injection. RT-MMDF algorithms add efficiency to the process of discriminating drug-related material from background noise, ensuring that a comprehensive collection of MS/MS fragments is obtained for structural assignment without sample re-injection. SWATH Acquisition also relies on a transformative algorithm to extract relevant fragment information from data acquired using a compound-independent approach – to ensure that no drug-related material is overlooked. Together, this technology leads the way for the finding, identifying, and confirming metabolites as quickly as possible, without compromising throughput. Even as the extraction of relevant drug-related peaks become increasingly efficient and accessible due to highly-selective filtering techniques and HRMS instrument enhancements, the true bottleneck in metabolite profiling remains in the fast and effective assignment of structure in the discovery stage, a process that is now very manual and hands-on, requiring expert interpretation and input. Progress in this area has been encouraging. Structure databases and software packages have begun to incorporate accurate mass HRMS and MS/MS data information to support automated metabolite structure predictions and localization of the site of metabolism from product ion spectra.4,6 MetabolitePilot Software, for example, includes functionality that assists in (but does not fully automate) the assignment of metabolite structures. As breakthroughs in automation of structural assignment are achieved, hands-on review of the data will ultimately still be needed by experienced scientists, but the number of compounds analyzed will grow exponentially, ultimately unleashing drug development pathways DRUG METABOLISM www.absciex.com Figure 6: Complementary workflows and complete product portfolios available to streamline drug metabolism workflows and to address key challenges. from this long-standing barrier to throughput. Looking to the future, structure elucidation will emerge as a fully automated solution, integrated with intelligently-filtered HRSM data and an integrated cheminformatics approach that predicts structures based on known biotransformation pathways, so that finding, identifying, and confirming metabolites is accelerated to a whole new level of productivity. Chemistry. 2014; 86(2):1202-9. 4 Pahler A, Brink A. “Software aided approaches to structure-based metabolite identification in drug discovery and development.” Drug Discovery Today: Technologies. 2013; 10 (1): 207-217. 5 Zhu, M, Zhang, H, Humphreys WG. “Drug metabolite profiling and identification by highresolution mass spectrometry.”J Biol Chem. 2011; 286 (29): 25419-25425. 6 Zelesky V, Schneider, R, Janiszewski J, Zamora I, Ferguson J, Troutman M. “Software automation tools for increased throughput metabolic soft-spot identification in early drug discovery.” Bioanalysis. 2013; 5(10): 1165-1179. 7 Erve JCL, Gauby S, Maynard, Jr. JW, Svensson MA, Tonn G, Quinn KP. “Bioactivation of sitaxentan in liver microsomes, hepatocytes, and expressed human P450s with characterization of the glutathione conjugate by liquid chromatography tandem mass spectrometry.” Chemical Research in Toxicology. 2013; 26: 926-936. 8 Qiao S, Shi X, Shi R, Liu M, Liu T, Zhang K, Wang Q, Yao M, Zhang L. “Identification of urinary metabolites of imperatorin with a single run on an LC/Triple TOF system based on multiple mass defect filter data acquisition and multiple data mining techniques.” Anal Bioanal Chem. 2013; 405: 6721-6738. 9 Liu M, Zhao S, Wang Z, Wang Y, Liu T, Song L, Wang C, Wang H, Tu P. “Identification of metabolites of deoxyschizandrin in rats by UPLC-Q-TOF-MS/MS based on multiple mass defect filter data acquisition and multiple data processing techniques.” Journal of Chromatography B. 2014; 949-950:115-126. 10 Bateman KP, Cohen L, Emary B, Pucci V. “Standardized workflows for increasing efficiency and productivity in discovery stage bioanalysis.” Bioanalysis. 2013; 5(14): 1783-1794. Abbreviations used MS/MS, tandem mass spectrometry; UHPLC, ultra-high pressure liquid chromatography; HRMS, high-resolution mass spectrometry; ADMET, adsorption, distribution, metabolism, excretion and toxicity; pMRM, predictive multiple reaction monitoring; RT-MMDF, real-time multiple mass defect filter; IDA, information dependent acquisition; DMPK, drug metabolism and pharmacokinetics; DMS, differential mobility spectroscopy; and PCVG, principal components variable grouping. References 1 Lin JH, Lu AYH. “Role of Pharmacokinetics and Metabolism in Drug Discovery and Development.” Pharmacological Reviews. 1997; 49(4): 403-449. 2 Ma S, Chowdhury SK. “Data acquisition and data mining techniques for metabolite identification using LC coupled to high-resolution MS.” Bioanalysis. 2013; 5(10): 1285-1297. 3 Zhu Z, Chen Y, Subramanian R. “Comparison of information-dependent acquisition, SWATH™ acquisition, and MSAll techniques in metabolite identification study employing ultrahighperformance liquid chromatography—quadrupole time-of-flight mass spectrometry.” Analytical www.absciex.com DRUG METABOLISM 15 Technology Drives High-Performance Biomolecular Mass Spectrometry Enhancing the sensitivity and dynamic range of the AB SCIEX QTRAP® 6500 System with IonDrive™ Technology Bruce Thompson and Bruce Collings AB SCIEX, Concord, ON, Canada In applications that range from proteomics to biomarker discovery to drug development, mass spectrometry has become the tool that provides the highest accuracy and specificity in trace chemical analysis. While there are many important metrics of analytical performance (accuracy, precision, limit of quantitation), they all rely heavily on two key instrumental performance characteristics – sensitivity and dynamic range. In mass spectrometry, instrument sensitivity can best be defined as the number of ions detected per molecule of analyte injected, thus accounting for all losses in ionization, transmission, and detection. Dynamic range is usually defined as the range of linear response of the instrument, limited at the low end by absolute sensitivity and, at the high end, by detector or other instrumentrelated saturation effects. Over the last thirty years of development at AB SCIEX, enormous strides have been made to improve both the instrument sensitivity and the dynamic range. This improved performance has enabled new applications to be addressed by mass spectrometry and has allowed analyses to be performed more rapidly and with greater confidence and higher precision. Higher sensitivity has also enabled the use of additional capabilities and techniques that provide improved analytical specificity – such as higher mass resolution, faster scans speeds, and shorter reaction monitoring times – and techniques such as ion mobility/mass spectrometry Figure 1: The growth in sensitivity of high-flow LC/MS/MS mass spectrometer systems over the last thirty years at AB SCIEX. 16 RUO-MKT-01-1583-A However, the sampling aperture into the vacuum still typically represents the largest area of ion losses. We have, therefore, increased the size of the orifice in order to sample more ions. Improved pumping in the interface helps maintain an acceptable core vacuum pressure without increasing the size of the turbo pumps. The gas expanding through the orifice forms a supersonic free jet with a characteristic barrel shock structure as shown in Figure 2. The high gas flow and pressure provide a strong drag force on the ions that are entrained in this jet, making it more challenging to effectively focus the ions through the next aperture. The new IonDrive™ QJet Ion Drive optics employs a two-stage RF quadrupole to capture and focus the ions to the centerline of the optics using the technique of collisional focusing, allowing the majority of gas to be pumped away. The first section is a large-diameter, RF-only quadrupole with narrow gaps between the rods in order to contain the ions. The narrow gaps minimize the radial outflow of gas, and, therefore, ion losses, while allowing the entrained ions to become collisionally focused. The second section is a smaller diameter quadrupole that provides the final stage of ion-beam focusing while the gas escapes. The transmission efficiency of the ions into the next chamber is approximately 50%, an impressive figure considering the larger orifice diameter, higher pressure, and higher gas velocity. TECHNOLOGY SPOTLIGHT TECHNOLOGY SPOTLIGHT For Research Use Only. Not for use in diagnostic procedures. Figure 2: Cross-sectional view of the IonDrive™ QJet Ion Guide showing the supersonic free jet (supersonic flow region in red) and the gas flow along the axis. combinations added levels of tandem mass spectrometry (MS/MS/MS). The growth curve of sensitivity in AB SCIEX triple quadrupole mass spectrometers over this time period is plotted in Figure 1, which shows a growth of nearly six orders of magnitude in absolute sensitivity since the first AB SCIEX LC/MS/MS product, the TAGA 6000. The AB SCIEX QTRAP® 6500 System, our newest and highest-performance instrument, reaches new levels in both sensitivity and dynamic range. New technologies in both the ion optics and ion counting detection system have driven these performance increases. A key step in achieving higher sensitivity is to create more ions in the source. Over the years, improvements in ionization efficiency have been achieved by increasing the efficiency of desolvation and declustering in the source. The new IonDrive™ Turbo V Source of the QTRAP 6500 system has reached a new level. By optimizing the design of the IonDrive Turbo V source heaters for better and more uniform distribution of heat in the region of droplet evaporation, the efficiency of creating ions in front of the sampling orifice has improved, especially at higher liquid flow rates and for less volatile compounds. DRUG METABOLISM www.absciex.com Figure 3: Dynamic range of the high energy conversion dynode (HED) detection system compared to the standard channel-electron multiplier (CEM) detection system. www.absciex.com DRUG METABOLISM 17 Technology Drives High-Performance Biomolecular Mass Spectrometry Enhancing the sensitivity and dynamic range of the AB SCIEX QTRAP® 6500 System with IonDrive™ Technology Bruce Thompson and Bruce Collings AB SCIEX, Concord, ON, Canada In applications that range from proteomics to biomarker discovery to drug development, mass spectrometry has become the tool that provides the highest accuracy and specificity in trace chemical analysis. While there are many important metrics of analytical performance (accuracy, precision, limit of quantitation), they all rely heavily on two key instrumental performance characteristics – sensitivity and dynamic range. In mass spectrometry, instrument sensitivity can best be defined as the number of ions detected per molecule of analyte injected, thus accounting for all losses in ionization, transmission, and detection. Dynamic range is usually defined as the range of linear response of the instrument, limited at the low end by absolute sensitivity and, at the high end, by detector or other instrumentrelated saturation effects. Over the last thirty years of development at AB SCIEX, enormous strides have been made to improve both the instrument sensitivity and the dynamic range. This improved performance has enabled new applications to be addressed by mass spectrometry and has allowed analyses to be performed more rapidly and with greater confidence and higher precision. Higher sensitivity has also enabled the use of additional capabilities and techniques that provide improved analytical specificity – such as higher mass resolution, faster scans speeds, and shorter reaction monitoring times – and techniques such as ion mobility/mass spectrometry Figure 1: The growth in sensitivity of high-flow LC/MS/MS mass spectrometer systems over the last thirty years at AB SCIEX. 16 RUO-MKT-01-1583-A However, the sampling aperture into the vacuum still typically represents the largest area of ion losses. We have, therefore, increased the size of the orifice in order to sample more ions. Improved pumping in the interface helps maintain an acceptable core vacuum pressure without increasing the size of the turbo pumps. The gas expanding through the orifice forms a supersonic free jet with a characteristic barrel shock structure as shown in Figure 2. The high gas flow and pressure provide a strong drag force on the ions that are entrained in this jet, making it more challenging to effectively focus the ions through the next aperture. The new IonDrive™ QJet Ion Drive optics employs a two-stage RF quadrupole to capture and focus the ions to the centerline of the optics using the technique of collisional focusing, allowing the majority of gas to be pumped away. The first section is a large-diameter, RF-only quadrupole with narrow gaps between the rods in order to contain the ions. The narrow gaps minimize the radial outflow of gas, and, therefore, ion losses, while allowing the entrained ions to become collisionally focused. The second section is a smaller diameter quadrupole that provides the final stage of ion-beam focusing while the gas escapes. The transmission efficiency of the ions into the next chamber is approximately 50%, an impressive figure considering the larger orifice diameter, higher pressure, and higher gas velocity. TECHNOLOGY SPOTLIGHT TECHNOLOGY SPOTLIGHT For Research Use Only. Not for use in diagnostic procedures. Figure 2: Cross-sectional view of the IonDrive™ QJet Ion Guide showing the supersonic free jet (supersonic flow region in red) and the gas flow along the axis. combinations added levels of tandem mass spectrometry (MS/MS/MS). The growth curve of sensitivity in AB SCIEX triple quadrupole mass spectrometers over this time period is plotted in Figure 1, which shows a growth of nearly six orders of magnitude in absolute sensitivity since the first AB SCIEX LC/MS/MS product, the TAGA 6000. The AB SCIEX QTRAP® 6500 System, our newest and highest-performance instrument, reaches new levels in both sensitivity and dynamic range. New technologies in both the ion optics and ion counting detection system have driven these performance increases. A key step in achieving higher sensitivity is to create more ions in the source. Over the years, improvements in ionization efficiency have been achieved by increasing the efficiency of desolvation and declustering in the source. The new IonDrive™ Turbo V Source of the QTRAP 6500 system has reached a new level. By optimizing the design of the IonDrive Turbo V source heaters for better and more uniform distribution of heat in the region of droplet evaporation, the efficiency of creating ions in front of the sampling orifice has improved, especially at higher liquid flow rates and for less volatile compounds. DRUG METABOLISM www.absciex.com Figure 3: Dynamic range of the high energy conversion dynode (HED) detection system compared to the standard channel-electron multiplier (CEM) detection system. www.absciex.com DRUG METABOLISM 17 The increased rate of ion generation in the source and improved transmission efficiency in the ion optics results in a higher ion flux reaching the detector for a given amount of sample injected. At the detector, ions are detected and registered with very high efficiency using a pulse counting system with a very low noise level. The challenge with pulse counting has always been the measurement of high ion signals without saturation. The new high energy conversion dynode (HED) detection system powered by IonDrive™ Technology provides a very significant improvement in this area, extending the upper level of ion counting while maintaining the ability to register single ion events for the best signal-to-noise ratios at the detection limit. The improved dynamic range can be seen in Figure 3, which compares the new HED detection system to the standard channel-electron multiplier (CEM) detection system. In Figure 3, the measured count rate of the first isotope of reserpine is plotted against the true count rate as determined from the known ratio and intensity of its fourth isotope. The new system uses high-energy, ion-to-electron conversion and a low-impedance, multi-channel continuous dynode detector with a closely coupled transimpedance amplifier system that allows high count rates to be sustained without loss of signal. Arrival rates of up to 200 million ions per second can be achieved resulting in a detector linear dynamic range of more than six orders of magnitude. With the sensitivity and dynamic range improvement described above, the QTRAP 6500 system provides a new level of analytical performance, as evidenced by the ability to detect and quantify sub-femtogram amounts of biomolecules injected on-column as shown in Figure 4. Demands for ever decreasing detection limits will continue to drive the need for newer and better methods of ionization, transmission, and detection in the future. However, the growth curve of sensitivity will become more and more difficult to maintain as we approach the limit of measuring and detecting nearly every ion injected. 18 RUO-MKT-01-1583-A TECHNOLOGY SPOTLIGHT TECHNOLOGY SPOTLIGHT For Research Use Only. Not for use in diagnostic procedures. Figure 4: Signal from 500 attograms of verapamil injected on-column monitored in MRM mode using the transition 455/165. DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 19 The increased rate of ion generation in the source and improved transmission efficiency in the ion optics results in a higher ion flux reaching the detector for a given amount of sample injected. At the detector, ions are detected and registered with very high efficiency using a pulse counting system with a very low noise level. The challenge with pulse counting has always been the measurement of high ion signals without saturation. The new high energy conversion dynode (HED) detection system powered by IonDrive™ Technology provides a very significant improvement in this area, extending the upper level of ion counting while maintaining the ability to register single ion events for the best signal-to-noise ratios at the detection limit. The improved dynamic range can be seen in Figure 3, which compares the new HED detection system to the standard channel-electron multiplier (CEM) detection system. In Figure 3, the measured count rate of the first isotope of reserpine is plotted against the true count rate as determined from the known ratio and intensity of its fourth isotope. The new system uses high-energy, ion-to-electron conversion and a low-impedance, multi-channel continuous dynode detector with a closely coupled transimpedance amplifier system that allows high count rates to be sustained without loss of signal. Arrival rates of up to 200 million ions per second can be achieved resulting in a detector linear dynamic range of more than six orders of magnitude. With the sensitivity and dynamic range improvement described above, the QTRAP 6500 system provides a new level of analytical performance, as evidenced by the ability to detect and quantify sub-femtogram amounts of biomolecules injected on-column as shown in Figure 4. Demands for ever decreasing detection limits will continue to drive the need for newer and better methods of ionization, transmission, and detection in the future. However, the growth curve of sensitivity will become more and more difficult to maintain as we approach the limit of measuring and detecting nearly every ion injected. 18 RUO-MKT-01-1583-A TECHNOLOGY SPOTLIGHT TECHNOLOGY SPOTLIGHT For Research Use Only. Not for use in diagnostic procedures. Figure 4: Signal from 500 attograms of verapamil injected on-column monitored in MRM mode using the transition 455/165. DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 19 High-Resolution Time-of-Flight MS for High-Quality Quantitative Analysis Yves Leblanc AB SCIEX, Concord, ON, Canada For many years, quantitative analysis of compounds was conducted on triple quadrupole (QqQ) mass spectrometry (MS) instruments, while qualitative analysis was achieved on mass spectrometry (MS) instruments with high resolution and mass accuracy, such as the time-of-flight Q-ToF MS sytems. As managers are looking into improving the efficiency of laboratory practices and streamlining their decision-making processes for projects ranging from drug metabolism to proteomics, instruments that provide both qualitative and quantitative data simultaneously are strongly desired. Examination of the attributes of MS systems that deliver typical quantitative analysis, reveals the following features: high duty cycle, high sensitivity, high selectivity, and a wide dynamic range. In addition to the innovative front-end developments associated with QqQ systems such as the Turbo V™ Source and the QJet® Ion Guide, key technologies were integrated into the TripleTOF® 5600 System that enabled high-performance quantitative analysis. First, operating the accelerator/pulser at 30 kHz provides high duty-cycle extraction of the ion beam exiting the collision cell. To match this capability, a 40 GHz multichannel time-to-digital (TDC) detection system that ensures a high rate data collection was integrated into the system. Secondly, the instrument operating at 15 kV TOF acceleration voltage with high transmission grids (~92% transparency) assisted in maintaining the sensitivity gains from the front-end changes. Both of these technologies ensure high-efficiency extraction of the ion beam to provide high sensitivity. On the qualitative front, improving system performance in terms of resolution as well as mass accuracy was important. To enhance resolution greater than 30K, the ion optic was optimized to transfer ions with coherent ion trajectories in the pulsar region over a distance of 2.5 m. The last key improvement was to ensure the maintenance of a mass accuracy < 2 ppm RMS over long periods of analysis. With the TripleTOF 5600 system, this is done in two steps: 1) the mass accuracy is established with a scheduled calibration with each batch, and 2) the mass precision is maintained by dynamic monitoring of background ions by adapting ion detection to variations in analysis conditions. As the XIC width is reduced from unit resolution (0.7 Da to mimic quadrupole isolation) to 10 mDa, all interferences are eliminated and a single LC peak is detected. This approach can be further extended to peptide analysis by summing the signal associated with both the charge state distribution as well as the isotope distribution for each ion. Figure 2 shows the observed isotope distribution associated with the +3 and +4 ions of neuromedin U (NMU, seq.: YKVNEYQGPVAPSGGFFLFRPRN). Here, the isotopes contribute to a large portion of the ion signal and each one of them can be combined to give the appropriate signal-to-noise (S/N) for selectivity and proper detection of LC peaks. When neuromedin U was spiked in protein-precipitated plasma, more than 80% of the signal was captured from the top four isotopes. This approach was shown to improve linearity and precision for the detection of NMU. An additional benefit of the TripleTOF 5600 is the ability to record MS/MS spectra retrospectively after separation by liquid chromatography. This mode of operation is referred to as MRM-HR where selective fragment(s) represent the peptide peak. Figure 4 shows an example of the extraction of multiple fragment ions associated a given peptide, in this particular case, a phosphorylated peptide. This allows the experimental mass spectrum to be compared to library entries to ensure that the proper peptide was detected. TECHNOLOGY SPOTLIGHT TECHNOLOGY SPOTLIGHT For Research Use Only. Not for use in diagnostic procedures. With the ability to collect data at rates as high as 50 Hz in either MS or MS/MS mode, the TripleTOF 5600 system offers unique way to support both qualitative and quantitative analysis. First and foremost, the TripleTOF 5600 system is compatible with UPLC separation, and more than 12 data points across most LC peaks can be collected. Secondly, the high resolution (> 30K) and the high mass stability (< 2 ppm rms) provides the ability to extract a narrow ion chromatogram (< 10 mDa) to achieve selectivity in MS mode that is comparable to MRM analysis on QqQ systems. The advantage of this approach is that generic data acquisition can be used for analysis, thus the need for tuning. Equally important is the ability to obtain reliable peak area from narrow extracted ion chromatograms (XIC), which is indicative of the instrument stability in terms of mass accuracy. Figure 1 shows the benefit of reducing the XIC width in order to gain selectivity in the detection of verapamil in diluted urine. Figure 1: Analysis of verapamil in urine samples diluted 2-fold. Data was collected at 30K resolution in MS mode. The width of the XIC was set to mimic single quadrupole analysis (XIC = 0.7 Da) and high resolution mode (XIC = 10 mDa). Figure 2: Isotope distribution of neuromedin U for the dominant charge states (Z=+3 and Z=+4). When the highlighted isotopes are extracted and combined, they represent 91% of the peptide signal, thus improving the overall sensitivity of the system. 20 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 21 High-Resolution Time-of-Flight MS for High-Quality Quantitative Analysis Yves Leblanc AB SCIEX, Concord, ON, Canada For many years, quantitative analysis of compounds was conducted on triple quadrupole (QqQ) mass spectrometry (MS) instruments, while qualitative analysis was achieved on mass spectrometry (MS) instruments with high resolution and mass accuracy, such as the time-of-flight Q-ToF MS sytems. As managers are looking into improving the efficiency of laboratory practices and streamlining their decision-making processes for projects ranging from drug metabolism to proteomics, instruments that provide both qualitative and quantitative data simultaneously are strongly desired. Examination of the attributes of MS systems that deliver typical quantitative analysis, reveals the following features: high duty cycle, high sensitivity, high selectivity, and a wide dynamic range. In addition to the innovative front-end developments associated with QqQ systems such as the Turbo V™ Source and the QJet® Ion Guide, key technologies were integrated into the TripleTOF® 5600 System that enabled high-performance quantitative analysis. First, operating the accelerator/pulser at 30 kHz provides high duty-cycle extraction of the ion beam exiting the collision cell. To match this capability, a 40 GHz multichannel time-to-digital (TDC) detection system that ensures a high rate data collection was integrated into the system. Secondly, the instrument operating at 15 kV TOF acceleration voltage with high transmission grids (~92% transparency) assisted in maintaining the sensitivity gains from the front-end changes. Both of these technologies ensure high-efficiency extraction of the ion beam to provide high sensitivity. On the qualitative front, improving system performance in terms of resolution as well as mass accuracy was important. To enhance resolution greater than 30K, the ion optic was optimized to transfer ions with coherent ion trajectories in the pulsar region over a distance of 2.5 m. The last key improvement was to ensure the maintenance of a mass accuracy < 2 ppm RMS over long periods of analysis. With the TripleTOF 5600 system, this is done in two steps: 1) the mass accuracy is established with a scheduled calibration with each batch, and 2) the mass precision is maintained by dynamic monitoring of background ions by adapting ion detection to variations in analysis conditions. As the XIC width is reduced from unit resolution (0.7 Da to mimic quadrupole isolation) to 10 mDa, all interferences are eliminated and a single LC peak is detected. This approach can be further extended to peptide analysis by summing the signal associated with both the charge state distribution as well as the isotope distribution for each ion. Figure 2 shows the observed isotope distribution associated with the +3 and +4 ions of neuromedin U (NMU, seq.: YKVNEYQGPVAPSGGFFLFRPRN). Here, the isotopes contribute to a large portion of the ion signal and each one of them can be combined to give the appropriate signal-to-noise (S/N) for selectivity and proper detection of LC peaks. When neuromedin U was spiked in protein-precipitated plasma, more than 80% of the signal was captured from the top four isotopes. This approach was shown to improve linearity and precision for the detection of NMU. An additional benefit of the TripleTOF 5600 is the ability to record MS/MS spectra retrospectively after separation by liquid chromatography. This mode of operation is referred to as MRM-HR where selective fragment(s) represent the peptide peak. Figure 4 shows an example of the extraction of multiple fragment ions associated a given peptide, in this particular case, a phosphorylated peptide. This allows the experimental mass spectrum to be compared to library entries to ensure that the proper peptide was detected. TECHNOLOGY SPOTLIGHT TECHNOLOGY SPOTLIGHT For Research Use Only. Not for use in diagnostic procedures. With the ability to collect data at rates as high as 50 Hz in either MS or MS/MS mode, the TripleTOF 5600 system offers unique way to support both qualitative and quantitative analysis. First and foremost, the TripleTOF 5600 system is compatible with UPLC separation, and more than 12 data points across most LC peaks can be collected. Secondly, the high resolution (> 30K) and the high mass stability (< 2 ppm rms) provides the ability to extract a narrow ion chromatogram (< 10 mDa) to achieve selectivity in MS mode that is comparable to MRM analysis on QqQ systems. The advantage of this approach is that generic data acquisition can be used for analysis, thus the need for tuning. Equally important is the ability to obtain reliable peak area from narrow extracted ion chromatograms (XIC), which is indicative of the instrument stability in terms of mass accuracy. Figure 1 shows the benefit of reducing the XIC width in order to gain selectivity in the detection of verapamil in diluted urine. Figure 1: Analysis of verapamil in urine samples diluted 2-fold. Data was collected at 30K resolution in MS mode. The width of the XIC was set to mimic single quadrupole analysis (XIC = 0.7 Da) and high resolution mode (XIC = 10 mDa). Figure 2: Isotope distribution of neuromedin U for the dominant charge states (Z=+3 and Z=+4). When the highlighted isotopes are extracted and combined, they represent 91% of the peptide signal, thus improving the overall sensitivity of the system. 20 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 21 TECHNOLOGY SPOTLIGHT TECHNOLOGY SPOTLIGHT For Research Use Only. Not for use in diagnostic procedures. Figure 4: Narrow-width extracted ion chromatogram (XIC) associated with dominant fragment masses of the illustrated peptides. The full scan mass spectrum can also be compared to library spectrum to gain further confidence in the detection of the peptide. Figure 3: Neuromedin U spiked into protein precipitated plasma. For each charge state, the top 4 isotopes were extracted and summed. The three charge states can also be combined to further improve the sensitivity as little noise is captured in the process. 22 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 23 TECHNOLOGY SPOTLIGHT TECHNOLOGY SPOTLIGHT For Research Use Only. Not for use in diagnostic procedures. Figure 4: Narrow-width extracted ion chromatogram (XIC) associated with dominant fragment masses of the illustrated peptides. The full scan mass spectrum can also be compared to library spectrum to gain further confidence in the detection of the peptide. Figure 3: Neuromedin U spiked into protein precipitated plasma. For each charge state, the top 4 isotopes were extracted and summed. The three charge states can also be combined to further improve the sensitivity as little noise is captured in the process. 22 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 23 PCVG: A Powerful Algorithm for Automating Comprehensive Xenobiotic Metabolite Identification TECHNOLOGY SPOTLIGHT TECHNOLOGY SPOTLIGHT For Research Use Only. Not for use in diagnostic procedures. Laura Baker 1, Suma Ramagiri 2 1 Contract Technical Writer at AB SCIEX, Pittsburgh, PA, 2AB SCIEX, Concord, Canada Key challenges of metabolite identification in complex biological matrices • • • • Overlooking low-level drug metabolites in complex biological matrices such as bile, plasma, and tissue extracts Incomplete metabolite information leading to repeated sample analysis and decreased productivity Non-definitive metabolite identification and characterization due to inadequate MS/MS information Figure 2: SWATH™ Acquisition sequentially collects MS/MS information for selected mass windows (swaths) across a total mass range of interest. Sequential Q1 isolation was stepped over mass range of interest (e.g., 25 Da or user defined). The high speed of the TripleTOF® 5600+ System allows for full coverage of the selected mass range in an LC time scale and for high-resolution XIC data for all fragment ions. Multiple, non-integrated software platforms complicate data processing, slowing metabolite ID and structure elucidation Key benefits of SWATH™ Acquisition and PCVG algorithm for metabolite ID • • • • Comprehensive metabolite fingerprinting of irreplaceable experimental samples using SWATH Acquisition. Having a complete array of spectra (both MS and MS/MS scans) provides a digital archive of all analytes for samples with restricted availability (e.g., pediatric studies, expensive toxicological studies). The ultimate safety net of 100% MS/MS coverage is realized by capturing structural information for both predicted and unpredicted metabolites, including low-level and genotoxic products. MetabolitePilot™ Software is an all-in-one integrated software tool that helps rapidly identify and confirm metabolites with structural elucidation capabilities built-in without the need to switch between multiple software tools. Easy method development and retrospective data-mining -- Requires no sample-specific method development -- Creates a digital archive of all analytes, enabling retrospective investigations without re-acquisition Key features of SWATH Acquisition and PCVG algorithm for metabolite identification • • • • • 24 Selective MS/MS quantitation is achieved using single or multiple product ions that are summed from multiple transitions during multiple reaction monitoring (MRM) experiments. PCVG-correlation of related peaks creates a less complex MS/MS spectrum than traditional data-independent acquisition strategies. Full retention of the isotopic pattern for each fragment due to a wider Q1 selection is ideal for stable-label drug studies (14Cmetabolism studies) and 100% MS/MS coverage for low-level metabolite/catabolite ID. PCVG algorithm enables simplified interpretation and reduction of data dimensionality for complex metabolite spectra generated using data-independent acquisition. PCVG algorithm is a fast, robust, and reliable approach for the deconvolution of multi-component fragment ion spectra that is applied within a research version of MetabolitePilot™ Software. RUO-MKT-01-1583-A Figure 1: SWATH™ Acquisition of all fragment ions for all precursors results in complex data sets. The non-specific fragmentation data collection strategy generates an unbiased record of all fragments of all precursors within one SWATH Acquisition isolation window. Fragments that belong to the same precursor follow the chromatographic profile of that precursor. Drug metabolism studies have traditionally relied upon compoundspecific LC/MS/MS analyses to quantitate and identify metabolites during the drug discovery and development process. Early stage identification of all metabolites – including low-abundance products – is not always possible, and biotransformation scientists occasionally must backtrack, revisiting samples to unearth information on previously-unidentified compounds. If MS/MS spectra could be acquired upfront for all metabolites, both known and unknown, during the drug discovery process, the chances of overlooking an unpredicted metabolite would decrease, saving time and satisfying regulatory requirements more quickly. Novel, data-independent acquisition (DIA) strategies such as SWATH Acquisition, now make non-targeted analyses a reality in complex biological matrices and provide an overall snapshot of lowabundance, genotoxic, and major metabolites. Having complete coverage creates a richer, more detailed picture, but wading through the expanse of data – MS/MS spectra for every fragment of every precursor ion – can be daunting and time-consuming. Deconstructing this amassed data into interpretable results requires a powerful algorithm called principal components variable grouping (PCVG) that effectively filters unabridged MS/MS data to extract comprehensive identification and quantitative information. DRUG METABOLISM www.absciex.com Generating complete metabolite fragment ion data sets using SWATH Acquisition Recently, AB SCIEX scientists showed that PCVG algorithms could rapidly identify and quantify drug metabolites formed in complex biological matrices using DIA methods.1 In these studies, all sample components were acquired in a single injection using SWATH Acquisition, an innovative, non-targeted LC/MS/MS data collection system on TripleTOF® 5600+ system, where an all-inclusive fingerprint is generated from MS/MS scans of every parent ion in the sample (Figure 1). SWATH Acquisition permits a full cluster of ions within a wide Q1 mass window to travel concurrently into the collision cell for fragmentation. Subsequent SWATH scans conducted during the same injection sequentially collect fragment ion information on incrementally increasing mass segments across the total mass range of interest (Figure 2). The resulting composite fragment ion data sets for each drug metabolite sample were laden with thousands of MS/MS spectra, rich in metabolic information. Additionally, a given MS/MS spectrum may be a combination of spectra for two or more metabolites, convoluted in such a way that interpretation of these raw, unprocessed spectra can be complex. grouping (PCVG) into a research version of MetabolitePilot™ Software. PCVG reduces the dimensionality of complex data sets by combining correlated variables into new representative groups that are related to a particular peak in the LC/MS chromatogram, delivering data that is easier to manipulate and understand. The PCVG algorithm uses an unsupervised method to assign related variables to groups, while also filtering out uncorrelated variables. Grouped variables are replaced by a single representative prior to additional data processing, reducing the data set for more efficient interpretation. Deconvolution by the PCVG algorithm proceeds in the following manner: • • • • • • The pre-processed, aligned, multiplexed fragment ion spectra (i.e., SWATH data) undergo principal component analysis (PCA). The variables are m/z values, the samples for the initial PCA are the raw, non-specific fragmentation spectra, and the resulting groups are the pure MS/MS spectra. PCVG analyzes the PCA loadings values to find correlated variables (fragment m/z values). PCVG automates data reduction, filtering variables that do not correlate with the target LC peak profile. The smaller, deconvoluted data set facilitates spectral simplification, aiding in data interpretation. PCVG processing correlates signals across the entire mass range examined, which, in turn, allows researchers to untangle complex relationships from among peaks of interest and to link information on isotopes, adducts, and fragments to related compounds. PCVG filtering finds and correlates related metabolite peaks PCVG deconvolution reduces and simplifies complex multivariate data sets Advances in LC/MS/MS DIA methods have produced fragment ion data sets so vast that discovering the critical connections amongst correlated data points is challenging without further data processing. To simplify multivariate LC/MS data processing within metabolite identification workflows, AB SCIEX scientists have integrated a novel algorithm called principal component variable www.absciex.com Figure 3: TOF MS/MS spectra of nefazodone metabolites collected with IDA and SWATH™ Acquisition methods. SWATH Acquisition with PCVG-filtering result in MS/ MS spectra that contain the full isotope pattern for fragment ions. Both background subtract and PCVG-filtering strategies yielded accurate mass information enabling more confident structure proposal. In the PCVG-filtered TOF MS/MS spectrum, an additional minor peak (m/z 317) that corresponds to a direct substructure of nefazodone was recovered from the raw data. Motivated by the successful reduction of the dimensionality of other LC/MS multivariate data sets obtained for proteomic2 and drug metabolite discoveries, AB SCIEX scientists applied PCVG to xenobiotic metabolite data generated by SWATH Acquisition to derive a fingerprint of all parent-related compounds, creating a more easily interpretable data set that equaled and – for some analytes – surpassed the results obtained using informationdependent acquisition (IDA) methods. After deconvoluting DRUG METABOLISM 25 PCVG: A Powerful Algorithm for Automating Comprehensive Xenobiotic Metabolite Identification TECHNOLOGY SPOTLIGHT TECHNOLOGY SPOTLIGHT For Research Use Only. Not for use in diagnostic procedures. Laura Baker 1, Suma Ramagiri 2 1 Contract Technical Writer at AB SCIEX, Pittsburgh, PA, 2AB SCIEX, Concord, Canada Key challenges of metabolite identification in complex biological matrices • • • • Overlooking low-level drug metabolites in complex biological matrices such as bile, plasma, and tissue extracts Incomplete metabolite information leading to repeated sample analysis and decreased productivity Non-definitive metabolite identification and characterization due to inadequate MS/MS information Figure 2: SWATH™ Acquisition sequentially collects MS/MS information for selected mass windows (swaths) across a total mass range of interest. Sequential Q1 isolation was stepped over mass range of interest (e.g., 25 Da or user defined). The high speed of the TripleTOF® 5600+ System allows for full coverage of the selected mass range in an LC time scale and for high-resolution XIC data for all fragment ions. Multiple, non-integrated software platforms complicate data processing, slowing metabolite ID and structure elucidation Key benefits of SWATH™ Acquisition and PCVG algorithm for metabolite ID • • • • Comprehensive metabolite fingerprinting of irreplaceable experimental samples using SWATH Acquisition. Having a complete array of spectra (both MS and MS/MS scans) provides a digital archive of all analytes for samples with restricted availability (e.g., pediatric studies, expensive toxicological studies). The ultimate safety net of 100% MS/MS coverage is realized by capturing structural information for both predicted and unpredicted metabolites, including low-level and genotoxic products. MetabolitePilot™ Software is an all-in-one integrated software tool that helps rapidly identify and confirm metabolites with structural elucidation capabilities built-in without the need to switch between multiple software tools. Easy method development and retrospective data-mining -- Requires no sample-specific method development -- Creates a digital archive of all analytes, enabling retrospective investigations without re-acquisition Key features of SWATH Acquisition and PCVG algorithm for metabolite identification • • • • • 24 Selective MS/MS quantitation is achieved using single or multiple product ions that are summed from multiple transitions during multiple reaction monitoring (MRM) experiments. PCVG-correlation of related peaks creates a less complex MS/MS spectrum than traditional data-independent acquisition strategies. Full retention of the isotopic pattern for each fragment due to a wider Q1 selection is ideal for stable-label drug studies (14Cmetabolism studies) and 100% MS/MS coverage for low-level metabolite/catabolite ID. PCVG algorithm enables simplified interpretation and reduction of data dimensionality for complex metabolite spectra generated using data-independent acquisition. PCVG algorithm is a fast, robust, and reliable approach for the deconvolution of multi-component fragment ion spectra that is applied within a research version of MetabolitePilot™ Software. RUO-MKT-01-1583-A Figure 1: SWATH™ Acquisition of all fragment ions for all precursors results in complex data sets. The non-specific fragmentation data collection strategy generates an unbiased record of all fragments of all precursors within one SWATH Acquisition isolation window. Fragments that belong to the same precursor follow the chromatographic profile of that precursor. Drug metabolism studies have traditionally relied upon compoundspecific LC/MS/MS analyses to quantitate and identify metabolites during the drug discovery and development process. Early stage identification of all metabolites – including low-abundance products – is not always possible, and biotransformation scientists occasionally must backtrack, revisiting samples to unearth information on previously-unidentified compounds. If MS/MS spectra could be acquired upfront for all metabolites, both known and unknown, during the drug discovery process, the chances of overlooking an unpredicted metabolite would decrease, saving time and satisfying regulatory requirements more quickly. Novel, data-independent acquisition (DIA) strategies such as SWATH Acquisition, now make non-targeted analyses a reality in complex biological matrices and provide an overall snapshot of lowabundance, genotoxic, and major metabolites. Having complete coverage creates a richer, more detailed picture, but wading through the expanse of data – MS/MS spectra for every fragment of every precursor ion – can be daunting and time-consuming. Deconstructing this amassed data into interpretable results requires a powerful algorithm called principal components variable grouping (PCVG) that effectively filters unabridged MS/MS data to extract comprehensive identification and quantitative information. DRUG METABOLISM www.absciex.com Generating complete metabolite fragment ion data sets using SWATH Acquisition Recently, AB SCIEX scientists showed that PCVG algorithms could rapidly identify and quantify drug metabolites formed in complex biological matrices using DIA methods.1 In these studies, all sample components were acquired in a single injection using SWATH Acquisition, an innovative, non-targeted LC/MS/MS data collection system on TripleTOF® 5600+ system, where an all-inclusive fingerprint is generated from MS/MS scans of every parent ion in the sample (Figure 1). SWATH Acquisition permits a full cluster of ions within a wide Q1 mass window to travel concurrently into the collision cell for fragmentation. Subsequent SWATH scans conducted during the same injection sequentially collect fragment ion information on incrementally increasing mass segments across the total mass range of interest (Figure 2). The resulting composite fragment ion data sets for each drug metabolite sample were laden with thousands of MS/MS spectra, rich in metabolic information. Additionally, a given MS/MS spectrum may be a combination of spectra for two or more metabolites, convoluted in such a way that interpretation of these raw, unprocessed spectra can be complex. grouping (PCVG) into a research version of MetabolitePilot™ Software. PCVG reduces the dimensionality of complex data sets by combining correlated variables into new representative groups that are related to a particular peak in the LC/MS chromatogram, delivering data that is easier to manipulate and understand. The PCVG algorithm uses an unsupervised method to assign related variables to groups, while also filtering out uncorrelated variables. Grouped variables are replaced by a single representative prior to additional data processing, reducing the data set for more efficient interpretation. Deconvolution by the PCVG algorithm proceeds in the following manner: • • • • • • The pre-processed, aligned, multiplexed fragment ion spectra (i.e., SWATH data) undergo principal component analysis (PCA). The variables are m/z values, the samples for the initial PCA are the raw, non-specific fragmentation spectra, and the resulting groups are the pure MS/MS spectra. PCVG analyzes the PCA loadings values to find correlated variables (fragment m/z values). PCVG automates data reduction, filtering variables that do not correlate with the target LC peak profile. The smaller, deconvoluted data set facilitates spectral simplification, aiding in data interpretation. PCVG processing correlates signals across the entire mass range examined, which, in turn, allows researchers to untangle complex relationships from among peaks of interest and to link information on isotopes, adducts, and fragments to related compounds. PCVG filtering finds and correlates related metabolite peaks PCVG deconvolution reduces and simplifies complex multivariate data sets Advances in LC/MS/MS DIA methods have produced fragment ion data sets so vast that discovering the critical connections amongst correlated data points is challenging without further data processing. To simplify multivariate LC/MS data processing within metabolite identification workflows, AB SCIEX scientists have integrated a novel algorithm called principal component variable www.absciex.com Figure 3: TOF MS/MS spectra of nefazodone metabolites collected with IDA and SWATH™ Acquisition methods. SWATH Acquisition with PCVG-filtering result in MS/ MS spectra that contain the full isotope pattern for fragment ions. Both background subtract and PCVG-filtering strategies yielded accurate mass information enabling more confident structure proposal. In the PCVG-filtered TOF MS/MS spectrum, an additional minor peak (m/z 317) that corresponds to a direct substructure of nefazodone was recovered from the raw data. Motivated by the successful reduction of the dimensionality of other LC/MS multivariate data sets obtained for proteomic2 and drug metabolite discoveries, AB SCIEX scientists applied PCVG to xenobiotic metabolite data generated by SWATH Acquisition to derive a fingerprint of all parent-related compounds, creating a more easily interpretable data set that equaled and – for some analytes – surpassed the results obtained using informationdependent acquisition (IDA) methods. After deconvoluting DRUG METABOLISM 25 For Research Use Only. Not for use in diagnostic procedures. TECHNOLOGY SPOTLIGHT In combination, SWATH Acquisition and the PCVG algorithm provide a powerful method for confident metabolic structure assignment and offer many advantages when processing complex MS/MS fragment ion data sets for quantitation and identification of metabolites. The following benefits allow for improved selectivity and specificity when pinpointing drug-related material: • • Figure 4: MS/MS obtained using SWATH™ Acquision and retention of the full isotopic pattern for a fragment from Biogen Idec drug candidate, BIIB021.5 MS/MS data were deconvoluted (A) with PCVG filtering and (B) without PCVG-filtering. • • the SWATH Acquisition data, researchers confirmed that the PCVG-filtered MS/MS spectra included identical peaks at similar intensities as those obtained by IDA.1 Shown here, SWATH Acquisition of nefazodone metabolites resulted in full retention of isotopic fragment ions and accurate mass information (Figure 3). The PCVG filters maintained the fidelity of the raw data and revealed a minor characteristic peak (m/z 317) that correlated to the nefazodone structure. In similar experiments, Biogen Idec scientist, Natasha Penner, used SWATH Acquisition to systematically identify metabolites for a drug candidate (Table 1). When comparing SWATH Acquisition data with results obtained using more conventional IDA techniques, Dr. Penner observed an increased number of PCVG-filtered metabolite peaks – 13 out of the 13 known metabolites – with complete MS/MS coverage when using SWATH Acquisition, surpassing results achieved with generic TOF MS data-dependent acquisition. Additionally, each fragment in the MS/MS data filtered by the PCVG algorithm, retained the full isotopic pattern and compared favorably to samples analyzed using traditional MS/MS approaches (Figure 4). Taken together, these data validate and confirm the versatility and accuracy of the PCVG method compared to well-established IDA methods. 26 RUO-MKT-01-1583-A • The ultimate safety net is realized by capturing both predicted and unpredicted metabolites. Having a complete array of spectra (both MS and MS/MS scans) provides researchers a digital archive of all analytes, so that data corresponding to unexpected products can be retrospectively probed without having to re-acquire a sample. Retention of full isotope pattern for each fragment is possible due to the relatively wide swath window, which significantly aids in the designation of metabolite structures and elemental composition during metabolite discovery. This isotopic data along with 100% MS/MS spectra provides sufficient structural information for the identification and quantitation of low-abundance metabolites. A less complex MS/MS spectrum is generated by MS/MSALL with SWATH Acquisition than other DIA techniques. PCVG correlates related peaks, and the relevant spectra make it easier to decide which parent ion goes with which fragment, resulting in higher quality data even with complex data from plasma or bile samples. Easy method development allows researchers to focus on data analysis instead of compound-specific methods. Because MS/MSALL with SWATH Acquisition is a data-independent scan providing quantitative information on all analytes, there is no need to create specialized, data collection strategies for a particular drug candidate; this saves time and unnecessary consumption of limited samples. Multicomponent quantitation is possible with multiple fragment ion transitions captured simultaneously during SWATH Acquisition in a single injection. This adds an additional layer of confidence to quantitative data by allowing multiple product ions to be summed. In summary, the PCVG algorithm provides a fast, robust, and reliable approach for deconvoluting non-specific fragmentation data from drug metabolism studies obtained using SWATH Acquisition. PCVG-filtering diminishes the complexity inherent in large, multivariate data sets, while still creating a global picture of xenobiotic drug metabolites and generating highlyinterpretable spectra for comprehensive metabolite identification and quantitation. Used during the preliminary stages of the drug discovery process, SWATH Acquisition coupled with the powerful PCVG algorithm (incorporated into MetabolitePilot Software) delivers complete metabolite coverage – even of minor products – eliminating the possibility of a missed or underestimated metabolite quantity, thereby streamlining the development of new drug candidates and furthering the understanding of their biotransformation pathways. DRUG METABOLISM www.absciex.com Metabolite Formula RT (min) (M+H)+ Generic TOF-MS SWATH HS Parent C14H15ClN6O 3.89 319.1068 √ (MS/MS) √ (MS/MS) Oxidation-1 C14H15N6O2Cl 3.63 335.1016 √ (MS/MS) √ (MS/MS) Oxidation-2 C14H15N6O2Cl 3.72 335.1016 √ (NO MS/MS) √ (MS/MS) Oxidation-3 C14H15N6O2Cl 3.81 335.1016 √ (MS/MS) √ (MS/MS) Oxidation-4 2 C H N O Cl 4.11 335.1016 √ (NO MS/MS) √ (MS/MS) Dechlorination & loss of CH2 C14H16N6O2 3.27 301.1405 X √ (MS/MS) Demethylation C13H13N6OCl 3.45 305.0914 √ (MS/MS) √ (MS/MS) Internal hydrolysis C14H17N6O2Cl 3.47 337.1174 √ (NO MS/MS) √ (MS/MS) Loss of C5H2N5Cl+oxidation C9H13NO2 0.99 168.1024 √ (NO MS/MS) √ (MS/MS) Loss of C9H11NO C5H4N5Cl 0.98 170.0232 √ (MS/MS) √ (MS/MS) Di-oxidation C14H15N6O3Cl 3.48 351.0967 √ (MS/MS) √ (MS/MS) Ketone formation 2 C H N O Cl 4.11 333.0861 x √ (MS/MS) Dechlorination, loss of CH2 + internal hydrolysis & di-oxidation C13H16N6O4 3.43 321.087 x √ (MS/MS) Loss of Cl di-oxidation C14H16N6O3 2.2 317.1354 x √ (MS/MS) Total metabolite coverage 9/13 (70%) 13/13 (100%) Total MS / MS coverage 5/9 (55%) 13/13 (100%) 14 14 15 13 6 6 TECHNOLOGY SPOTLIGHT Advantages of metabolite discovery with SWATH Acquisition and PCVG-filtering Table 1: Metabolite coverage for Biogen Idec drug candidate BIIB021 in complex biological matrix using generic TOF-MS IDA and SWATH™ Acquisition.5 References 1 Duchoslav E, Ivosev G, Shilov I, Ghobarah H, Burton L. “Automated metabolite identification and profiling in non-specific fragmentation high-resolution accurate MS data.” Poster session presented at: the 61st annual conference of the American Society for Mass Spectromtery; 2013 June 9-13; Minneapolis, MN. 2 Gillet LC, Navarro P, Tate S, Röst H, Slevsek N, Reiter L, Bonner R, Aebersold R. “Targeted data extraction of the MS/MS spectra generated by data-independent acquisition: a new concept for consistent and accurate proteome analysis.” Mol. Cell Proteomics. June 2012. DOI 10.1074/ mcp.0111.016717 3 Ivosev G., Burton L., Bonner R. “Dimensionality reduction and visualization in principal component analysis.” Anal. Chem. July 2008. 80: 4933-4944. 4 Hopfgartner, G. “High-resolution mass spectrometry for integrated qualitative and quantitative analysis of pharmaceuticals in biological matrices.” Anal Bioanal Chem, March 2012. 402(8): 2587-96. 5 Penner N. “High throughput metabolite ID: Are we there yet?” AB SCIEX Mass Spec Webinar Series. July 2013. Retrieved at: http://www.absciex.com/events/webinars/high-throughput metabolite-id-are-we-there-yet www.absciex.com DRUG METABOLISM 27 For Research Use Only. Not for use in diagnostic procedures. TECHNOLOGY SPOTLIGHT In combination, SWATH Acquisition and the PCVG algorithm provide a powerful method for confident metabolic structure assignment and offer many advantages when processing complex MS/MS fragment ion data sets for quantitation and identification of metabolites. The following benefits allow for improved selectivity and specificity when pinpointing drug-related material: • • Figure 4: MS/MS obtained using SWATH™ Acquision and retention of the full isotopic pattern for a fragment from Biogen Idec drug candidate, BIIB021.5 MS/MS data were deconvoluted (A) with PCVG filtering and (B) without PCVG-filtering. • • the SWATH Acquisition data, researchers confirmed that the PCVG-filtered MS/MS spectra included identical peaks at similar intensities as those obtained by IDA.1 Shown here, SWATH Acquisition of nefazodone metabolites resulted in full retention of isotopic fragment ions and accurate mass information (Figure 3). The PCVG filters maintained the fidelity of the raw data and revealed a minor characteristic peak (m/z 317) that correlated to the nefazodone structure. In similar experiments, Biogen Idec scientist, Natasha Penner, used SWATH Acquisition to systematically identify metabolites for a drug candidate (Table 1). When comparing SWATH Acquisition data with results obtained using more conventional IDA techniques, Dr. Penner observed an increased number of PCVG-filtered metabolite peaks – 13 out of the 13 known metabolites – with complete MS/MS coverage when using SWATH Acquisition, surpassing results achieved with generic TOF MS data-dependent acquisition. Additionally, each fragment in the MS/MS data filtered by the PCVG algorithm, retained the full isotopic pattern and compared favorably to samples analyzed using traditional MS/MS approaches (Figure 4). Taken together, these data validate and confirm the versatility and accuracy of the PCVG method compared to well-established IDA methods. 26 RUO-MKT-01-1583-A • The ultimate safety net is realized by capturing both predicted and unpredicted metabolites. Having a complete array of spectra (both MS and MS/MS scans) provides researchers a digital archive of all analytes, so that data corresponding to unexpected products can be retrospectively probed without having to re-acquire a sample. Retention of full isotope pattern for each fragment is possible due to the relatively wide swath window, which significantly aids in the designation of metabolite structures and elemental composition during metabolite discovery. This isotopic data along with 100% MS/MS spectra provides sufficient structural information for the identification and quantitation of low-abundance metabolites. A less complex MS/MS spectrum is generated by MS/MSALL with SWATH Acquisition than other DIA techniques. PCVG correlates related peaks, and the relevant spectra make it easier to decide which parent ion goes with which fragment, resulting in higher quality data even with complex data from plasma or bile samples. Easy method development allows researchers to focus on data analysis instead of compound-specific methods. Because MS/MSALL with SWATH Acquisition is a data-independent scan providing quantitative information on all analytes, there is no need to create specialized, data collection strategies for a particular drug candidate; this saves time and unnecessary consumption of limited samples. Multicomponent quantitation is possible with multiple fragment ion transitions captured simultaneously during SWATH Acquisition in a single injection. This adds an additional layer of confidence to quantitative data by allowing multiple product ions to be summed. In summary, the PCVG algorithm provides a fast, robust, and reliable approach for deconvoluting non-specific fragmentation data from drug metabolism studies obtained using SWATH Acquisition. PCVG-filtering diminishes the complexity inherent in large, multivariate data sets, while still creating a global picture of xenobiotic drug metabolites and generating highlyinterpretable spectra for comprehensive metabolite identification and quantitation. Used during the preliminary stages of the drug discovery process, SWATH Acquisition coupled with the powerful PCVG algorithm (incorporated into MetabolitePilot Software) delivers complete metabolite coverage – even of minor products – eliminating the possibility of a missed or underestimated metabolite quantity, thereby streamlining the development of new drug candidates and furthering the understanding of their biotransformation pathways. DRUG METABOLISM www.absciex.com Metabolite Formula RT (min) (M+H)+ Generic TOF-MS SWATH HS Parent C14H15ClN6O 3.89 319.1068 √ (MS/MS) √ (MS/MS) Oxidation-1 C14H15N6O2Cl 3.63 335.1016 √ (MS/MS) √ (MS/MS) Oxidation-2 C14H15N6O2Cl 3.72 335.1016 √ (NO MS/MS) √ (MS/MS) Oxidation-3 C14H15N6O2Cl 3.81 335.1016 √ (MS/MS) √ (MS/MS) Oxidation-4 2 C H N O Cl 4.11 335.1016 √ (NO MS/MS) √ (MS/MS) Dechlorination & loss of CH2 C14H16N6O2 3.27 301.1405 X √ (MS/MS) Demethylation C13H13N6OCl 3.45 305.0914 √ (MS/MS) √ (MS/MS) Internal hydrolysis C14H17N6O2Cl 3.47 337.1174 √ (NO MS/MS) √ (MS/MS) Loss of C5H2N5Cl+oxidation C9H13NO2 0.99 168.1024 √ (NO MS/MS) √ (MS/MS) Loss of C9H11NO C5H4N5Cl 0.98 170.0232 √ (MS/MS) √ (MS/MS) Di-oxidation C14H15N6O3Cl 3.48 351.0967 √ (MS/MS) √ (MS/MS) Ketone formation 2 C H N O Cl 4.11 333.0861 x √ (MS/MS) Dechlorination, loss of CH2 + internal hydrolysis & di-oxidation C13H16N6O4 3.43 321.087 x √ (MS/MS) Loss of Cl di-oxidation C14H16N6O3 2.2 317.1354 x √ (MS/MS) Total metabolite coverage 9/13 (70%) 13/13 (100%) Total MS / MS coverage 5/9 (55%) 13/13 (100%) 14 14 15 13 6 6 TECHNOLOGY SPOTLIGHT Advantages of metabolite discovery with SWATH Acquisition and PCVG-filtering Table 1: Metabolite coverage for Biogen Idec drug candidate BIIB021 in complex biological matrix using generic TOF-MS IDA and SWATH™ Acquisition.5 References 1 Duchoslav E, Ivosev G, Shilov I, Ghobarah H, Burton L. “Automated metabolite identification and profiling in non-specific fragmentation high-resolution accurate MS data.” Poster session presented at: the 61st annual conference of the American Society for Mass Spectromtery; 2013 June 9-13; Minneapolis, MN. 2 Gillet LC, Navarro P, Tate S, Röst H, Slevsek N, Reiter L, Bonner R, Aebersold R. “Targeted data extraction of the MS/MS spectra generated by data-independent acquisition: a new concept for consistent and accurate proteome analysis.” Mol. Cell Proteomics. June 2012. DOI 10.1074/ mcp.0111.016717 3 Ivosev G., Burton L., Bonner R. “Dimensionality reduction and visualization in principal component analysis.” Anal. Chem. July 2008. 80: 4933-4944. 4 Hopfgartner, G. “High-resolution mass spectrometry for integrated qualitative and quantitative analysis of pharmaceuticals in biological matrices.” Anal Bioanal Chem, March 2012. 402(8): 2587-96. 5 Penner N. “High throughput metabolite ID: Are we there yet?” AB SCIEX Mass Spec Webinar Series. July 2013. Retrieved at: http://www.absciex.com/events/webinars/high-throughput metabolite-id-are-we-there-yet www.absciex.com DRUG METABOLISM 27 DiscoveryQuant™ Software 2.0: The Definitive Solution for LC/MS/MS Early-ADME Workflows Key scientific challenges of early-ADME workflows • • • Overview Productivity is one of the top, if not the main, driver in drug discovery. Sample Pooling – To increase productivity, multiple drugs are often cassette-dosed. Data Sharing – Global companies need to share information globally. Key benefits of the DiscoveryQuant™ Software early-ADME workflows • • • Speed – The software can both optimize the system and prosystems to provide the high levels of sensitivity for MRM analysis. Rapid data review – The ability to quickly visualize the data from a microtiter plate speeds data review. Time-saving templates – Pre-set methods allow users to spend less time setting up runs and more time acquiring data. Key features of DiscoveryQuant Software early-ADME workflows • • • • Cassetting wizard – A pre-set method for pooling samples makes setup fast and simple. Multiply injected assay support – Maximum autosampler speed (Shimadzu, CTC, Agilent, or Acuity) is allowed while continually acquiring data for increased throughput. LIMS connectivity – Improved laboratory informatics are enabled through easily configurable output column mapping taking full advantage of established enterprise-wide systems. Global database support – Compounds optimized in one lab can be used at another without re-optimizing, improving efficiency and productivity across organizations. Today’s high-performance discovery lab performs many different assays on an increasing number of novel compounds. To improve the discovery pipeline, drug discovery labs must gather more information on a greater number of new chemical entities. Platebased technologies, shared conditions information, and pooling strategies are often used to increase throughput in the lab. New software tools can also greatly improve the situation with process automation, information sharing, and functionality monitoring. DiscoveryQuant™ Software 2.0 has been designed specifically for early-ADME workflows in a drug discovery environment. Using a simple and elegant workflow, DiscoveryQuant Software 2.0 is a very powerful and comprehensive software solution that streamlines the drug discovery process by providing better efficiency and productivity. With the Optimize and Analyze modules, researchers can work in parallel on multiple MS systems. The Optimize module will automatically determine the optimal ion path parameters (DP and CE) for maximum quantitative sensitivity for test compounds and then populate a database with this information. The Analyze module can also access MS methods from the database for use in standard assays. Implementing a shared database accessed by labs around the world, different teams can seamlessly collaborate with distant colleagues, working together to avoid duplicating effort. Figure 1: The Optimize module in DiscoveryQuant Software maximizes compound-dependent parameters that are populated into a database. The Analyze module allows users to create quantitative methods for their analytes based on the stored values in the DiscoveryQuant Software database. Analyze is also used to create batches, run studies, integrate data, and generate final reports. 28 RUO-MKT-01-1583-A EARLY DISCOVERY EARLY DISCOVERY For Research Use Only. Not for use in diagnostic procedures. DRUG METABOLISM www.absciex.com Figure 2: The intuitive software interface makes plate review simple and fast. Intuitive interface The graphical user interface has been designed to be intuitive and easy-to-navigate. The main workflows are set out on consecutive tabs at the top of the page that are completed consecutively to optimize method development. Data can be easily imported to avoid manual re-entry, and the information can be saved and recalled as needed. Concise plate review The results of an entire plate can be seen graphically. The colorcoded display gives immediate and clear visualization of results and quick access to individual problem samples with a single click. Express cassetting wizard Compounds for dosing can be grouped directly within the software to facilitate straightforward analytical setup. The intelligently designed DiscoveryQuant Software 2.0 will warn of any conflicts in polarity or interfering parent or fragement masses. Because the positive and negative ion experiments are run separately, it takes twice as long to obtain complete data potentially complicating data analysis with LC retention time Figure 4: Plate templates make large-batch sample entry efficient and easy. www.absciex.com Figure 3: Peak Review allows you to quickly check injection and integration in one interactive window. shifts between samples. Furthermore, greater sample amounts are required for a two-injection workflow. Use of the express cassetting wizard will bypass these issues with assays that require polarity switching. Support for multiple autosampler platforms Maximize costly MS instrument time by continually acquiring over multiple injections, improving throughput and productivity. When the run is done, the software deconvolutes the analysis into individual compound results, reducing instrument calibration and delaying overhead during device communication and synchronization. Rapid review capability Multiple concentrations of a single compound can be reviewed across many injections. The software stitches runs together automatically to give a clear and concise picture of test results. Figure 5: The calibration curve window displays the calibration data and the linear regression for each MRM transition. DRUG METABOLISM 29 DiscoveryQuant™ Software 2.0: The Definitive Solution for LC/MS/MS Early-ADME Workflows Key scientific challenges of early-ADME workflows • • • Overview Productivity is one of the top, if not the main, driver in drug discovery. Sample Pooling – To increase productivity, multiple drugs are often cassette-dosed. Data Sharing – Global companies need to share information globally. Key benefits of the DiscoveryQuant™ Software early-ADME workflows • • • Speed – The software can both optimize the system and prosystems to provide the high levels of sensitivity for MRM analysis. Rapid data review – The ability to quickly visualize the data from a microtiter plate speeds data review. Time-saving templates – Pre-set methods allow users to spend less time setting up runs and more time acquiring data. Key features of DiscoveryQuant Software early-ADME workflows • • • • Cassetting wizard – A pre-set method for pooling samples makes setup fast and simple. Multiply injected assay support – Maximum autosampler speed (Shimadzu, CTC, Agilent, or Acuity) is allowed while continually acquiring data for increased throughput. LIMS connectivity – Improved laboratory informatics are enabled through easily configurable output column mapping taking full advantage of established enterprise-wide systems. Global database support – Compounds optimized in one lab can be used at another without re-optimizing, improving efficiency and productivity across organizations. Today’s high-performance discovery lab performs many different assays on an increasing number of novel compounds. To improve the discovery pipeline, drug discovery labs must gather more information on a greater number of new chemical entities. Platebased technologies, shared conditions information, and pooling strategies are often used to increase throughput in the lab. New software tools can also greatly improve the situation with process automation, information sharing, and functionality monitoring. DiscoveryQuant™ Software 2.0 has been designed specifically for early-ADME workflows in a drug discovery environment. Using a simple and elegant workflow, DiscoveryQuant Software 2.0 is a very powerful and comprehensive software solution that streamlines the drug discovery process by providing better efficiency and productivity. With the Optimize and Analyze modules, researchers can work in parallel on multiple MS systems. The Optimize module will automatically determine the optimal ion path parameters (DP and CE) for maximum quantitative sensitivity for test compounds and then populate a database with this information. The Analyze module can also access MS methods from the database for use in standard assays. Implementing a shared database accessed by labs around the world, different teams can seamlessly collaborate with distant colleagues, working together to avoid duplicating effort. Figure 1: The Optimize module in DiscoveryQuant Software maximizes compound-dependent parameters that are populated into a database. The Analyze module allows users to create quantitative methods for their analytes based on the stored values in the DiscoveryQuant Software database. Analyze is also used to create batches, run studies, integrate data, and generate final reports. 28 RUO-MKT-01-1583-A EARLY DISCOVERY EARLY DISCOVERY For Research Use Only. Not for use in diagnostic procedures. DRUG METABOLISM www.absciex.com Figure 2: The intuitive software interface makes plate review simple and fast. Intuitive interface The graphical user interface has been designed to be intuitive and easy-to-navigate. The main workflows are set out on consecutive tabs at the top of the page that are completed consecutively to optimize method development. Data can be easily imported to avoid manual re-entry, and the information can be saved and recalled as needed. Concise plate review The results of an entire plate can be seen graphically. The colorcoded display gives immediate and clear visualization of results and quick access to individual problem samples with a single click. Express cassetting wizard Compounds for dosing can be grouped directly within the software to facilitate straightforward analytical setup. The intelligently designed DiscoveryQuant Software 2.0 will warn of any conflicts in polarity or interfering parent or fragement masses. Because the positive and negative ion experiments are run separately, it takes twice as long to obtain complete data potentially complicating data analysis with LC retention time Figure 4: Plate templates make large-batch sample entry efficient and easy. www.absciex.com Figure 3: Peak Review allows you to quickly check injection and integration in one interactive window. shifts between samples. Furthermore, greater sample amounts are required for a two-injection workflow. Use of the express cassetting wizard will bypass these issues with assays that require polarity switching. Support for multiple autosampler platforms Maximize costly MS instrument time by continually acquiring over multiple injections, improving throughput and productivity. When the run is done, the software deconvolutes the analysis into individual compound results, reducing instrument calibration and delaying overhead during device communication and synchronization. Rapid review capability Multiple concentrations of a single compound can be reviewed across many injections. The software stitches runs together automatically to give a clear and concise picture of test results. Figure 5: The calibration curve window displays the calibration data and the linear regression for each MRM transition. DRUG METABOLISM 29 Time-saving templates Highlights Plate layouts can be chosen from pre-defined templates that are user-built and then shared. Less time can be spent entering sample locations by entering a few samples for a new plate and then clicking “Propagate Groups” to auto-fill the remaining sample names based on the currently defined pattern. With multiple plates of samples to run, easy sample entry reduces the tedium typically associated with large-batch analysis. • Quick-view calibration curves • • • EARLY DISCOVERY EARLY DISCOVERY For Research Use Only. Not for use in diagnostic procedures. The cassetting wizard makes setup of pooled samples fast and simple. Support for multiple autosampler platforms allows maximum injection speed (Shimadzu, CTC, Agilent, or Acuity) while continually acquiring data for increased throughput. LIMS connectivity enabled through easily configurable output column mapping takes full advantage of established, enterprise-wide systems. Global database support allows compounds optimized in one lab to be used at another without re-optimization, improving efficiency and productivity across organizations. Data export to an external package for regression is eliminated with quick-view calibration curves incorporated within the software package. Regression analysis can be conducted immediately after data acquisition, and graphical results can be viewed internally. Custom column mapping The order and name of column headers can be user-defined, outputting results appear in a .CSV output. This allows files to legacy LIMS systems for a final report, without any intervention or manual modification of the results. Global database setup The global database support in DiscoveryQuant Software 2.0 can be used to store and share mass spectrometry methods. The size of research laboratories can be leveraged to create greater sample capacity and productivity by drastically reducing redundant work. When enabled, the software will automatically look for a method in the database before acquiring data to build a new one. This allows for methods from distant colleagues around the globe to be shared to keep even the most remote labs running consistently and efficiently. Summary DiscoveryQuant Software 2.0 has been designed by experts in early-ADME drug discovery who understand the intricacies and needs of this area. This software has been carefully designed to deliver high-throughput analyses without compromising results – so that efficiency is not sacrificed over quality. This software allows researchers to easily stay connected with colleagues in other ADME research areas throughout an organization. Methods can be stored in the database and shared globally. In an environment where efficiency is vital, duplication of efforts can be avoided to get the most out of mass spectrometry analysis during ADME research. With DiscoveryQuant Software 2.0, the LC/MS/MS early-ADME workflow is completely covered – no more partial solutions or manual data transport to other systems. DiscoveryQuant Software provides a total solution from beginning to end and everywhere in between. Early-ADME workflows have never seen anything like this. For a free 90-day trial of this software, visit: www.absciex.com/discoveryquant 30 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 31 Time-saving templates Highlights Plate layouts can be chosen from pre-defined templates that are user-built and then shared. Less time can be spent entering sample locations by entering a few samples for a new plate and then clicking “Propagate Groups” to auto-fill the remaining sample names based on the currently defined pattern. With multiple plates of samples to run, easy sample entry reduces the tedium typically associated with large-batch analysis. • Quick-view calibration curves • • • EARLY DISCOVERY EARLY DISCOVERY For Research Use Only. Not for use in diagnostic procedures. The cassetting wizard makes setup of pooled samples fast and simple. Support for multiple autosampler platforms allows maximum injection speed (Shimadzu, CTC, Agilent, or Acuity) while continually acquiring data for increased throughput. LIMS connectivity enabled through easily configurable output column mapping takes full advantage of established, enterprise-wide systems. Global database support allows compounds optimized in one lab to be used at another without re-optimization, improving efficiency and productivity across organizations. Data export to an external package for regression is eliminated with quick-view calibration curves incorporated within the software package. Regression analysis can be conducted immediately after data acquisition, and graphical results can be viewed internally. Custom column mapping The order and name of column headers can be user-defined, outputting results appear in a .CSV output. This allows files to legacy LIMS systems for a final report, without any intervention or manual modification of the results. Global database setup The global database support in DiscoveryQuant Software 2.0 can be used to store and share mass spectrometry methods. The size of research laboratories can be leveraged to create greater sample capacity and productivity by drastically reducing redundant work. When enabled, the software will automatically look for a method in the database before acquiring data to build a new one. This allows for methods from distant colleagues around the globe to be shared to keep even the most remote labs running consistently and efficiently. Summary DiscoveryQuant Software 2.0 has been designed by experts in early-ADME drug discovery who understand the intricacies and needs of this area. This software has been carefully designed to deliver high-throughput analyses without compromising results – so that efficiency is not sacrificed over quality. This software allows researchers to easily stay connected with colleagues in other ADME research areas throughout an organization. Methods can be stored in the database and shared globally. In an environment where efficiency is vital, duplication of efforts can be avoided to get the most out of mass spectrometry analysis during ADME research. With DiscoveryQuant Software 2.0, the LC/MS/MS early-ADME workflow is completely covered – no more partial solutions or manual data transport to other systems. DiscoveryQuant Software provides a total solution from beginning to end and everywhere in between. Early-ADME workflows have never seen anything like this. For a free 90-day trial of this software, visit: www.absciex.com/discoveryquant 30 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 31 Confirmation of In Vitro Nefazodone Metabolites Using the Superior Fragmentation of the QTRAP® 5500 LC/MS/MS System Claire Bramwell-German, Elliott Jones, and Daniel Lebre AB SCIEX, Redwood City, California and Concord, Ontario, Canada Figure 1: Predictive multiple reaction monitoring (pMRM) example. This shows the pMRM transition prediction for a transformation that is a combination of oxidation and demethylation. In this case LightSight® Software 2.1 creates four MRMs which will cover any fragment/transformation possibility. Since the MRM method has a better duty cycle than any other MS method, sensitivity and selectivity are very important in this mode. Key scientific challenges of metabolite ID • • • Productivity – Increasing productivity in drug discovery and development continues to be a primary goal in pharmaceutical research. Sensitivity – The ability to detect metabolites at physiologically relevant concentrations is critical to drug development, and early detection minimizes development costs for drugs that shouldn’t progress to the development stage. Introduction Metabolite profiling plays a critical role in the discovery and developmental level of analysis for lead drug candidates. Many aspects of drug activity are affected by metabolism (pharmacokinetics, bioavailability, drug distribution, toxicity, and adverse drug reactions). The enhanced scan rate (4-fold for quadrupole and 5-fold for LIT modes) and sensitivity increases of (8- to 10-fold response in quadrupole mode and 10- to 100-fold in LIT mode) allow for a number of unique experiments with unprecedented sensitivity. Ease of use – Most metabolite ID workflows require a skilled operator to acquire and process the data, often using multiple software packages. Key benefits of the QTRAP® 5500 Systems and LightSight® Software 2.1 for metabolite ID • • • Comprehensive software package – LightSight Software can be used to build compound-specific methods, acquire the data, and then process it. Selective and sensitive scans – The QTRAP systems have selective and sensitive scan modes that can be combined to find the maximum number of metabolites in the minimal number of injections. Increased throughput – The faster speed of this instrument makes it UHPLC compatible, providing greater sample throughput. Key features of the QTRAP 5500 systems and LightSight Software 2.1 for metabolite ID • • • Unique capabilities – With combinable quadrupole and ion trap scan modes available in one instrument coupled with advanced technologies, rapid, thorough metabolite ID is easily achievable. Speed – Hardware improvements provide a system that is UHPLC-compatible. One software package – With the built-in ability to both acquire and process data, LightSight Software makes the work of the biotransformation chemist much easier. Overview Recently, nefazodone (an antidepressant) has been targeted for metabolite studies due to the quantity and diversity of biotransformations resulting from CYP450 enzymatic activity. Several scientific publications have evaluated different workflows and mass spectrometry platforms for this specific application.1-3 Herein, we report an effective and sensitive method to identify nefazodone metabolites using the new QTRAP 5500 LC/MS/MS system. The system’s quadrupole and linear ion trap sensitivity and scan speed have been significantly improved, allowing for highly effective detection and confirmation of in vitro and in vivo metabolites. The faster scan rates are a perfect match for small particle, high-pressure LC workflows for better separation and throughput. Using a predictive multiple reaction monitoring (pMRM) approach and a rapid, 10-minute chromatographic analysis, a significant number of metabolites were found and confirmed in a single injection – more than any other MS approach found in previous literature references. This level of identification in a rapid chromatographic analysis demonstrates the abilities of the QTRAP 5500 system for high throughput and a detailed metabolite analysis, with a depth of detection not possible when using other MS technologies. Furthermore, the improved sensitivity in the linear ion trap (LIT) mode allows for fine structural confirmation, since a greater number of diagnostic low-level fragments can be detected compared to less sensitive accurate mass systems. The most effective scan mode for first pass metabolic screening on the QTRAP 5500 system is pMRM. Figure 1 shows an example of pMRM detection in which a comprehensive list of possible biotransformations along with parent drug fragment pattern are used to create a range of theoretically derived MRMs to cover all possible metabolite transitions. The only type of fragmentation pattern where pMRM might miss a metabolite is N-dealkylation and other exotic breaks in the drug core structure. In this case, a second injection using a dual precursor and neutral loss survey based on the parent drug’s fragmentation pattern can be employed to pick up any overlooked metabolites. This second injection would not be required if the molecule is unlikely to undergo an N-dealkylation transformation. The strength of the QTRAP 5500 system is that a cycle of ~300 pMRMs or a dual precursor and neutral loss survey with four dependent ion trap MS/MS can be done approximately 4-fold faster than on the current 3200 and 4000 QTRAP systems. For example, a dual scan experiment for both a precursor and neutral loss, with four enhanced product ion (EPI) scans running at the fastest scan rate possible on the 4000 QTRAP system could take ~7.5 sec. Because this cycle rate is too slow for most chromatographic peaks, only a single survey and one or two EPI scans could be done within an LC/MS time frame. However, as the QTRAP 5500 system can scan 4- to 5-fold faster, this same cycle would be reduced to around ~2 sec. A 2 sec cycle rate will allow for at least three data points to be acquired over even a very sharp 6 sec chromatographic peak. The combined precursor and neutral loss experiment is also particularly effective because few metabolic changes would affect both the neutral and charged parts of the molecule, allowing for broad metabolite detection. In the case of many co-eluting metabolites, a greater number of dependent EPI scans is possible based on the 5-fold faster LIT scan rate, which will also allow for more MS/MS spectra to be acquired in a single injection. Thus the dual scan survey with four EPI scans can allow for greater metabolite detection without compromising scan rate. (Dynamic Background Subtraction (DBS) is another way to ensure efficient ion selection during an automated experiment and doesn’t require more than one dependent EPI scan, significantly reducing the cycle time.) This optimal mode is only possible on a QTRAP 5500 system. Similarly to the dual survey mode, pMRM experiments also benefit from a 4-fold reduction in scan rate compared to the 4000 QTRAP system in much the same way. EARLY DISCOVERY EARLY DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Figure 2: Sensitivity gains on the QTRAP® 5500 LC/MS/MS System over the 4000 QTRAP® System. Three metabolites with corresponding linear ion trap (LIT) MS/MS spectra are compared for the QTRAP 5500 system (left panels) and the 4000 QTRAP system (right panels.) In each case, a significant increase in the number of interpretable fragments is obtained by the QTRAP 5500 system. 32 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 33 Confirmation of In Vitro Nefazodone Metabolites Using the Superior Fragmentation of the QTRAP® 5500 LC/MS/MS System Claire Bramwell-German, Elliott Jones, and Daniel Lebre AB SCIEX, Redwood City, California and Concord, Ontario, Canada Figure 1: Predictive multiple reaction monitoring (pMRM) example. This shows the pMRM transition prediction for a transformation that is a combination of oxidation and demethylation. In this case LightSight® Software 2.1 creates four MRMs which will cover any fragment/transformation possibility. Since the MRM method has a better duty cycle than any other MS method, sensitivity and selectivity are very important in this mode. Key scientific challenges of metabolite ID • • • Productivity – Increasing productivity in drug discovery and development continues to be a primary goal in pharmaceutical research. Sensitivity – The ability to detect metabolites at physiologically relevant concentrations is critical to drug development, and early detection minimizes development costs for drugs that shouldn’t progress to the development stage. Introduction Metabolite profiling plays a critical role in the discovery and developmental level of analysis for lead drug candidates. Many aspects of drug activity are affected by metabolism (pharmacokinetics, bioavailability, drug distribution, toxicity, and adverse drug reactions). The enhanced scan rate (4-fold for quadrupole and 5-fold for LIT modes) and sensitivity increases of (8- to 10-fold response in quadrupole mode and 10- to 100-fold in LIT mode) allow for a number of unique experiments with unprecedented sensitivity. Ease of use – Most metabolite ID workflows require a skilled operator to acquire and process the data, often using multiple software packages. Key benefits of the QTRAP® 5500 Systems and LightSight® Software 2.1 for metabolite ID • • • Comprehensive software package – LightSight Software can be used to build compound-specific methods, acquire the data, and then process it. Selective and sensitive scans – The QTRAP systems have selective and sensitive scan modes that can be combined to find the maximum number of metabolites in the minimal number of injections. Increased throughput – The faster speed of this instrument makes it UHPLC compatible, providing greater sample throughput. Key features of the QTRAP 5500 systems and LightSight Software 2.1 for metabolite ID • • • Unique capabilities – With combinable quadrupole and ion trap scan modes available in one instrument coupled with advanced technologies, rapid, thorough metabolite ID is easily achievable. Speed – Hardware improvements provide a system that is UHPLC-compatible. One software package – With the built-in ability to both acquire and process data, LightSight Software makes the work of the biotransformation chemist much easier. Overview Recently, nefazodone (an antidepressant) has been targeted for metabolite studies due to the quantity and diversity of biotransformations resulting from CYP450 enzymatic activity. Several scientific publications have evaluated different workflows and mass spectrometry platforms for this specific application.1-3 Herein, we report an effective and sensitive method to identify nefazodone metabolites using the new QTRAP 5500 LC/MS/MS system. The system’s quadrupole and linear ion trap sensitivity and scan speed have been significantly improved, allowing for highly effective detection and confirmation of in vitro and in vivo metabolites. The faster scan rates are a perfect match for small particle, high-pressure LC workflows for better separation and throughput. Using a predictive multiple reaction monitoring (pMRM) approach and a rapid, 10-minute chromatographic analysis, a significant number of metabolites were found and confirmed in a single injection – more than any other MS approach found in previous literature references. This level of identification in a rapid chromatographic analysis demonstrates the abilities of the QTRAP 5500 system for high throughput and a detailed metabolite analysis, with a depth of detection not possible when using other MS technologies. Furthermore, the improved sensitivity in the linear ion trap (LIT) mode allows for fine structural confirmation, since a greater number of diagnostic low-level fragments can be detected compared to less sensitive accurate mass systems. The most effective scan mode for first pass metabolic screening on the QTRAP 5500 system is pMRM. Figure 1 shows an example of pMRM detection in which a comprehensive list of possible biotransformations along with parent drug fragment pattern are used to create a range of theoretically derived MRMs to cover all possible metabolite transitions. The only type of fragmentation pattern where pMRM might miss a metabolite is N-dealkylation and other exotic breaks in the drug core structure. In this case, a second injection using a dual precursor and neutral loss survey based on the parent drug’s fragmentation pattern can be employed to pick up any overlooked metabolites. This second injection would not be required if the molecule is unlikely to undergo an N-dealkylation transformation. The strength of the QTRAP 5500 system is that a cycle of ~300 pMRMs or a dual precursor and neutral loss survey with four dependent ion trap MS/MS can be done approximately 4-fold faster than on the current 3200 and 4000 QTRAP systems. For example, a dual scan experiment for both a precursor and neutral loss, with four enhanced product ion (EPI) scans running at the fastest scan rate possible on the 4000 QTRAP system could take ~7.5 sec. Because this cycle rate is too slow for most chromatographic peaks, only a single survey and one or two EPI scans could be done within an LC/MS time frame. However, as the QTRAP 5500 system can scan 4- to 5-fold faster, this same cycle would be reduced to around ~2 sec. A 2 sec cycle rate will allow for at least three data points to be acquired over even a very sharp 6 sec chromatographic peak. The combined precursor and neutral loss experiment is also particularly effective because few metabolic changes would affect both the neutral and charged parts of the molecule, allowing for broad metabolite detection. In the case of many co-eluting metabolites, a greater number of dependent EPI scans is possible based on the 5-fold faster LIT scan rate, which will also allow for more MS/MS spectra to be acquired in a single injection. Thus the dual scan survey with four EPI scans can allow for greater metabolite detection without compromising scan rate. (Dynamic Background Subtraction (DBS) is another way to ensure efficient ion selection during an automated experiment and doesn’t require more than one dependent EPI scan, significantly reducing the cycle time.) This optimal mode is only possible on a QTRAP 5500 system. Similarly to the dual survey mode, pMRM experiments also benefit from a 4-fold reduction in scan rate compared to the 4000 QTRAP system in much the same way. EARLY DISCOVERY EARLY DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Figure 2: Sensitivity gains on the QTRAP® 5500 LC/MS/MS System over the 4000 QTRAP® System. Three metabolites with corresponding linear ion trap (LIT) MS/MS spectra are compared for the QTRAP 5500 system (left panels) and the 4000 QTRAP system (right panels.) In each case, a significant increase in the number of interpretable fragments is obtained by the QTRAP 5500 system. 32 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 33 Another differential feature is the LIT MS/MS sensitivity. Compared to the 4000 QTRAP system, an improvement of 10- to 100-fold in intensity is apparent for data collected on the QTRAP 5500 system. This has two main benefits, with the first being the detection of metabolites at an unprecedented low level. Often in in vivo pharmacokinetic work, MRM methods track metabolites at very low levels. For the first time, the QTRAP 5500 system can now allow qualitative identification of metabolites at nearly equivalent levels. The second benefit is the acquisition of greater number of fragments available for detailed structural identification on the QTRAP 5500 system; the presence of more fragments aids in greater structural refinement of interpretation. Figure 2 shows an example of the QTRAP 5500 system and 4000 QTRAP system in terms of the number of interpretable fragments for the same metabolites. To illustrate these points, a nefazodone incubation was analyzed on the QTRAP 5500 system, and two survey modes were used: 1) pMRM information-dependent acquisition (IDA) and 2) dual scan precursor and neutral loss IDA. A number of highly refined MS/MS confirmations will be presented to demonstrate the level of refinement possible using the high sensitivity LIT MS/MS of the new QTRAP 5500 system. Experimental conditions Samples for analysis: Control and incubations (20 µL each) were diluted with 200 µL 95% (5.0 mM ammonium formate)/5% ACN (v/v) prior to injection. After dilution, the final concentration of nefazodone in the control solution was 2.5 µM. Chromatography (LC) System: Shimadzu Prominence (Shimadzu Corp, Kyoto, Japan) Column: Stable Bond C18 (4.6 mm ID x 50 mm) 1.8 µm (Agilent, Palo Alto, CA) Mobile phase: A) 0.1% Formic Acid B) Acetonitrile + 0.1% Formic Acid Column oven temperature: 45 ºC Flow rate: 1.00 mL/min Injection volume: 10 µL Gradient runtime: 10.0 min (Table 1) Mass spectrometry Incubations Solutions: 50 mM phosphate buffer (pH 7.4) 5.0 mM NADPH in phosphate buffer Solution A. 2.0 mg/mL rat microsomes in phosphate buffer Solution B. 20 µM nefazodone in 5.0 mM NADPH Solution C. Acetronitrile Control (t = 0 min): 200 µL A, 200 µL B, and 400 µL C were added to a 1.8 mL Eppendorf tube (final volume, 800 µL) EARLY DISCOVERY EARLY DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Step Time (min) Flow Rate (µL/min) %A %B 0 0.0 1,000 95 5 1 1.0 1,000 95 5 2 2.0 1,000 70 30 3 10.0 1,000 60 40 4 10.1 1,000 5 95 95 5 10.5 1,000 5 6 10.6 1,000 95 5 7 15.0 1,000 95 5 Figure 3: Sample/control comparison in LightSight® Software. In this example, a low level of N-dealkylation, oxidation, and bis-dehydrogenation metabolite was observed. The MS/MS comparison list (middle right) allows for a quick comparison of metabolite versus parent fragmentation patterns. The LIT MS/MS sensitivity QTRAP® 5500 System is excellent for the detection of low-level (0.1%) metabolites based on the MRM total ion current (TIC). Figure 4: Fragment analysis of the same N-dealkylation, oxidation, and bisdehydrogenation metabolites from Figure 3. The ACD fragmenter overlays structural fragment assignments on top of the MS spectra. The user can then scroll through the list deselecting any incorrect predictions. Results Figure 4 shows the fragment assignment accomplished using the ACD Fragmenter. In this case, 80% of the fragments were assigned using the automated workflow. The metabolically introduced carbonyl group is likely to be on the 6, 7, 8, or 12 carbon atom. A number of novel metabolites will be identified that demonstrate the ultra-high sensitivity of the LIT mode on the QTRAP 5500 system. Initial screening of the metabolites was accomplished by sample and control comparison using LightSight Software. Figure 3 shows the extracted ion chromatogram (XIC) trace, MS/MS spectrum, and other relevant information in the LightSight Software interface for an N-dealkylation, oxidation, and bis-dehydrogenation metabolite, representing 0.1% of the total ion chromatogram (TIC) area, a good example of a lowlevel species. Even for this trace-level metabolite, 18 high-quality fragments are apparent in the spectrum. LightSight Software 2.1 allows spectra to be directly imported into ACD Spectrum Manager and Fragmenter™ for MS/MS automated analysis. One of the other interesting metabolic changes is, an N-dealkylation plus oxidation and dehydrogenation, found at a retention time of 4.13 minutes. Figure 5 shows the XIC and MS/MS spectra for fragments identified by LightSight Software. Examination of the MS/MS spectra reveals 19 fragments of high quality. The ACD Fragmenter was able to assign 70% of these fragments to the predicted structure with minimal manual review (Figure 6). The metabolically introduced ketone group is likely to be at positions 21, 20, 11, and 10 on the backbone of the structure. Table 1: Gradient composition. The tube was vortexed for 1 min, incubated 2-3 min on ice, and then centrifuged for 2 min at 10,000 rpm. Final concentration of nefazodone is 5.0 µM in 1.25 mM NADPH and 25 mM phosphate buffer. Incubation (t = 60 min): 200 µL A and 200 µL B were added to a 1.8 mL Eppendorf tube (final volume, 800 µL) This mixture was incubated for 60 min at 37 °C with shaking (Eppendorf apparatus, 350 rpm agitation) The reaction was quenched with 400 µL of C The QTRAP 5500 system and LightSight Software 2.1 were used for all LC/MS experiments. A single injection pMRM approach was employed, triggering the collection of confirmatory MS/MS spectra in an automated experiment (IDA). A second injection used a dual scan parent precursor/neutral loss IDA method, although no major metabolites were missed in the first run. In both cases, the 20,000 Da/sec scan rate was used with four dependent EPI scans. All methods were generated using the LightSight Software 2.1 automatic method builder. The mixture was vortexed for 1 min, incubated on ice for 2-3 min, and then centrifuged for 2 min at 10,000 rpm. Figure 5: Extracted ion chromatogram (XIC) and MS/MS spectra for a N-dealkylation fragment of nefazodone. A peak representing the N-dealkylation of the chlorinecontaining ring on the left half of nefazodone is shown and includes additional sites of oxidation and dehydrogenation. The high LIT sensitivity of the QTRAP® 5500 System provides superior fragmentation detail. 34 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 35 Another differential feature is the LIT MS/MS sensitivity. Compared to the 4000 QTRAP system, an improvement of 10- to 100-fold in intensity is apparent for data collected on the QTRAP 5500 system. This has two main benefits, with the first being the detection of metabolites at an unprecedented low level. Often in in vivo pharmacokinetic work, MRM methods track metabolites at very low levels. For the first time, the QTRAP 5500 system can now allow qualitative identification of metabolites at nearly equivalent levels. The second benefit is the acquisition of greater number of fragments available for detailed structural identification on the QTRAP 5500 system; the presence of more fragments aids in greater structural refinement of interpretation. Figure 2 shows an example of the QTRAP 5500 system and 4000 QTRAP system in terms of the number of interpretable fragments for the same metabolites. To illustrate these points, a nefazodone incubation was analyzed on the QTRAP 5500 system, and two survey modes were used: 1) pMRM information-dependent acquisition (IDA) and 2) dual scan precursor and neutral loss IDA. A number of highly refined MS/MS confirmations will be presented to demonstrate the level of refinement possible using the high sensitivity LIT MS/MS of the new QTRAP 5500 system. Experimental conditions Samples for analysis: Control and incubations (20 µL each) were diluted with 200 µL 95% (5.0 mM ammonium formate)/5% ACN (v/v) prior to injection. After dilution, the final concentration of nefazodone in the control solution was 2.5 µM. Chromatography (LC) System: Shimadzu Prominence (Shimadzu Corp, Kyoto, Japan) Column: Stable Bond C18 (4.6 mm ID x 50 mm) 1.8 µm (Agilent, Palo Alto, CA) Mobile phase: A) 0.1% Formic Acid B) Acetonitrile + 0.1% Formic Acid Column oven temperature: 45 ºC Flow rate: 1.00 mL/min Injection volume: 10 µL Gradient runtime: 10.0 min (Table 1) Mass spectrometry Incubations Solutions: 50 mM phosphate buffer (pH 7.4) 5.0 mM NADPH in phosphate buffer Solution A. 2.0 mg/mL rat microsomes in phosphate buffer Solution B. 20 µM nefazodone in 5.0 mM NADPH Solution C. Acetronitrile Control (t = 0 min): 200 µL A, 200 µL B, and 400 µL C were added to a 1.8 mL Eppendorf tube (final volume, 800 µL) EARLY DISCOVERY EARLY DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Step Time (min) Flow Rate (µL/min) %A %B 0 0.0 1,000 95 5 1 1.0 1,000 95 5 2 2.0 1,000 70 30 3 10.0 1,000 60 40 4 10.1 1,000 5 95 95 5 10.5 1,000 5 6 10.6 1,000 95 5 7 15.0 1,000 95 5 Figure 3: Sample/control comparison in LightSight® Software. In this example, a low level of N-dealkylation, oxidation, and bis-dehydrogenation metabolite was observed. The MS/MS comparison list (middle right) allows for a quick comparison of metabolite versus parent fragmentation patterns. The LIT MS/MS sensitivity QTRAP® 5500 System is excellent for the detection of low-level (0.1%) metabolites based on the MRM total ion current (TIC). Figure 4: Fragment analysis of the same N-dealkylation, oxidation, and bisdehydrogenation metabolites from Figure 3. The ACD fragmenter overlays structural fragment assignments on top of the MS spectra. The user can then scroll through the list deselecting any incorrect predictions. Results Figure 4 shows the fragment assignment accomplished using the ACD Fragmenter. In this case, 80% of the fragments were assigned using the automated workflow. The metabolically introduced carbonyl group is likely to be on the 6, 7, 8, or 12 carbon atom. A number of novel metabolites will be identified that demonstrate the ultra-high sensitivity of the LIT mode on the QTRAP 5500 system. Initial screening of the metabolites was accomplished by sample and control comparison using LightSight Software. Figure 3 shows the extracted ion chromatogram (XIC) trace, MS/MS spectrum, and other relevant information in the LightSight Software interface for an N-dealkylation, oxidation, and bis-dehydrogenation metabolite, representing 0.1% of the total ion chromatogram (TIC) area, a good example of a lowlevel species. Even for this trace-level metabolite, 18 high-quality fragments are apparent in the spectrum. LightSight Software 2.1 allows spectra to be directly imported into ACD Spectrum Manager and Fragmenter™ for MS/MS automated analysis. One of the other interesting metabolic changes is, an N-dealkylation plus oxidation and dehydrogenation, found at a retention time of 4.13 minutes. Figure 5 shows the XIC and MS/MS spectra for fragments identified by LightSight Software. Examination of the MS/MS spectra reveals 19 fragments of high quality. The ACD Fragmenter was able to assign 70% of these fragments to the predicted structure with minimal manual review (Figure 6). The metabolically introduced ketone group is likely to be at positions 21, 20, 11, and 10 on the backbone of the structure. Table 1: Gradient composition. The tube was vortexed for 1 min, incubated 2-3 min on ice, and then centrifuged for 2 min at 10,000 rpm. Final concentration of nefazodone is 5.0 µM in 1.25 mM NADPH and 25 mM phosphate buffer. Incubation (t = 60 min): 200 µL A and 200 µL B were added to a 1.8 mL Eppendorf tube (final volume, 800 µL) This mixture was incubated for 60 min at 37 °C with shaking (Eppendorf apparatus, 350 rpm agitation) The reaction was quenched with 400 µL of C The QTRAP 5500 system and LightSight Software 2.1 were used for all LC/MS experiments. A single injection pMRM approach was employed, triggering the collection of confirmatory MS/MS spectra in an automated experiment (IDA). A second injection used a dual scan parent precursor/neutral loss IDA method, although no major metabolites were missed in the first run. In both cases, the 20,000 Da/sec scan rate was used with four dependent EPI scans. All methods were generated using the LightSight Software 2.1 automatic method builder. The mixture was vortexed for 1 min, incubated on ice for 2-3 min, and then centrifuged for 2 min at 10,000 rpm. Figure 5: Extracted ion chromatogram (XIC) and MS/MS spectra for a N-dealkylation fragment of nefazodone. A peak representing the N-dealkylation of the chlorinecontaining ring on the left half of nefazodone is shown and includes additional sites of oxidation and dehydrogenation. The high LIT sensitivity of the QTRAP® 5500 System provides superior fragmentation detail. 34 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 35 EARLY DISCOVERY EARLY DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Figure 6: The fragmentation pattern of an N-demethylation metabolite of nefazodone. Fragmentation of a nefazodine metabolite with an N-dealkylation of the chlorinecontaining ring on the left half of the molecule with additional sites of oxidation and dehydrogenation. ACD Fragmenter™ confirmed around 70 percent of the major fragments automatically. The data suggests that the ketone group may be found at positions 21, 20, 10, and 11 on nefazodone’s aliphatic backbone. Figure 8: Structural assignment of the m/z 274 fragment from nefazodone. The ACD Fragmenter™ assigns the m/z 274 nefazodone fragment to the right half the molecule. This is consistent with oxidative dechlorination taking place on the left ring. Positions 29, 30, or 31 are the most likely sites for oxidation. Figure 7: XIC and MS/MS spectra of a nefazodone metabolite displaying of oxidative de-chlorination identified using LightSight® Software. Figure 9: Structural assignment of the m/z 246 fragment of nefazodone. The ACD Fragmenter™ assigns the m/z 246 nefazodone fragment to the right half the molecule. This is consistent with oxidative dechlorination taking place on the left ring. Position 29, 30, or 31 are the most likely sites for oxidation. Two novel metabolites that were displaying oxidative and dechlorination changes were detected by pMRM IDA. Figure 7 shows an example of the oxidative dechlorination metabolite found at 4.25 min in the LC run. The LIT MS/MS spectrum shows 14 diagnostic fragments at excellent sensitivity. Figures 8 and 9 contain the ACD Fragmenter assignments for this metabolite. 36 RUO-MKT-01-1583-A Conclusions • • • The QTRAP 5500 system has excellent sensitivity for both detection and confirmation of metabolites The pMRM method as well as dual scan precursor and neutral loss IDA methods, provide comprehensive detection of in vitro metabolites in a single injection. In most cases, a single injection using pMRM methods alone is more than adequate for a complete analysis. DRUG METABOLISM www.absciex.com • The 10- to 100-fold LIT MS/MS sensitivity improvement allows for superior detection of fragments that was not possible on less sensitive trap or TOF instruments. This greater degree of fragmentation allows for a high level of structural assignment. The scan rate is improved 4- or 5-fold over previous QTRAP systems, allowing for greater IDA coverage, even with sharp peaks delivered by the most progressive small particle high-pressure chromatography methods. www.absciex.com References 1 Peterman, S. M.; Duczak, N. J.; Kalgutkar, A. S.; Lame, M. E. ; Soglia, J. R. J Am Soc Mass Spectrom 2006; 17: 363. 2 Li, A. C.; Gohdes, M. A.; Shou, W. Z. Rapid Commun Mass Spectrom 2007; 21: 1421. 3 Li, A. C.; Shou, W. Z.; Mai, T. T.; Jiang, X.y. Rapid Commun Mass Spectrom 2007; 21: 4001. DRUG METABOLISM 37 EARLY DISCOVERY EARLY DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Figure 6: The fragmentation pattern of an N-demethylation metabolite of nefazodone. Fragmentation of a nefazodine metabolite with an N-dealkylation of the chlorinecontaining ring on the left half of the molecule with additional sites of oxidation and dehydrogenation. ACD Fragmenter™ confirmed around 70 percent of the major fragments automatically. The data suggests that the ketone group may be found at positions 21, 20, 10, and 11 on nefazodone’s aliphatic backbone. Figure 8: Structural assignment of the m/z 274 fragment from nefazodone. The ACD Fragmenter™ assigns the m/z 274 nefazodone fragment to the right half the molecule. This is consistent with oxidative dechlorination taking place on the left ring. Positions 29, 30, or 31 are the most likely sites for oxidation. Figure 7: XIC and MS/MS spectra of a nefazodone metabolite displaying of oxidative de-chlorination identified using LightSight® Software. Figure 9: Structural assignment of the m/z 246 fragment of nefazodone. The ACD Fragmenter™ assigns the m/z 246 nefazodone fragment to the right half the molecule. This is consistent with oxidative dechlorination taking place on the left ring. Position 29, 30, or 31 are the most likely sites for oxidation. Two novel metabolites that were displaying oxidative and dechlorination changes were detected by pMRM IDA. Figure 7 shows an example of the oxidative dechlorination metabolite found at 4.25 min in the LC run. The LIT MS/MS spectrum shows 14 diagnostic fragments at excellent sensitivity. Figures 8 and 9 contain the ACD Fragmenter assignments for this metabolite. 36 RUO-MKT-01-1583-A Conclusions • • • The QTRAP 5500 system has excellent sensitivity for both detection and confirmation of metabolites The pMRM method as well as dual scan precursor and neutral loss IDA methods, provide comprehensive detection of in vitro metabolites in a single injection. In most cases, a single injection using pMRM methods alone is more than adequate for a complete analysis. DRUG METABOLISM www.absciex.com • The 10- to 100-fold LIT MS/MS sensitivity improvement allows for superior detection of fragments that was not possible on less sensitive trap or TOF instruments. This greater degree of fragmentation allows for a high level of structural assignment. The scan rate is improved 4- or 5-fold over previous QTRAP systems, allowing for greater IDA coverage, even with sharp peaks delivered by the most progressive small particle high-pressure chromatography methods. www.absciex.com References 1 Peterman, S. M.; Duczak, N. J.; Kalgutkar, A. S.; Lame, M. E. ; Soglia, J. R. J Am Soc Mass Spectrom 2006; 17: 363. 2 Li, A. C.; Gohdes, M. A.; Shou, W. Z. Rapid Commun Mass Spectrom 2007; 21: 1421. 3 Li, A. C.; Shou, W. Z.; Mai, T. T.; Jiang, X.y. Rapid Commun Mass Spectrom 2007; 21: 4001. DRUG METABOLISM 37 Breakthrough Productivity for ADME Studies Using the AB SCIEX TripleTOF® 5600 System Speed, sensitivity, resolution, mass accuracy and linearity – together in one instrument for the first time Hesham Ghobarah, James Ferguson, and Suma Ramagiri AB SCIEX, Toronto, Ontario and Framingham, Massachusetts Key scientific challenges of ADME studies • • • Productivity – Increasing productivity in drug discovery and development continues to be a primary goal in pharmaceutical research. Separate quantitative and qualitative analysis – In the past, the optimal instrumentation for quantitative analysis was not suitable for qualitative analysis and vice versa. Typically, one ADME research group completed metabolite ID studies and another group with doing clearance studies. Analysis at non-relevant doses – For many compounds, ADME studies have been conducted at a higher than therapeutic concentration, occasionally leading to false conclusions due to concentrationdependent metabolic pathways for some compounds. • • Quant and qual in one run – Combining the best attributes of triple quadrupole systems (the workhorses of quantitative analysis) with a high-performance accurate mass analyzer into one instrument makes it possible to conduct both quantitative and qualitative analyses in one run. Triple-quad-like sensitivity – TripleTOF systems show quantitative sensitivity on par with the API 4000™ LC/MS/MS System, known industry-wide for its sensitivity and robustness. Speed – Most ADME labs are selecting UHPLC as the separation method of choice. UHPLC’s narrower chromatographic peaks result in higher concentrations at peak apices and, therefore, lower LOQs. To accommodate the UHPLC time scale, mass spectrometers must be able to perform the needed experiments on short cycle times. (Typical cycle times for the TripleTOF system are below 0.5 s). Key features of TripleTOF systems for ADME analyses • • • 38 Resolution and mass accuracy regardless of scan speed – Instruments employing orbital trapping require slower scanning to get higher resolution. The TripleTOF systems maintain approximately 30,000 resolution regardless of analysis speed. Calibration stability and linearity – Traditional time-of-flight (TOF) instruments typically require internal calibration to maintain mass accuracy resulting in lowered signals and non-linear quantitative response. The TripleTOF systems have been designed to overcome both of these limitations. Resolution and mass accuracy even at low m/z – Traditional TOF instruments provide good mass accuracy and resolution at high m/z. The high detector speed of the TripleTOF systems allows for sufficient resolution at low m/z, providing unambiguous formulae assigments using accurate mass, as well as the isotope and fragmention pattern. RUO-MKT-01-1583-A Due to their excellent sensitivity and high throughput, triple quadrupole instruments have been the workhorse instrument for the quantitative aspects of ADME analysis. Accurate mass instruments such as TOF and orbital trapping analyzers have been used for metabolite identification due to their high mass accuracy and resolution. Unfortunately, high-resolution orbital trapping approaches compromises speed at the expense of resolution. Traditional TOF technology has shown limitations in linearity and requires internal calibration to maintain mass accuracy. Modifying study designs, sample preparation procedures, or chromatography in order to accommodate for these instrument limitations is not acceptable in a highthroughput laboratory. Advanced technology enables true quant/qual without compromises Key benefits of the TripleTOF® 5600 System for ADME assays • compounds need to be analyzed weekly. Therefore, a generic data acquisition method that eliminates the need to optimize for individual compounds is highly desirable. • • Integrated software packages – To date, no manufacturer has provided one software package capable of finding metabolites, confirming the site of metabolism, tracking time course, experiments and doing interspecies comparisons. MetabolitePilot™ Software does all these tasks. Smarter acquisition – The automatic acquisition of MS/MS on low- level metabolites has been elusive before the introduction of real-time mass defect triggering enabled on the TripleTOF system. Now, the TripleTOF system can trigger on ions with mass defects related to the parent drug even at intensities lower than other compounds, allowing the proverbial needle to be pulled from its haystack. is highlighted for the AB SCIEX TripleTOF 5600 system. Modifications of existing assays are not necessary to realize the benefits of combined quant/qual features in a single experiment, a true breakthrough in productivity. Materials and methods Sample preparation Sample preparation. Six compounds (clomipramine, diclofenac, imipramine, haloperidol, verapamil, and midazolam) were incubated in rat liver microsomes at an initial substrate concentration of 1 µM in 96-well format. Standard highthroughput incubation conditions were used with time points of 0, 5, 15, 30, 60, and 120 min. Protein concentration was 1 mg/mL and NADPH was present at 4 mM. Reaction was quenched using an equal volume of acetonitrile and then diluted 1:1 with water prior to analysis. The final substrate concentration at t = 0 was 0.25 µM. EARLY DISCOVERY EARLY DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Chromatography The AB SCIEX TripleTOF 5600 system combines the best attributes of triple quadrupoles and accurate mass analyzers in a single instrument. The highly innovative design of the AB SCIEX TripleTOF 5600 system combines a proven high-performance triple quadrupole front-end with the Accelerator TOF™ analyzer, a state-of-the-art accurate mass analyzer with unprecedented performance and stability (Figure 1). This results in the linearity and sensitivity of a high performance triple quadrupole, combined with the speed, high mass accuracy (2 ppm or less), and high resolution (30,000) of an accurate mass instrument, even for lowmass for small molecules. System: Prominence UFLC-XR HPLC system Column: Phenomenex Synergi Polar-RP column 2 × 50 mm, 2.5 µm Injection volume: 10 μL Flow rate: 0.750 ml/min Mobile phase: A) Water/0.1% formic acid B) Acetonitrile/0.1% formic acid A generic gradient was used. In this technical note, a true quant/qual ADME application Introduction Metabolic stability experiments in microsomes are a classic example of in vitro studies that have traditionally been used for estimation of hepatic clearance. For compounds exhibiting rapid metabolism, additional qualitative studies are performed to identify the major metabolites in order to identify metabolic “soft spots.” This information aids the medicinal chemist in optimizing the structure of the unknown metabolite. However, performing a separate analysis for metabolite identification is time consuming. A major improvement in productivity can be achieved if both quantitative clearance and qualitative soft spot analysis can be performed in a single analytical run. A key requirement is to obtain accurate mass MS/MS spectra at high speed on a fast chromatographic time scale. In addition, due to the high- throughput nature of in vitro assays, hundreds of DRUG METABOLISM www.absciex.com Figure 1: AB SCIEX TripleTOF® 5600 System ion path. Proven front end technology coupled to a state of the art Accelerator TOF™ Analyzer results in unparalleled performance for both quantitative and qualitative analysis. www.absciex.com DRUG METABOLISM 39 Breakthrough Productivity for ADME Studies Using the AB SCIEX TripleTOF® 5600 System Speed, sensitivity, resolution, mass accuracy and linearity – together in one instrument for the first time Hesham Ghobarah, James Ferguson, and Suma Ramagiri AB SCIEX, Toronto, Ontario and Framingham, Massachusetts Key scientific challenges of ADME studies • • • Productivity – Increasing productivity in drug discovery and development continues to be a primary goal in pharmaceutical research. Separate quantitative and qualitative analysis – In the past, the optimal instrumentation for quantitative analysis was not suitable for qualitative analysis and vice versa. Typically, one ADME research group completed metabolite ID studies and another group with doing clearance studies. Analysis at non-relevant doses – For many compounds, ADME studies have been conducted at a higher than therapeutic concentration, occasionally leading to false conclusions due to concentrationdependent metabolic pathways for some compounds. • • Quant and qual in one run – Combining the best attributes of triple quadrupole systems (the workhorses of quantitative analysis) with a high-performance accurate mass analyzer into one instrument makes it possible to conduct both quantitative and qualitative analyses in one run. Triple-quad-like sensitivity – TripleTOF systems show quantitative sensitivity on par with the API 4000™ LC/MS/MS System, known industry-wide for its sensitivity and robustness. Speed – Most ADME labs are selecting UHPLC as the separation method of choice. UHPLC’s narrower chromatographic peaks result in higher concentrations at peak apices and, therefore, lower LOQs. To accommodate the UHPLC time scale, mass spectrometers must be able to perform the needed experiments on short cycle times. (Typical cycle times for the TripleTOF system are below 0.5 s). Key features of TripleTOF systems for ADME analyses • • • 38 Resolution and mass accuracy regardless of scan speed – Instruments employing orbital trapping require slower scanning to get higher resolution. The TripleTOF systems maintain approximately 30,000 resolution regardless of analysis speed. Calibration stability and linearity – Traditional time-of-flight (TOF) instruments typically require internal calibration to maintain mass accuracy resulting in lowered signals and non-linear quantitative response. The TripleTOF systems have been designed to overcome both of these limitations. Resolution and mass accuracy even at low m/z – Traditional TOF instruments provide good mass accuracy and resolution at high m/z. The high detector speed of the TripleTOF systems allows for sufficient resolution at low m/z, providing unambiguous formulae assigments using accurate mass, as well as the isotope and fragmention pattern. RUO-MKT-01-1583-A Due to their excellent sensitivity and high throughput, triple quadrupole instruments have been the workhorse instrument for the quantitative aspects of ADME analysis. Accurate mass instruments such as TOF and orbital trapping analyzers have been used for metabolite identification due to their high mass accuracy and resolution. Unfortunately, high-resolution orbital trapping approaches compromises speed at the expense of resolution. Traditional TOF technology has shown limitations in linearity and requires internal calibration to maintain mass accuracy. Modifying study designs, sample preparation procedures, or chromatography in order to accommodate for these instrument limitations is not acceptable in a highthroughput laboratory. Advanced technology enables true quant/qual without compromises Key benefits of the TripleTOF® 5600 System for ADME assays • compounds need to be analyzed weekly. Therefore, a generic data acquisition method that eliminates the need to optimize for individual compounds is highly desirable. • • Integrated software packages – To date, no manufacturer has provided one software package capable of finding metabolites, confirming the site of metabolism, tracking time course, experiments and doing interspecies comparisons. MetabolitePilot™ Software does all these tasks. Smarter acquisition – The automatic acquisition of MS/MS on low- level metabolites has been elusive before the introduction of real-time mass defect triggering enabled on the TripleTOF system. Now, the TripleTOF system can trigger on ions with mass defects related to the parent drug even at intensities lower than other compounds, allowing the proverbial needle to be pulled from its haystack. is highlighted for the AB SCIEX TripleTOF 5600 system. Modifications of existing assays are not necessary to realize the benefits of combined quant/qual features in a single experiment, a true breakthrough in productivity. Materials and methods Sample preparation Sample preparation. Six compounds (clomipramine, diclofenac, imipramine, haloperidol, verapamil, and midazolam) were incubated in rat liver microsomes at an initial substrate concentration of 1 µM in 96-well format. Standard highthroughput incubation conditions were used with time points of 0, 5, 15, 30, 60, and 120 min. Protein concentration was 1 mg/mL and NADPH was present at 4 mM. Reaction was quenched using an equal volume of acetonitrile and then diluted 1:1 with water prior to analysis. The final substrate concentration at t = 0 was 0.25 µM. EARLY DISCOVERY EARLY DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Chromatography The AB SCIEX TripleTOF 5600 system combines the best attributes of triple quadrupoles and accurate mass analyzers in a single instrument. The highly innovative design of the AB SCIEX TripleTOF 5600 system combines a proven high-performance triple quadrupole front-end with the Accelerator TOF™ analyzer, a state-of-the-art accurate mass analyzer with unprecedented performance and stability (Figure 1). This results in the linearity and sensitivity of a high performance triple quadrupole, combined with the speed, high mass accuracy (2 ppm or less), and high resolution (30,000) of an accurate mass instrument, even for lowmass for small molecules. System: Prominence UFLC-XR HPLC system Column: Phenomenex Synergi Polar-RP column 2 × 50 mm, 2.5 µm Injection volume: 10 μL Flow rate: 0.750 ml/min Mobile phase: A) Water/0.1% formic acid B) Acetonitrile/0.1% formic acid A generic gradient was used. In this technical note, a true quant/qual ADME application Introduction Metabolic stability experiments in microsomes are a classic example of in vitro studies that have traditionally been used for estimation of hepatic clearance. For compounds exhibiting rapid metabolism, additional qualitative studies are performed to identify the major metabolites in order to identify metabolic “soft spots.” This information aids the medicinal chemist in optimizing the structure of the unknown metabolite. However, performing a separate analysis for metabolite identification is time consuming. A major improvement in productivity can be achieved if both quantitative clearance and qualitative soft spot analysis can be performed in a single analytical run. A key requirement is to obtain accurate mass MS/MS spectra at high speed on a fast chromatographic time scale. In addition, due to the high- throughput nature of in vitro assays, hundreds of DRUG METABOLISM www.absciex.com Figure 1: AB SCIEX TripleTOF® 5600 System ion path. Proven front end technology coupled to a state of the art Accelerator TOF™ Analyzer results in unparalleled performance for both quantitative and qualitative analysis. www.absciex.com DRUG METABOLISM 39 Mass spectrometry A generic method was used for data acquisition on all compounds and samples. The method consisted of a TOF MS survey scan followed by two IDA TOF MS/MS scans. The mass range was m/z 100–1000 for both MS and MS/MS. An accumulation time of 100 ms was used for each scan. Dynamic background subtraction was applied for IDA criteria, and a collision energy of 35 eV with a spread of +/-10 eV was used for the MS/MS scans. External mass calibration was performed automatically using a calibrant delivery system. Data processing was performed using MultiQuant™ Software and MetabolitePilot Software (Figure 2). Results – sensitivity and speed MultiQuant Software was used to process the TOF MS data and generate all quantitative information. An XIC window of ±10mDa was used for all compounds. Peak areas were used to plot the % remaining relative to t = 0 (Figure 3), and MetabolitePilot Software was used to process the data for metabolites. Metabolic stability studies are best performed at a lower substrate concentration (1 µM), which is more physiologically relevant. Enough sensitivity is required to detect at least 1% of remaining parent molecule in order to obtain meaningful kinetic data. The TripleTOF 5600 system demonstrated excellent sensitivity and speed. For example, 0.1% of midazolam was easily detected (Figure 4). This represents a concentration of 0.25 nM or 0.8 pg on column. Sensitivity does not mean compromising on speed with a TripleTOF system. A minimum of eight points was obtained in IDA mode (with two dependent MS/MS scans) across a 4 sec peak while maintaining 30K resolution in both MS and MS/MS mode. In fact, each data point consisted of three scans (one TOF MS scan and two MS/MS scans) for a total of 24 experiments done in less than 4 sec. High resolution and mass accuracy No compromises Excellent mass accuracy and resolution were achieved in both MS and MS/MS scans (Figure 5). The high mass accuracy obtained in MS/MS scans greatly facilitates structure elucidation because it allows unambiguous assignment of elemental composition. Successful application of quant/qual technology to in vitro ADME was demonstrated using the TripleTOF 5600 system. No compromises were made in resolution in order to achieve speed or sensitivity. A relatively low substrate concentration of 1 µm was successfully used. Using MetabolitePilot Software, data from multiple samples and compounds was processed unattended in batch mode. The software reported metabolites detected and elemental compositions in a concise and powerful, yet user-friendly, interface (Figure 2). Standard incubation and HPLC conditions were used with no need to make any modifications in order to gain the benefits of quant/qual. EARLY DISCOVERY EARLY DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Excellent separation, metabolite coverage, and speed were demonstrated using a generic IDA method (Figure 6). Figure 5: Mass accuracy and resolution in TOF MS and MS/MS Modes. The MS scan of haloperidol parent from the 5 min time point shows excellent accuracy and resolution (left panel) with a near perfect match of isotope pattern (inset). The product ion spectrum of hydroxy midazolam from the 5 min time point acquired with IDA is shown (right panel). Excellent mass accuracy and 30K resolution allow easy assignment of elemental composition, even to the product ions. Figure 2: Easy data processing in MetabolitePilot™ Software. Using this nextgeneration software simplifies accurate mass metabolite identification with its automated data processing in batch mode. Figure 4: Sensitivity and Speed. The 30 minutes time point from Midazolam microsome incubation with 0.1% of parent remaining (0.8 pg on column). Analyte is easily detected in TOF MS IDA mode with 8 data points. Each timepoint consists of a survey scan and two MS/MS dependent scans. Figure 3: Data Processing in MultiQuant™ Software. (a) TOF MS data was easily processed for multiple analytes to obtain quantitative data. (b) Metabolic stability profiles of 6 common substrates incubated at 1 µM and analyzed using TOF MS with IDA triggered MS/MS. 40 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com Figure 6: Broad coverage for phase I metabolites. Overlay of accurate mass XICs for imipramine incubation at 5 min. A wide range of imipramine phase I metabolites were easily detected using a completely generic TOF MS IDA method in under 2.5 min. www.absciex.com DRUG METABOLISM 41 Mass spectrometry A generic method was used for data acquisition on all compounds and samples. The method consisted of a TOF MS survey scan followed by two IDA TOF MS/MS scans. The mass range was m/z 100–1000 for both MS and MS/MS. An accumulation time of 100 ms was used for each scan. Dynamic background subtraction was applied for IDA criteria, and a collision energy of 35 eV with a spread of +/-10 eV was used for the MS/MS scans. External mass calibration was performed automatically using a calibrant delivery system. Data processing was performed using MultiQuant™ Software and MetabolitePilot Software (Figure 2). Results – sensitivity and speed MultiQuant Software was used to process the TOF MS data and generate all quantitative information. An XIC window of ±10mDa was used for all compounds. Peak areas were used to plot the % remaining relative to t = 0 (Figure 3), and MetabolitePilot Software was used to process the data for metabolites. Metabolic stability studies are best performed at a lower substrate concentration (1 µM), which is more physiologically relevant. Enough sensitivity is required to detect at least 1% of remaining parent molecule in order to obtain meaningful kinetic data. The TripleTOF 5600 system demonstrated excellent sensitivity and speed. For example, 0.1% of midazolam was easily detected (Figure 4). This represents a concentration of 0.25 nM or 0.8 pg on column. Sensitivity does not mean compromising on speed with a TripleTOF system. A minimum of eight points was obtained in IDA mode (with two dependent MS/MS scans) across a 4 sec peak while maintaining 30K resolution in both MS and MS/MS mode. In fact, each data point consisted of three scans (one TOF MS scan and two MS/MS scans) for a total of 24 experiments done in less than 4 sec. High resolution and mass accuracy No compromises Excellent mass accuracy and resolution were achieved in both MS and MS/MS scans (Figure 5). The high mass accuracy obtained in MS/MS scans greatly facilitates structure elucidation because it allows unambiguous assignment of elemental composition. Successful application of quant/qual technology to in vitro ADME was demonstrated using the TripleTOF 5600 system. No compromises were made in resolution in order to achieve speed or sensitivity. A relatively low substrate concentration of 1 µm was successfully used. Using MetabolitePilot Software, data from multiple samples and compounds was processed unattended in batch mode. The software reported metabolites detected and elemental compositions in a concise and powerful, yet user-friendly, interface (Figure 2). Standard incubation and HPLC conditions were used with no need to make any modifications in order to gain the benefits of quant/qual. EARLY DISCOVERY EARLY DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Excellent separation, metabolite coverage, and speed were demonstrated using a generic IDA method (Figure 6). Figure 5: Mass accuracy and resolution in TOF MS and MS/MS Modes. The MS scan of haloperidol parent from the 5 min time point shows excellent accuracy and resolution (left panel) with a near perfect match of isotope pattern (inset). The product ion spectrum of hydroxy midazolam from the 5 min time point acquired with IDA is shown (right panel). Excellent mass accuracy and 30K resolution allow easy assignment of elemental composition, even to the product ions. Figure 2: Easy data processing in MetabolitePilot™ Software. Using this nextgeneration software simplifies accurate mass metabolite identification with its automated data processing in batch mode. Figure 4: Sensitivity and Speed. The 30 minutes time point from Midazolam microsome incubation with 0.1% of parent remaining (0.8 pg on column). Analyte is easily detected in TOF MS IDA mode with 8 data points. Each timepoint consists of a survey scan and two MS/MS dependent scans. Figure 3: Data Processing in MultiQuant™ Software. (a) TOF MS data was easily processed for multiple analytes to obtain quantitative data. (b) Metabolic stability profiles of 6 common substrates incubated at 1 µM and analyzed using TOF MS with IDA triggered MS/MS. 40 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com Figure 6: Broad coverage for phase I metabolites. Overlay of accurate mass XICs for imipramine incubation at 5 min. A wide range of imipramine phase I metabolites were easily detected using a completely generic TOF MS IDA method in under 2.5 min. www.absciex.com DRUG METABOLISM 41 Conclusions • • • • EARLY DISCOVERY EARLY DISCOVERY For Research Use Only. Not for use in diagnostic procedures. TripleTOF 5600 system is the first accurate mass instrument to offer quantitative performance comparable to a high performance triple quadrupole. The combination of speed, sensitivity, mass accuracy, and resolution enables routine quant/qual analysis. For metabolic stability, the ability to perform parent quantification, metabolite detection, and accurate mass information all in a single run. MultiQuant and MetabolitePilot Software are powerful tools for efficient analysis of quantitative and qualitative data. With proven front-end technology and high performance, the TripleTOF 5600 system fits well with existing high-throughput sample preparation and fast chromatography. Acknowledgements The authors would like to thank Dr. Christie Hunter and Tanya Gamble for their invaluable contributions and review. 42 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 43 Conclusions • • • • EARLY DISCOVERY EARLY DISCOVERY For Research Use Only. Not for use in diagnostic procedures. TripleTOF 5600 system is the first accurate mass instrument to offer quantitative performance comparable to a high performance triple quadrupole. The combination of speed, sensitivity, mass accuracy, and resolution enables routine quant/qual analysis. For metabolic stability, the ability to perform parent quantification, metabolite detection, and accurate mass information all in a single run. MultiQuant and MetabolitePilot Software are powerful tools for efficient analysis of quantitative and qualitative data. With proven front-end technology and high performance, the TripleTOF 5600 system fits well with existing high-throughput sample preparation and fast chromatography. Acknowledgements The authors would like to thank Dr. Christie Hunter and Tanya Gamble for their invaluable contributions and review. 42 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 43 Rapid Metabolite Identification Using MetabolitePilot™ Software and the TripleTOF® 5600 System Easily find, identify, and confirm metabolites in accurate mass data Carmai Seto, Tanya Gamble, and Hesham Ghobarah AB SCIEX, Concord, Ontario, Canada Key scientific challenges of metabolite ID workflows • • • High-performance, integrated software – In the past, the work of finding, confirming, and quantifying metabolites has required the use of multiple software packages. MetabolitePilot™ Software can perform all three functions in one software package. Complicated data correlation – Typically after processing quantitative data another software package must be used to correlate data, (e.g., interspecies comparisons, lot-to-lot variations, time course studies). Structural elucidation – The ability to match fragments to proposed structures speeds the structural elucidation process, providing critical “soft-spot” information to the medicinal chemist in a timely manner. based on the structure of the parent compound. It also automatically calculates mass-defect filters for phase I metabolites, including the parent drug, as well as phase II and potential cleavage metabolites (Figure 1). MetabolitePilot Software makes the work of identifying and confirming metabolites more efficient with an easy-to-use, integrated workspace that displays all relevant information. The software displays the analog, MS, and MS/MS data associated with each metabolite in a single view (Figure 2). A confirmation score is also provided to facilitiate metabolite peak detection (Figure 3). To aid with the confirmation of metabolite peak identification, the software enables faster comparison of metabolite and parent product ion spectra with a simple and effective display. The product ion spectrum of the parent drug is automatically compared with each metabolite, and the results of the comparison are displayed in the MS/MS panel of the results workspace. Materials and methods Sample preparation Bromocriptine was incubated in rat liver microsomes under oxidative conditions at 37° C for 1 hr, along with a control. The reactions were quenched by the addition of an equal volume of acetonitrile to the microsomal solutions and then centrifuged at 14,000 rpm for 5 min. Chromatography The incubations were analyzed using a Shimadzu Prominence UFLC-XR LC system with UV detector set at 302 nm. All analyses were performed using an Imtakt Cadenza CD-C18 (2×50 mm) 3µm, ODS column that was held at 40° C. Solvent A contained water with 0.1% formic acid and solvent B contained acetonitrile with 0.1% formic acid, and the flow rate was set at 600 µL/min. The gradient is summarized in Table 1. Gradient:Time Gradient (min) composition (%B) Key benefits of MetabolitePilot Software • • • • Speed – Data can be batch-processed, saving time during analysis. A comprehensive software package – MetabolitePilot Software finds metabolites, speeds confirmation the site of metabolism with fragmentation interpretation tools, and correlates data across userdefined domains, with multiple options for visualizing the data. Structure-driven processing – Processing parameters are based on the structure of the parent drug and can be rapidly and easily built. Correlation with analog (UV, DAD, or radioactivity) data – Peak areas from both mass spectrometry and analog data are both provided. Key features of MetabolitePilot Software • • • • Multiple methods for finding metabolites – Data can be mined for expected and unexpected metabolites (unpredicted chromatographic peaks), as well as by mass defect, isotope patterns and similarity of the product ion spectra with that of the parent drug. Data comparisons against up to five controls – For example, during GSH screenings, the software encorporates a t0 control, a second control without the test article, a third control without GSH, and another control without NADPH. Integrated MS/MS fragment interpretation and structure editing – These features speed the determination of the site of metabolism. Weighted scoring – Scoring may be customized and is based on mass accuracy, isotopic pattern fidelity, similarity and quality of the MS/MS data to that of the parent drug, and mass-defect matching. Introduction The ability to find, identify, and confirm metabolites as quickly as possible is critical at multiple stages of drug discovery and development. Advances in instrumentation have enabled the generation of very information-rich raw data. New software 44 RUO-MKT-01-1583-A LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. programs that automate portions of the metabolite identification workflow provide for considerable benefits to drug metabolism researchers. For example, accurate mass instruments such as the time-of-flight (TOF) MS have the capability to provide both qualitative and quantitative information in a single analysis. However, the data analysis can be quite complicated and very time consuming if performed manually, especially if analog data was acquired along with MS and MS/MS data. Software programs can automate the process of finding, identifying, and confirming metabolites in accurate mass data. This technical note describes how MetabolitePilot Software and the AB SCIEX TripleTOF® 5600 System can make the process of identifying and confirming metabolites more efficient than ever before. Figure 1: Multiple mass-defect filtering setup in the processing parameters. MetabolitePilot™ Software automatically calculates the mass-defect filters for multiple metabolite classes using the parent structure. DRUG METABOLISM www.absciex.com 10 0.5 10 0.75 20 5.5 40 6.5 60 6.8 90 6.9 90 7 10 Table 1: LC gradient conditions. Mass spectrometry The TripleTOF 5600 system method used for data acquisition consisted of a TOF MS survey scan followed by two IDA TOF MS/ MS scans. Mass Defect triggered IDA with Dynamic Background Subtraction™ algorithm was used to collect the MS/MS data. External mass calibration was performed automatically using a calibrant delivery system. MetabolitePilot Software: Automated identification and confirmation of metabolites MetabolitePilot Software employs multiple, powerful data processing algorithms to perform both non-targeted and targeted processing in parallel. In addition, generic peak-finding, multiple mass-defect filtering, isotope-pattern matching, and metabolite detection based on common product ions or neutral losses can all be used simultaneously to find and identify metabolites efficiently. To harness the power of these algorithms, the software automatically calculates the most appropriate compound-specific processing parameters for a given compound. MetabolitePilot Software automatically predicts potential cleavage metabolites 0 Figure 2: Results workspace of MetabolitePilot™ Software. All data is displayed in a single workspace for efficient data review. www.absciex.com DRUG METABOLISM 45 Rapid Metabolite Identification Using MetabolitePilot™ Software and the TripleTOF® 5600 System Easily find, identify, and confirm metabolites in accurate mass data Carmai Seto, Tanya Gamble, and Hesham Ghobarah AB SCIEX, Concord, Ontario, Canada Key scientific challenges of metabolite ID workflows • • • High-performance, integrated software – In the past, the work of finding, confirming, and quantifying metabolites has required the use of multiple software packages. MetabolitePilot™ Software can perform all three functions in one software package. Complicated data correlation – Typically after processing quantitative data another software package must be used to correlate data, (e.g., interspecies comparisons, lot-to-lot variations, time course studies). Structural elucidation – The ability to match fragments to proposed structures speeds the structural elucidation process, providing critical “soft-spot” information to the medicinal chemist in a timely manner. based on the structure of the parent compound. It also automatically calculates mass-defect filters for phase I metabolites, including the parent drug, as well as phase II and potential cleavage metabolites (Figure 1). MetabolitePilot Software makes the work of identifying and confirming metabolites more efficient with an easy-to-use, integrated workspace that displays all relevant information. The software displays the analog, MS, and MS/MS data associated with each metabolite in a single view (Figure 2). A confirmation score is also provided to facilitiate metabolite peak detection (Figure 3). To aid with the confirmation of metabolite peak identification, the software enables faster comparison of metabolite and parent product ion spectra with a simple and effective display. The product ion spectrum of the parent drug is automatically compared with each metabolite, and the results of the comparison are displayed in the MS/MS panel of the results workspace. Materials and methods Sample preparation Bromocriptine was incubated in rat liver microsomes under oxidative conditions at 37° C for 1 hr, along with a control. The reactions were quenched by the addition of an equal volume of acetonitrile to the microsomal solutions and then centrifuged at 14,000 rpm for 5 min. Chromatography The incubations were analyzed using a Shimadzu Prominence UFLC-XR LC system with UV detector set at 302 nm. All analyses were performed using an Imtakt Cadenza CD-C18 (2×50 mm) 3µm, ODS column that was held at 40° C. Solvent A contained water with 0.1% formic acid and solvent B contained acetonitrile with 0.1% formic acid, and the flow rate was set at 600 µL/min. The gradient is summarized in Table 1. Gradient:Time Gradient (min) composition (%B) Key benefits of MetabolitePilot Software • • • • Speed – Data can be batch-processed, saving time during analysis. A comprehensive software package – MetabolitePilot Software finds metabolites, speeds confirmation the site of metabolism with fragmentation interpretation tools, and correlates data across userdefined domains, with multiple options for visualizing the data. Structure-driven processing – Processing parameters are based on the structure of the parent drug and can be rapidly and easily built. Correlation with analog (UV, DAD, or radioactivity) data – Peak areas from both mass spectrometry and analog data are both provided. Key features of MetabolitePilot Software • • • • Multiple methods for finding metabolites – Data can be mined for expected and unexpected metabolites (unpredicted chromatographic peaks), as well as by mass defect, isotope patterns and similarity of the product ion spectra with that of the parent drug. Data comparisons against up to five controls – For example, during GSH screenings, the software encorporates a t0 control, a second control without the test article, a third control without GSH, and another control without NADPH. Integrated MS/MS fragment interpretation and structure editing – These features speed the determination of the site of metabolism. Weighted scoring – Scoring may be customized and is based on mass accuracy, isotopic pattern fidelity, similarity and quality of the MS/MS data to that of the parent drug, and mass-defect matching. Introduction The ability to find, identify, and confirm metabolites as quickly as possible is critical at multiple stages of drug discovery and development. Advances in instrumentation have enabled the generation of very information-rich raw data. New software 44 RUO-MKT-01-1583-A LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. programs that automate portions of the metabolite identification workflow provide for considerable benefits to drug metabolism researchers. For example, accurate mass instruments such as the time-of-flight (TOF) MS have the capability to provide both qualitative and quantitative information in a single analysis. However, the data analysis can be quite complicated and very time consuming if performed manually, especially if analog data was acquired along with MS and MS/MS data. Software programs can automate the process of finding, identifying, and confirming metabolites in accurate mass data. This technical note describes how MetabolitePilot Software and the AB SCIEX TripleTOF® 5600 System can make the process of identifying and confirming metabolites more efficient than ever before. Figure 1: Multiple mass-defect filtering setup in the processing parameters. MetabolitePilot™ Software automatically calculates the mass-defect filters for multiple metabolite classes using the parent structure. DRUG METABOLISM www.absciex.com 10 0.5 10 0.75 20 5.5 40 6.5 60 6.8 90 6.9 90 7 10 Table 1: LC gradient conditions. Mass spectrometry The TripleTOF 5600 system method used for data acquisition consisted of a TOF MS survey scan followed by two IDA TOF MS/ MS scans. Mass Defect triggered IDA with Dynamic Background Subtraction™ algorithm was used to collect the MS/MS data. External mass calibration was performed automatically using a calibrant delivery system. MetabolitePilot Software: Automated identification and confirmation of metabolites MetabolitePilot Software employs multiple, powerful data processing algorithms to perform both non-targeted and targeted processing in parallel. In addition, generic peak-finding, multiple mass-defect filtering, isotope-pattern matching, and metabolite detection based on common product ions or neutral losses can all be used simultaneously to find and identify metabolites efficiently. To harness the power of these algorithms, the software automatically calculates the most appropriate compound-specific processing parameters for a given compound. MetabolitePilot Software automatically predicts potential cleavage metabolites 0 Figure 2: Results workspace of MetabolitePilot™ Software. All data is displayed in a single workspace for efficient data review. www.absciex.com DRUG METABOLISM 45 For Research Use Only. Not for use in diagnostic procedures. LATE STAGE DISCOVERY Along with the interpreted spectrum of bromocriptine, structural elucidation of the metabolites was quickly achieved using the excellent mass accuracies achieved by the TripleTOF 5600 system. When comparing product ion spectra, common product ions between metabolites and parent drug (bromocriptine) indicate where metabolism has not occurred, while common neutral losses indicate where metabolism has occurred. The comparison process is made more efficient by the automated product ion comparisons and fully integrated, automatic MS/MS fragment interpretation and assignment performed by MetabolitePilot Software. Editing and storage of metabolite structures can be performed directly within the software. Interpretation and inter-sample correlation capabilities are covered in detail in reference 3. • An example of the product ion spectra used for structural elucidation in this study is shown in Figure 5; the spectrum for an oxidation metabolite (M6), a desaturation metabolite (M23) and bromocriptine are shown. Due to the presence of the common product ion, m/z 426.0809, the sites of metabolites were determined not to be localized on the substructure represented by m/z 426.0809. There is no ambiguity in the elemental composition of the product ion due to the mass accuracies achieved by the TripleTOF 5600 system for this experiment. • Conclusions • • The TripleTOF 5600 system and MetabolitePilot Software effectively address the data analysis bottleneck in metabolite identification, from peak detection to structural elucidation. Efficiently find metabolites using powerful, multiple, peak-finding strategies applied in parallel, such as non-targeted, generic peak-finding and mass-defect filtering. • • Automatic generation of structure-based processing parameters such as multiple mass-defect ranges and potential cleavage metabolites eases metabolite structural assignments. Efficient processing of multiple sample sets together occurs in the Batch Workspace. Structural elucidation is rapidly performed using integrated MS/MS fragment interpretation and assignment, all in a single, user-friendly workspace. Automatic correlation of UV, MS, and MS/MS data for multiple samples and plotting of response across samples enables faster and easier evaluation of the time course studies and inter-species comparison. References 1 “Breakthrough Productivity for ADME Studies Using the AB SCIEX TripleTOF® 5600 System.” AB SCIEX Technical Note. Publication 0480110-01. 2 “Simultaneous Pharmacokinetic Profiling and Automated Metabolite Identification Using the AB SCIEX TripleTOF® 5600 System and MetabolitePilot™ Software.” AB SCIEX Technical Note. Publication 1270210-01. 3 “Solving Bottlenecks in Metabolite ID Data Analysis With MetabolitePilot™ Software”, AB SCIEX Technical Note, Publication 3610211-01 4 To download a trial version of MetabolitePilot™ Software, please visit: http://www.absciex.com/ Products/Software/MetabolitePilot-Software LATE STAGE DISCOVERY The identified peaks were confirmed as metabolites using the confirmation score provided by the software, the exact mass product ion spectra and the UV data. Figure 3: Detailed information about a confirmation score. Each potential metabolite is given a confirmation score to help determine if the peak is likely to be a metabolite. Results MetabolitePilot Software was used to process the data acquired from the incubation. In the results workspace, the MS and UV data were correlated, with the potential metabolites, indicating a corresponding analog peak for any MS peaks (Figure 2). Figure 4 illustrates the structures proposed for some of the metabolites that were identified by the software. Figure 4: Proposed structures for some bromocriptine metabolites identified in a rat microsomal incubation. 46 RUO-MKT-01-1583-A Figure 5: Product ion spectra of two metabolites and bromocriptine. Automated MS/MS comparisons and excellent mass accuracies allowed for the rapid structural elucidation of metabolites. DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 47 For Research Use Only. Not for use in diagnostic procedures. LATE STAGE DISCOVERY Along with the interpreted spectrum of bromocriptine, structural elucidation of the metabolites was quickly achieved using the excellent mass accuracies achieved by the TripleTOF 5600 system. When comparing product ion spectra, common product ions between metabolites and parent drug (bromocriptine) indicate where metabolism has not occurred, while common neutral losses indicate where metabolism has occurred. The comparison process is made more efficient by the automated product ion comparisons and fully integrated, automatic MS/MS fragment interpretation and assignment performed by MetabolitePilot Software. Editing and storage of metabolite structures can be performed directly within the software. Interpretation and inter-sample correlation capabilities are covered in detail in reference 3. • An example of the product ion spectra used for structural elucidation in this study is shown in Figure 5; the spectrum for an oxidation metabolite (M6), a desaturation metabolite (M23) and bromocriptine are shown. Due to the presence of the common product ion, m/z 426.0809, the sites of metabolites were determined not to be localized on the substructure represented by m/z 426.0809. There is no ambiguity in the elemental composition of the product ion due to the mass accuracies achieved by the TripleTOF 5600 system for this experiment. • Conclusions • • The TripleTOF 5600 system and MetabolitePilot Software effectively address the data analysis bottleneck in metabolite identification, from peak detection to structural elucidation. Efficiently find metabolites using powerful, multiple, peak-finding strategies applied in parallel, such as non-targeted, generic peak-finding and mass-defect filtering. • • Automatic generation of structure-based processing parameters such as multiple mass-defect ranges and potential cleavage metabolites eases metabolite structural assignments. Efficient processing of multiple sample sets together occurs in the Batch Workspace. Structural elucidation is rapidly performed using integrated MS/MS fragment interpretation and assignment, all in a single, user-friendly workspace. Automatic correlation of UV, MS, and MS/MS data for multiple samples and plotting of response across samples enables faster and easier evaluation of the time course studies and inter-species comparison. References 1 “Breakthrough Productivity for ADME Studies Using the AB SCIEX TripleTOF® 5600 System.” AB SCIEX Technical Note. Publication 0480110-01. 2 “Simultaneous Pharmacokinetic Profiling and Automated Metabolite Identification Using the AB SCIEX TripleTOF® 5600 System and MetabolitePilot™ Software.” AB SCIEX Technical Note. Publication 1270210-01. 3 “Solving Bottlenecks in Metabolite ID Data Analysis With MetabolitePilot™ Software”, AB SCIEX Technical Note, Publication 3610211-01 4 To download a trial version of MetabolitePilot™ Software, please visit: http://www.absciex.com/ Products/Software/MetabolitePilot-Software LATE STAGE DISCOVERY The identified peaks were confirmed as metabolites using the confirmation score provided by the software, the exact mass product ion spectra and the UV data. Figure 3: Detailed information about a confirmation score. Each potential metabolite is given a confirmation score to help determine if the peak is likely to be a metabolite. Results MetabolitePilot Software was used to process the data acquired from the incubation. In the results workspace, the MS and UV data were correlated, with the potential metabolites, indicating a corresponding analog peak for any MS peaks (Figure 2). Figure 4 illustrates the structures proposed for some of the metabolites that were identified by the software. Figure 4: Proposed structures for some bromocriptine metabolites identified in a rat microsomal incubation. 46 RUO-MKT-01-1583-A Figure 5: Product ion spectra of two metabolites and bromocriptine. Automated MS/MS comparisons and excellent mass accuracies allowed for the rapid structural elucidation of metabolites. DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 47 In Vivo Metabolic Profiling of Carbamazepine Using the QTRAP® 5500 System and LightSight® Software 2.2 Key features of the QTRAP 5500® System and LightSight Software 2.2 for in vivo metabolitic profiling • • Tanya Gamble 1, Henrianna Pang 2, Sophie Pang 2, Yingbo Yang 2, William Cui 2, Julia Izhakova 2, Douglas Turk 2, and Hesham Ghobarah 1 1 AB SCIEX, Concord, Ontario; 2NoAb BioDiscoveries Inc., Mississauga, Ontario, Canada • Key scientific challenges of in vivo metabolitic profiling • • Matrix interferences – Extracting or finding metabolites in complex biological matrices used for in vivo metabolic studies often requires both sensitive and specific scan types. Speed – High-throughput analyses, driven by the desire for efficient data collection and reporting, enables rapid detection and identification of metabolites in drug discovery laboratories. Key benefits of the QTRAP® 5500 System and LightSight Software 2.2 for in vivo metabolitic profiling • • • Ease-of-use – LightSight Software can be used to process qualitative and quantitative data simultaneously. High sensitivity – The high sensitivity of the QTRAP 5500 system allows for metabolic profiling at physiologically relevant dosing. High specificity – The unique scanning functions of the QTRAP 5500 system combine high specificity and sensitivity for powerful metabolite ID workflows. LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. TripleTrap™ Scanning – These scanning functions unique to the QTRAP systems combine highly specific triple quadrupole scans with very fast and sensitive linear ion trap (LIT) scans for powerful metabolite ID workflows. An integrated software package – Quantitative and qualitative data are easily and automatically generated using LightSight Software. (Figure 1 shows processed data from a dual survey acquisition method.) LightSight Software will also build acquisition methods and acquire data. Sensitivity – The high sensitivity of the QTRAP 5500 system allows it to find more metabolites than previous systems. Overview This application note demonstrates the use of ultra-high pressure liquid chromatography (UHPLC) with fast-scanning mass spectrometric techniques for the identification of carbamazepine and its metabolites in rat plasma. The unique, fast-scanning capabilities of the QTRAP 5500 system couples highly specific triple quadrupole scan modes with highly sensitive LIT product ion scans for the rapid detection and identification of metabolites. LightSight Software 2.2 was employed to create and submit acquisition methods. All data processing was performed using a single processing method in LightSight Software. Figure 2: Experimental conditions with UHPLC gradient. Introduction Traditionally, in vitro and in vivo metabolic profiling has been performed at high, non-clinically relevant doses due to the sensitivity and selectivity limitiations of mass spectrometric detection. A metabolic profile at these high doses can be significantly different from the profile at physiologically relevant concentrations. It is highly desirable to perform in vivo studies at clinically relevant dose levels; however, these studies are often performed at such a low concentration that significant analytical challenges are posed during qualitative analysis. For this study, researchers evaluated the use of UPLC coupled to a hybrid triple quadrupole linear ion trap mass spectrometer for metabolic profiling of carbamazepine at physiologically relevant concentrations. Experimental conditions Carbamazepine was administered intravenously to SpragueDawley rats at 4 mg/kg dose. Plasma samples were collected prior to dosing and at 15, 60, 180, and 360 min and stored at -20° C prior to analysis. Each sample was prepared using simple protein precipitation with 50/50 methanol/acetonitrile, centrifuged, dried down, and reconstituted in 95/5/0.1 water/acetonitrile/ formic acid. Figure 1: Data processed using LightSight® Software. Dual survey data from a precursor ion and neutral loss information-dependent acquisition (IDA) method processed in LightSight Software 2.2. 48 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com Carbamazepine and its metabolites were chromatographically separated using an Acquity UPLC system. Mass spectrometric detection was performed using a QTRAP 5500 system using the conditions shown in Figure 2. This unique hybrid triple www.absciex.com quadrupole/linear ion trap instrument couples the ability to use highly specific triple quadrupole scan modes such as precursor ion (Prec) or neutral loss (NL) scanning to trigger highly sensitive linear ion trap product ion scans using information-dependent acquisition (IDA). The acquisition method wizard in LightSight Software 2.2 (Figure 3) was used to create all methods, including a predictive MRM (pMRM) approach and a method that combines a neutral loss and precursor ion scan to trigger MS/MS. Results and discussion Figure 4 shows the molecular structure of carbamazepine with fragmentation sites incurred during MS/MS analysis indicated along with the nominal mass of the fragment or neutral loss mass (NL). The m/z 194 fragment ion was chosen as the Q3 mass for the pMRM method, as well as the fragment for the precursor/ neutral loss method. A two-injection strategy was used to find the majority of the metabolites. The first injection used the pMRM IDA method and was able to detect 18 potential metabolites. The dual precursor ion/neutral loss IDA method ensured that unpredicted metabolites were not missed. Screens for sulfate and glucuronide conjugation were completed using a neutral loss scan m/z of 80 and 176 in a third injection. In total, 20 potential metabolites were identified with information-rich MS/MS spectra collected using the highly sensitive ion trap. DRUG METABOLISM 49 In Vivo Metabolic Profiling of Carbamazepine Using the QTRAP® 5500 System and LightSight® Software 2.2 Key features of the QTRAP 5500® System and LightSight Software 2.2 for in vivo metabolitic profiling • • Tanya Gamble 1, Henrianna Pang 2, Sophie Pang 2, Yingbo Yang 2, William Cui 2, Julia Izhakova 2, Douglas Turk 2, and Hesham Ghobarah 1 1 AB SCIEX, Concord, Ontario; 2NoAb BioDiscoveries Inc., Mississauga, Ontario, Canada • Key scientific challenges of in vivo metabolitic profiling • • Matrix interferences – Extracting or finding metabolites in complex biological matrices used for in vivo metabolic studies often requires both sensitive and specific scan types. Speed – High-throughput analyses, driven by the desire for efficient data collection and reporting, enables rapid detection and identification of metabolites in drug discovery laboratories. Key benefits of the QTRAP® 5500 System and LightSight Software 2.2 for in vivo metabolitic profiling • • • Ease-of-use – LightSight Software can be used to process qualitative and quantitative data simultaneously. High sensitivity – The high sensitivity of the QTRAP 5500 system allows for metabolic profiling at physiologically relevant dosing. High specificity – The unique scanning functions of the QTRAP 5500 system combine high specificity and sensitivity for powerful metabolite ID workflows. LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. TripleTrap™ Scanning – These scanning functions unique to the QTRAP systems combine highly specific triple quadrupole scans with very fast and sensitive linear ion trap (LIT) scans for powerful metabolite ID workflows. An integrated software package – Quantitative and qualitative data are easily and automatically generated using LightSight Software. (Figure 1 shows processed data from a dual survey acquisition method.) LightSight Software will also build acquisition methods and acquire data. Sensitivity – The high sensitivity of the QTRAP 5500 system allows it to find more metabolites than previous systems. Overview This application note demonstrates the use of ultra-high pressure liquid chromatography (UHPLC) with fast-scanning mass spectrometric techniques for the identification of carbamazepine and its metabolites in rat plasma. The unique, fast-scanning capabilities of the QTRAP 5500 system couples highly specific triple quadrupole scan modes with highly sensitive LIT product ion scans for the rapid detection and identification of metabolites. LightSight Software 2.2 was employed to create and submit acquisition methods. All data processing was performed using a single processing method in LightSight Software. Figure 2: Experimental conditions with UHPLC gradient. Introduction Traditionally, in vitro and in vivo metabolic profiling has been performed at high, non-clinically relevant doses due to the sensitivity and selectivity limitiations of mass spectrometric detection. A metabolic profile at these high doses can be significantly different from the profile at physiologically relevant concentrations. It is highly desirable to perform in vivo studies at clinically relevant dose levels; however, these studies are often performed at such a low concentration that significant analytical challenges are posed during qualitative analysis. For this study, researchers evaluated the use of UPLC coupled to a hybrid triple quadrupole linear ion trap mass spectrometer for metabolic profiling of carbamazepine at physiologically relevant concentrations. Experimental conditions Carbamazepine was administered intravenously to SpragueDawley rats at 4 mg/kg dose. Plasma samples were collected prior to dosing and at 15, 60, 180, and 360 min and stored at -20° C prior to analysis. Each sample was prepared using simple protein precipitation with 50/50 methanol/acetonitrile, centrifuged, dried down, and reconstituted in 95/5/0.1 water/acetonitrile/ formic acid. Figure 1: Data processed using LightSight® Software. Dual survey data from a precursor ion and neutral loss information-dependent acquisition (IDA) method processed in LightSight Software 2.2. 48 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com Carbamazepine and its metabolites were chromatographically separated using an Acquity UPLC system. Mass spectrometric detection was performed using a QTRAP 5500 system using the conditions shown in Figure 2. This unique hybrid triple www.absciex.com quadrupole/linear ion trap instrument couples the ability to use highly specific triple quadrupole scan modes such as precursor ion (Prec) or neutral loss (NL) scanning to trigger highly sensitive linear ion trap product ion scans using information-dependent acquisition (IDA). The acquisition method wizard in LightSight Software 2.2 (Figure 3) was used to create all methods, including a predictive MRM (pMRM) approach and a method that combines a neutral loss and precursor ion scan to trigger MS/MS. Results and discussion Figure 4 shows the molecular structure of carbamazepine with fragmentation sites incurred during MS/MS analysis indicated along with the nominal mass of the fragment or neutral loss mass (NL). The m/z 194 fragment ion was chosen as the Q3 mass for the pMRM method, as well as the fragment for the precursor/ neutral loss method. A two-injection strategy was used to find the majority of the metabolites. The first injection used the pMRM IDA method and was able to detect 18 potential metabolites. The dual precursor ion/neutral loss IDA method ensured that unpredicted metabolites were not missed. Screens for sulfate and glucuronide conjugation were completed using a neutral loss scan m/z of 80 and 176 in a third injection. In total, 20 potential metabolites were identified with information-rich MS/MS spectra collected using the highly sensitive ion trap. DRUG METABOLISM 49 For Research Use Only. Not for use in diagnostic procedures. LATE STAGE DISCOVERY • • Figure 4: Carbamazepine structure. The portion of the molecule corresponding to the m/z 194 fragment used for precursor scans is shown in blue, and the m/z 43 fragment used for neutral loss scans is shown in orange. • • • Figure 3: The acquisition method wizard in LightSight Software 2.2. A single processing method was created to detect metabolites in all scan modes explored (Figure 5). The identities of several of the metabolites were confirmed using standards. Figure 6 illustrates the metabolite separation achieved using UHPLC. Table 1 summarizes the potential metabolites detected. Confirmatory enhance product ion (EPI) spectra were obtained for all species. The fast-scanning capabilities of the QTRAP 5500 system enable the coupling of specific and sensitive triple quadrupole Figure 5: The processing method for carbamazepine metabolite data. A single processing method can be used for all scan types needed for metabolic profiling on QTRAP systems and includes scan-type specific parameters. 50 RUO-MKT-01-1583-A Metabolic profiling of carbamazepine was performed using LightSight Software 2.2 and the QTRAP 5500 system. LightSight Software 2.2 accelerates metabolic profiling by: • Creating optimized acquisition methods for metabolite detection. • Automatically creating IDA acquisition methods with two survey scans for the QTRAP 5500 system. • Automatically creating glucuronide neutral loss methods. • Creating and submitting acquisition methods using the Acquity® UHPLC system. • Allowing users to create and save processing methods. Use of a single processing method can be used for data reduction and identification of metabolites from dual-survey scan data acquisition. A dual-survey scan approach combining neutral loss and precursor ions scans provides complementary information to the predictive MRM IDA approach. The high speed and sensitivity of the QTRAP 5500 system allows for metabolic-profiling studies at more physiologically relevant concentrations. LATE STAGE DISCOVERY Conclusions scan modes (Prec/NL/MRM) with highly sensitive product ion scans on a UHPLC time-scale. The use of quadrupole mode survey scans can be especially useful for sensitive determination of low-level metabolites in complex matrices. The ability to combine multiple survey scans is an extremely powerful solution for the targeted detection of phase II metabolites such as glucuronide and glutathione conjugates. The aforementioned strategies for detecting metabolites are fully enabled by LightSight Software 2.2. Table 1: Potential metabolites of carbamazepine and their proposed structures (RT = retention time in min). Figure 6: UHPLC separation of carbamazepine metabolites. DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 51 For Research Use Only. Not for use in diagnostic procedures. LATE STAGE DISCOVERY • • Figure 4: Carbamazepine structure. The portion of the molecule corresponding to the m/z 194 fragment used for precursor scans is shown in blue, and the m/z 43 fragment used for neutral loss scans is shown in orange. • • • Figure 3: The acquisition method wizard in LightSight Software 2.2. A single processing method was created to detect metabolites in all scan modes explored (Figure 5). The identities of several of the metabolites were confirmed using standards. Figure 6 illustrates the metabolite separation achieved using UHPLC. Table 1 summarizes the potential metabolites detected. Confirmatory enhance product ion (EPI) spectra were obtained for all species. The fast-scanning capabilities of the QTRAP 5500 system enable the coupling of specific and sensitive triple quadrupole Figure 5: The processing method for carbamazepine metabolite data. A single processing method can be used for all scan types needed for metabolic profiling on QTRAP systems and includes scan-type specific parameters. 50 RUO-MKT-01-1583-A Metabolic profiling of carbamazepine was performed using LightSight Software 2.2 and the QTRAP 5500 system. LightSight Software 2.2 accelerates metabolic profiling by: • Creating optimized acquisition methods for metabolite detection. • Automatically creating IDA acquisition methods with two survey scans for the QTRAP 5500 system. • Automatically creating glucuronide neutral loss methods. • Creating and submitting acquisition methods using the Acquity® UHPLC system. • Allowing users to create and save processing methods. Use of a single processing method can be used for data reduction and identification of metabolites from dual-survey scan data acquisition. A dual-survey scan approach combining neutral loss and precursor ions scans provides complementary information to the predictive MRM IDA approach. The high speed and sensitivity of the QTRAP 5500 system allows for metabolic-profiling studies at more physiologically relevant concentrations. LATE STAGE DISCOVERY Conclusions scan modes (Prec/NL/MRM) with highly sensitive product ion scans on a UHPLC time-scale. The use of quadrupole mode survey scans can be especially useful for sensitive determination of low-level metabolites in complex matrices. The ability to combine multiple survey scans is an extremely powerful solution for the targeted detection of phase II metabolites such as glucuronide and glutathione conjugates. The aforementioned strategies for detecting metabolites are fully enabled by LightSight Software 2.2. Table 1: Potential metabolites of carbamazepine and their proposed structures (RT = retention time in min). Figure 6: UHPLC separation of carbamazepine metabolites. DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 51 Comprehensive Detection of Metabolites Using Polarity Switching Data Collection with the QTRAP® 5500 LC/MS/MS System Carmai Seto AB SCIEX, Concord, Ontario, Canada Key scientific challenges of metabolite ID • • • Thoroughness – In the early stages of drug discovery and development, it is important to find as many metabolites as possible. To eliminate unviable candidates from the pipeline. Restricted sample availability – In some metabolism studies, the supply of sample for analysis may be limited. Acquiringe data in one injection rather than two means using 50% less of an irreplaceable, precious sample. Productivity – Increasing productivity in drug discovery and development continues to be a primary goal in pharmaceutical research. Key benefits of the QTRAP® 5500 System for metabolite ID • • • High sensitivity – The QTRAP 5500 system provides high levels of sensitivity for multiple reaction monitoring (MRM) analysis. Rapid polarity switching – Collecting MS data in both positive and negative ion modes while doing chromatography with narrow peaks is critical for throughput. Fast scanning – Trap scan speeds of up to 20,000 Da/s are available for rapid analyses. Key features of QTRAP 5500 systems for metabolite ID • Unique Workflows – workflows such as positive/negative MRMtriggered information-dependent acquisition (IDA) are supported on the QTRAP 5500 system. • Ease-of-use – LightSight® Software can be used to build acquisition methods and process the data. Positive/negative workflows – More metabolites of varying polarities. Speed – The QTRAP 5500 system is fully compatible with UHPLC, providing better throughput and sensitivity for low-level metabolites. • • 52 RUO-MKT-01-1583-A Experimental conditions In discovery stage pharmaceutical research, information about the extent of metabolism and metabolite structures is used in lead optimization. Identified metabolites can be a source of new therapeutics, toxicity assessment and drug soft spot optimization. LC/MS is widely used for the identification of metabolites at all stages of drug development. Metabolites most often ionize with the same polarity as the parent compound of interest. However, there are cases where this assumption does not hold true thus leading to cases of overlooked metabolites. Polarity switching data collection ensures a more comprehensive way of detecting metabolites of opposite polarities. Simvastatin was incubated in rabbit microsomes at a concentration of 10 µM under oxidative conditions using an NADPH regenerating system. The incubation was quenched with acetonitrile at time zero and 60 min and then centrifuged to pellet the protein. The supernatant was then diluted three-fold with water. A QTRAP 5500 LC/MS/MS system was used for acquisition and LightSight Software for metabolite identification, version 2.1, was used for data interpretation. There are two approaches to collecting polarity switching data; 1) single injection polarity switching and 2) the two injection discrete polarity analysis (one injection in positive and another in negative ion mode). The one injection approach is where the positive and negative ion survey experiments are looped throughout the run with or without dependent IDA. In the two injection approach, the positive and negative ion experiments are in two separate runs. This approach, therefore, takes twice as long to obtain complete data, and LC retention time shifts between samples could complicate data analysis. Furthermore, greater sample amounts are required for a two-injection workflow. Polarity switching data collection can be achieved using various survey scans, such as enhanced mass spectra (EMS) (trap single MS), or quadrupole MS/MS modes such as precursor ion, neutral loss, or MRM. For this study, a pMRM method was used. With prior knowledge of the parent drug and its fragmentation pathway, theoretical MRM transitions for metabolites can be determined based on a particular biotransformation list. For this experiment, two pMRM methods (one positive and one negative ion) were combined into a single experiment. Each of the pMRM surveys contained a total of 48 MRM transitions that covered the range of phase I metabolites. MS/MS data was collected automatically in negative ion via IDA at a scan rate of 10,000 Da/sec. The structure of the method is shown in Figure 1. The overall duty cycle time for this method was approximately 2.6 sec. In this work, metabolites of simvastatin were detected and confirmed in an in vitro incubation: using a single injection polarity switching IDA methodology. Overview The majority of xenobiotic metabolites tend to ionize in the same polarity as the parent drug. Typically, the majority of therapeutics and their downstream metabolites well ionize in positive electrospray ionization (ESI) to provide excellent sensitivity. However, some phase II conjugates, such as phosphate or sulfate additions, completely change the ionization character of the molecule, switching the polarity to negative ESI. Polarity switching data collection ensures the comprehensive detection of metabolites in both polarities within a single LC/MS run. Furthermore, for GSH-conjugate detection, a number of positive/ negative precursor/neutral loss or MRM IDA experiments provide effective coverage for all possible formations1. The QTRAP 5500 system is especially effective at positive/negative IDA methods. The system’s ability to perform fast quadrupole and linear ion trap (LIT) scanning (a 4-fold increase in quadrupole scanning and a 5-fold increase in LIT scanning over the 4000 QTRAP® System), 50 ms polarity switching, and enhanced sensitivity in both quadrupole and LIT mode allow for efficient metabolic detection. These groundbreaking workflows are further optimized by the addition of LightSight Software 2.1 for automated data processing and IDA method creation. DRUG METABOLISM Introduction www.absciex.com 5% A – 85% A in 12.5 min 85% - 95% A in 1 min 95% A isocratic for 1.5 min A is 0.1% formic acid in acetonitrile and B is 0.1% formic acid in water. A 400 uL/min gradient was used with a Phenomenex Luna C18(2) 3 µm, 50 × 2 mm HPLC column. LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Figure 1: IDA method options. The polarity switching IDA method shown here was used to detect metabolites of simvastatin. www.absciex.com DRUG METABOLISM 53 Comprehensive Detection of Metabolites Using Polarity Switching Data Collection with the QTRAP® 5500 LC/MS/MS System Carmai Seto AB SCIEX, Concord, Ontario, Canada Key scientific challenges of metabolite ID • • • Thoroughness – In the early stages of drug discovery and development, it is important to find as many metabolites as possible. To eliminate unviable candidates from the pipeline. Restricted sample availability – In some metabolism studies, the supply of sample for analysis may be limited. Acquiringe data in one injection rather than two means using 50% less of an irreplaceable, precious sample. Productivity – Increasing productivity in drug discovery and development continues to be a primary goal in pharmaceutical research. Key benefits of the QTRAP® 5500 System for metabolite ID • • • High sensitivity – The QTRAP 5500 system provides high levels of sensitivity for multiple reaction monitoring (MRM) analysis. Rapid polarity switching – Collecting MS data in both positive and negative ion modes while doing chromatography with narrow peaks is critical for throughput. Fast scanning – Trap scan speeds of up to 20,000 Da/s are available for rapid analyses. Key features of QTRAP 5500 systems for metabolite ID • Unique Workflows – workflows such as positive/negative MRMtriggered information-dependent acquisition (IDA) are supported on the QTRAP 5500 system. • Ease-of-use – LightSight® Software can be used to build acquisition methods and process the data. Positive/negative workflows – More metabolites of varying polarities. Speed – The QTRAP 5500 system is fully compatible with UHPLC, providing better throughput and sensitivity for low-level metabolites. • • 52 RUO-MKT-01-1583-A Experimental conditions In discovery stage pharmaceutical research, information about the extent of metabolism and metabolite structures is used in lead optimization. Identified metabolites can be a source of new therapeutics, toxicity assessment and drug soft spot optimization. LC/MS is widely used for the identification of metabolites at all stages of drug development. Metabolites most often ionize with the same polarity as the parent compound of interest. However, there are cases where this assumption does not hold true thus leading to cases of overlooked metabolites. Polarity switching data collection ensures a more comprehensive way of detecting metabolites of opposite polarities. Simvastatin was incubated in rabbit microsomes at a concentration of 10 µM under oxidative conditions using an NADPH regenerating system. The incubation was quenched with acetonitrile at time zero and 60 min and then centrifuged to pellet the protein. The supernatant was then diluted three-fold with water. A QTRAP 5500 LC/MS/MS system was used for acquisition and LightSight Software for metabolite identification, version 2.1, was used for data interpretation. There are two approaches to collecting polarity switching data; 1) single injection polarity switching and 2) the two injection discrete polarity analysis (one injection in positive and another in negative ion mode). The one injection approach is where the positive and negative ion survey experiments are looped throughout the run with or without dependent IDA. In the two injection approach, the positive and negative ion experiments are in two separate runs. This approach, therefore, takes twice as long to obtain complete data, and LC retention time shifts between samples could complicate data analysis. Furthermore, greater sample amounts are required for a two-injection workflow. Polarity switching data collection can be achieved using various survey scans, such as enhanced mass spectra (EMS) (trap single MS), or quadrupole MS/MS modes such as precursor ion, neutral loss, or MRM. For this study, a pMRM method was used. With prior knowledge of the parent drug and its fragmentation pathway, theoretical MRM transitions for metabolites can be determined based on a particular biotransformation list. For this experiment, two pMRM methods (one positive and one negative ion) were combined into a single experiment. Each of the pMRM surveys contained a total of 48 MRM transitions that covered the range of phase I metabolites. MS/MS data was collected automatically in negative ion via IDA at a scan rate of 10,000 Da/sec. The structure of the method is shown in Figure 1. The overall duty cycle time for this method was approximately 2.6 sec. In this work, metabolites of simvastatin were detected and confirmed in an in vitro incubation: using a single injection polarity switching IDA methodology. Overview The majority of xenobiotic metabolites tend to ionize in the same polarity as the parent drug. Typically, the majority of therapeutics and their downstream metabolites well ionize in positive electrospray ionization (ESI) to provide excellent sensitivity. However, some phase II conjugates, such as phosphate or sulfate additions, completely change the ionization character of the molecule, switching the polarity to negative ESI. Polarity switching data collection ensures the comprehensive detection of metabolites in both polarities within a single LC/MS run. Furthermore, for GSH-conjugate detection, a number of positive/ negative precursor/neutral loss or MRM IDA experiments provide effective coverage for all possible formations1. The QTRAP 5500 system is especially effective at positive/negative IDA methods. The system’s ability to perform fast quadrupole and linear ion trap (LIT) scanning (a 4-fold increase in quadrupole scanning and a 5-fold increase in LIT scanning over the 4000 QTRAP® System), 50 ms polarity switching, and enhanced sensitivity in both quadrupole and LIT mode allow for efficient metabolic detection. These groundbreaking workflows are further optimized by the addition of LightSight Software 2.1 for automated data processing and IDA method creation. DRUG METABOLISM Introduction www.absciex.com 5% A – 85% A in 12.5 min 85% - 95% A in 1 min 95% A isocratic for 1.5 min A is 0.1% formic acid in acetonitrile and B is 0.1% formic acid in water. A 400 uL/min gradient was used with a Phenomenex Luna C18(2) 3 µm, 50 × 2 mm HPLC column. LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Figure 1: IDA method options. The polarity switching IDA method shown here was used to detect metabolites of simvastatin. www.absciex.com DRUG METABOLISM 53 LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Figure 2: Structures of simvastatin and simvastatin hydroxyl acid. Results and discussion Simvastatin is a lactone prodrug and is hydrolyzed to the active compound (simvastatin hydroxy acid, Figure 2). Both simvastatin and the hydroxy acid ionize in positive ion mode. However, if the MS/MS acquisition method was only in positive ion mode, some metabolites may not be detected because the carboxylic acid moiety ionizes predominately in the negative mode (Figure 3). Since both simvastatin and its carboxylic acid form can be metabolized, it is important to monitor both polarities. Previously, it was not possible to collect data in both polarities as well as confirmatory information in a single injection due to IDA cycle time limitations. However, with improvements to the polarity switching capabilities (50 ms) of the QTRAP 5500 system combined with the fast trap and quadrupole scanning of the new system; many single and dual survey pos/neg IDA workflows are now possible. Proposed structures of some of the metabolites detected are given in Figure 4; some were metabolites of simvastatin, while the majority of those metabolites detected were modifications to the hydroxy acid. Using LightSight Software, a total of 34 metabolites were detected in the microsomal incubation using automated data processing. The metabolites detected in both polarities were correlated in one session (Figure 5), making Figure 3: Pos/Neg switching. The detection of simvastatin carboxylic acid in both positive (top) and negative ion (bottom); the compound is better detected using negative ion mode. reporting of results easier. MS/MS data was collected in both positive ion and negative ion modes, where the data in the negative polarity was collected automatically through IDA, and the data in the positive polarity was collected through multi-experiment, period-based MS/MS methods. Figure 4: Metabolites of simvastatin detected. Metabolites of simvastatin and simvastatin hydroxy acid identified in a rabbit microsomal incubation. Of the 34 metabolites, 5 metabolites were only detected in positive ion, 28 metabolites were detected in negative ion and 1 metabolite (Mx) was detected in both polarities. An advantage of detecting metabolites in both polarities is that MS/MS data can be collected in both polarities, which provides more information for structural elucidation. The MS/MS spectra of Mx are depicted in Figure 6. If the MS/MS spectra was only collected in negative ion Figure 6 (top), the site of oxidation could only be localized to one half of the molecule. However, the positive ion MS/MS spectrum provided enough additional structural information so that the site of oxidation could be further pinpointed to the core ring structure. Figure 5: Polarity switching data processing. An oxidative metabolite of simvastatin hydroxy acid was found using polarity switching data collection. The LightSight Software allows this data to be processed in the same session. 54 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 55 LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Figure 2: Structures of simvastatin and simvastatin hydroxyl acid. Results and discussion Simvastatin is a lactone prodrug and is hydrolyzed to the active compound (simvastatin hydroxy acid, Figure 2). Both simvastatin and the hydroxy acid ionize in positive ion mode. However, if the MS/MS acquisition method was only in positive ion mode, some metabolites may not be detected because the carboxylic acid moiety ionizes predominately in the negative mode (Figure 3). Since both simvastatin and its carboxylic acid form can be metabolized, it is important to monitor both polarities. Previously, it was not possible to collect data in both polarities as well as confirmatory information in a single injection due to IDA cycle time limitations. However, with improvements to the polarity switching capabilities (50 ms) of the QTRAP 5500 system combined with the fast trap and quadrupole scanning of the new system; many single and dual survey pos/neg IDA workflows are now possible. Proposed structures of some of the metabolites detected are given in Figure 4; some were metabolites of simvastatin, while the majority of those metabolites detected were modifications to the hydroxy acid. Using LightSight Software, a total of 34 metabolites were detected in the microsomal incubation using automated data processing. The metabolites detected in both polarities were correlated in one session (Figure 5), making Figure 3: Pos/Neg switching. The detection of simvastatin carboxylic acid in both positive (top) and negative ion (bottom); the compound is better detected using negative ion mode. reporting of results easier. MS/MS data was collected in both positive ion and negative ion modes, where the data in the negative polarity was collected automatically through IDA, and the data in the positive polarity was collected through multi-experiment, period-based MS/MS methods. Figure 4: Metabolites of simvastatin detected. Metabolites of simvastatin and simvastatin hydroxy acid identified in a rabbit microsomal incubation. Of the 34 metabolites, 5 metabolites were only detected in positive ion, 28 metabolites were detected in negative ion and 1 metabolite (Mx) was detected in both polarities. An advantage of detecting metabolites in both polarities is that MS/MS data can be collected in both polarities, which provides more information for structural elucidation. The MS/MS spectra of Mx are depicted in Figure 6. If the MS/MS spectra was only collected in negative ion Figure 6 (top), the site of oxidation could only be localized to one half of the molecule. However, the positive ion MS/MS spectrum provided enough additional structural information so that the site of oxidation could be further pinpointed to the core ring structure. Figure 5: Polarity switching data processing. An oxidative metabolite of simvastatin hydroxy acid was found using polarity switching data collection. The LightSight Software allows this data to be processed in the same session. 54 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 55 LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Figure 6: Structural elucidation. The MS/MS spectra of Mx (oxidation of simvastatin hydroxy acid) in positive (bottom) and negative (top) ion modes is shown. The site of oxidation could be better pinpointed when using both spectra. 56 Summary Highlights Single-injection polarity switching IDA experiments on the QTRAP 5500 system provide improved metabolite detection compared to two-injection single polarity experiments. There are four key factors which support this conclusion. One, a single-injection workflow requires less sample, thus allowing for a large injection volume and improved sensitivity for a single analysis. Two, data reduction is made easier, because only one LC/MS experiment is required to be processed. Three, combined positive and negative ESI metabolite profiles allow for complete confirmation of varied species in a single analysis. Four, the fast-scanning attributes in quadrupole and LIT modes of the QTRAP 5500 system along with fast polarity switching capabilities allow for higher throughput analyses by incorporating faster LC strategies. • RUO-MKT-01-1583-A • • • • • • Improved quadrupole sensitivity (MRM response and signal-to-noise ratio, PI, and NL) Sensitivity improvement in trap modes (EPI, 10-100-fold) Four times faster triple quadrupole scanning Faster polarity switching (50 ms) Five times faster LIT scanning (20,000 amu/sec) Shorter fill time capabilities (50 μs) Unique workflows ideal for metabolite identification DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 57 LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Figure 6: Structural elucidation. The MS/MS spectra of Mx (oxidation of simvastatin hydroxy acid) in positive (bottom) and negative (top) ion modes is shown. The site of oxidation could be better pinpointed when using both spectra. 56 Summary Highlights Single-injection polarity switching IDA experiments on the QTRAP 5500 system provide improved metabolite detection compared to two-injection single polarity experiments. There are four key factors which support this conclusion. One, a single-injection workflow requires less sample, thus allowing for a large injection volume and improved sensitivity for a single analysis. Two, data reduction is made easier, because only one LC/MS experiment is required to be processed. Three, combined positive and negative ESI metabolite profiles allow for complete confirmation of varied species in a single analysis. Four, the fast-scanning attributes in quadrupole and LIT modes of the QTRAP 5500 system along with fast polarity switching capabilities allow for higher throughput analyses by incorporating faster LC strategies. • RUO-MKT-01-1583-A • • • • • • Improved quadrupole sensitivity (MRM response and signal-to-noise ratio, PI, and NL) Sensitivity improvement in trap modes (EPI, 10-100-fold) Four times faster triple quadrupole scanning Faster polarity switching (50 ms) Five times faster LIT scanning (20,000 amu/sec) Shorter fill time capabilities (50 μs) Unique workflows ideal for metabolite identification DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 57 Simultaneous Pharmacokinetic Profiling and Automated Metabolite Identification Using the AB SCIEX TripleTOF® 5600 System and MetabolitePilot™ Software The AB SCIEX TripleTOF 5600 System is a state-of-the-art high-resolution accurate mass instrument possessing the speed, sensitivity, and linearity to deliver triple-quadrupolelike quantitative performance and accurate mass metabolite identification in the same injection and at the same time. Suma Ramagiri, Jeff Miller, and Hesham Ghobarah Materials and methods AB SCIEX, Toronto, Ontario and Framingham, Massachusetts This application note describes the use of the TripleTOF 5600 system for simultaneous in vivo PK quantification and identification of the major metabolites of buspirone following administration in rat using the workflow in Figure 1. In vivo administration Three Sprague-Dawley rats were dosed with buspirone at 10 mg/kg intravenously. Plasma samples were collected at 0, 15, 30, 60, 120, 240, 360, and 480 min. Key scientific challenges of combined quant and qual workflows Instrumentation – Prior to the debut of the TripleTOF® 5600 System, obtaining accurate quantification and high-quality qualitative information required using a triple quad mass spectrometer for quant and an accurate mass instrument for qual. • Analysis at relevant doses – For many compounds, ADME studies were previously conducted at a higher, non-clinically relevant concentrations to monitor drug metabolism, occasionally leading to false conclusions as the metabolic pathway has been found to be concentration-dependent metabolic pathways for some compounds. Productivity – The ability to combine quantitative bioanalysis and in vivo metabolite ID into one analysis will increase productivity in both drug discovery and development. Protein was precipitated from plasma samples using acetonitrile containing an internal standard (buspirone-d8) at a 3:1 ratio followed by centrifugation at 10,000 x g. The supernatant (100 µL) was mixed with 50 µL water, transferred into autosampler vials, and injected. • • Quant and qual in one run – The sensitivity of TripleTOF systems on par with the API 4000™ System, which are known throughout the industry for their sensitivity and robustness. In the same instrument, the high performance accurate mass analyzer allows for both quantitative and qualitative analyses in a single method. Intelligent data acquisition – Real-time multiple mass-defect filtering (MDF) for information-dependent acquisition (IDA) increases workflow efficiency by acquiring relevant MS/MS data in every run, eliminating the need for re-injection in many cases. Speed – Both drug discovery and development labs are standardizing on ultra-high pressure liquid chromatography (UHPLC) as the separation method of choice. UHPLC’s narrower chromatographic peaks result in higher concentrations at peak apices and, therefore, lower LOQs. To accommodate the UHPLC time scale, mass spectrometers must perform the needed experiments on a short time scale. The TripleTOF systems are designed to keep up with UHPLC peaks. Key features of TripleTOF systems for quant/qual workflows • 58 Resolution and mass accuracy regardless of scan speed – Instruments employing orbital trapping require slower scanning to get higher resolution. The TripleTOF system maintains approximately 30K resolution regardless of analysis speed. RUO-MKT-01-1583-A • • Resolution and mass accuracy even at low m/z – Traditional time-offlight (TOF) instruments provided good mass accuracy and resolution at high m/z. The high detector speed of the TripleTOF 5600 system allows for sufficient resolution at low m/z, providing unambiguous formulae ssignments when using accurate mass information, as well as the isotope and fragmentation pattern. Integrated software packages – To date, no manufacturer has provided one software package capable of finding metabolites, confirming sites of metabolism, tracking time course experiments, and doing inter-species comparisons. MetabolitePilot Software does all these. DRUG METABOLISM www.absciex.com The system was operated in positive electrospray mode using a DuoSpray™ Ion Source. The information-dependent acquisition (IDA) method consisted of a TOF MS survey scan (m/z 100–1000) followed by three TOF MS/MS dependent scans (m/z 50–1000). The TOF MS scan data was used for quantification, while the MS/MS data was used for the accurate mass structure elucidation of detected metabolites. Mass defect filtering (MDF) has been shown to be a powerful tool in detecting metabolites that are similar in elemental composition to the parent. Traditionally, MDF is a two-step process, where full-scan data is acquired first, then MDF analysis is performed post-processing to identify peaks of interest. A second injection is then performed to acquire the MS/MS spectra of these potential metabolites. Parent metabolite quantification The high-speed TOF MS survey scan was used for the dual purpose of parent compound quantification and detection of potential metabolites. Using real-time MDF to increase IDA efficiency, accurate mass product ion spectra were triggered on potential metabolites and used for structural elucidation. Quantitative PK data for buspirone parent compounds were easily processed in MultiQuant™ Software (Figure 3). Introduction Combining quantitative bioanalysis and in vivo metabolite identification (quant/qual) in the same workflow has the potential to significantly increase the productivity of pharmaceutical drug discovery and development. Traditionally, quantitative bioanalysis is performed on samples obtained from pharmacokinetic (PK) studies, while metabolite identification is done in a separate analysis often on two different instruments. Due to sensitivity limitations of traditional LC/MS/MS instruments, studies for metabolite identification are often performed at much higher, non-physiologically relevant doses than for PK studies. Mass spectrometry Using real-time mass defect filtering to trigger IDA, the TripleTOF 5600 system can acquire both MS and MS/MS data in the same run, eliminating the need for a second injection. Multiple mass defect ranges are often required to detect different metabolite classes. The system is capable of applying multiple filters at the same time (Figure 2). The software automatically calculates the mass defects based on the elemental compositions provided and performs intelligent, priority-based MS/MS triggering. This gives priority to peaks matching the mass defect range for MS/MS data acquisition so that both expected and unexpected metabolites can be detected. Key benefits of the TripleTOF 5600 system for quant/qual workflows • Sample analysis was performed on the TripleTOF 5600 system coupled to an Acquity UPLC system. An acetonitrile/water /0.1% formic acid gradient was used on a Waters BEH C18, 2.1×100 mm, 1.7 μm column. The flow rate was 0.6 mL/min, and the column was heated to 50°C. Real-time multiple mass defect filter Sample preparation • • Chromatography LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Figure 1: Automated pharmacokinetic (PK) profiling and metabolite identification using the AB SCIEX TripleTOF® 5600 System with MetabolitePilot™ Software and MultiQuant™ Software packages. www.absciex.com DRUG METABOLISM 59 Simultaneous Pharmacokinetic Profiling and Automated Metabolite Identification Using the AB SCIEX TripleTOF® 5600 System and MetabolitePilot™ Software The AB SCIEX TripleTOF 5600 System is a state-of-the-art high-resolution accurate mass instrument possessing the speed, sensitivity, and linearity to deliver triple-quadrupolelike quantitative performance and accurate mass metabolite identification in the same injection and at the same time. Suma Ramagiri, Jeff Miller, and Hesham Ghobarah Materials and methods AB SCIEX, Toronto, Ontario and Framingham, Massachusetts This application note describes the use of the TripleTOF 5600 system for simultaneous in vivo PK quantification and identification of the major metabolites of buspirone following administration in rat using the workflow in Figure 1. In vivo administration Three Sprague-Dawley rats were dosed with buspirone at 10 mg/kg intravenously. Plasma samples were collected at 0, 15, 30, 60, 120, 240, 360, and 480 min. Key scientific challenges of combined quant and qual workflows Instrumentation – Prior to the debut of the TripleTOF® 5600 System, obtaining accurate quantification and high-quality qualitative information required using a triple quad mass spectrometer for quant and an accurate mass instrument for qual. • Analysis at relevant doses – For many compounds, ADME studies were previously conducted at a higher, non-clinically relevant concentrations to monitor drug metabolism, occasionally leading to false conclusions as the metabolic pathway has been found to be concentration-dependent metabolic pathways for some compounds. Productivity – The ability to combine quantitative bioanalysis and in vivo metabolite ID into one analysis will increase productivity in both drug discovery and development. Protein was precipitated from plasma samples using acetonitrile containing an internal standard (buspirone-d8) at a 3:1 ratio followed by centrifugation at 10,000 x g. The supernatant (100 µL) was mixed with 50 µL water, transferred into autosampler vials, and injected. • • Quant and qual in one run – The sensitivity of TripleTOF systems on par with the API 4000™ System, which are known throughout the industry for their sensitivity and robustness. In the same instrument, the high performance accurate mass analyzer allows for both quantitative and qualitative analyses in a single method. Intelligent data acquisition – Real-time multiple mass-defect filtering (MDF) for information-dependent acquisition (IDA) increases workflow efficiency by acquiring relevant MS/MS data in every run, eliminating the need for re-injection in many cases. Speed – Both drug discovery and development labs are standardizing on ultra-high pressure liquid chromatography (UHPLC) as the separation method of choice. UHPLC’s narrower chromatographic peaks result in higher concentrations at peak apices and, therefore, lower LOQs. To accommodate the UHPLC time scale, mass spectrometers must perform the needed experiments on a short time scale. The TripleTOF systems are designed to keep up with UHPLC peaks. Key features of TripleTOF systems for quant/qual workflows • 58 Resolution and mass accuracy regardless of scan speed – Instruments employing orbital trapping require slower scanning to get higher resolution. The TripleTOF system maintains approximately 30K resolution regardless of analysis speed. RUO-MKT-01-1583-A • • Resolution and mass accuracy even at low m/z – Traditional time-offlight (TOF) instruments provided good mass accuracy and resolution at high m/z. The high detector speed of the TripleTOF 5600 system allows for sufficient resolution at low m/z, providing unambiguous formulae ssignments when using accurate mass information, as well as the isotope and fragmentation pattern. Integrated software packages – To date, no manufacturer has provided one software package capable of finding metabolites, confirming sites of metabolism, tracking time course experiments, and doing inter-species comparisons. MetabolitePilot Software does all these. DRUG METABOLISM www.absciex.com The system was operated in positive electrospray mode using a DuoSpray™ Ion Source. The information-dependent acquisition (IDA) method consisted of a TOF MS survey scan (m/z 100–1000) followed by three TOF MS/MS dependent scans (m/z 50–1000). The TOF MS scan data was used for quantification, while the MS/MS data was used for the accurate mass structure elucidation of detected metabolites. Mass defect filtering (MDF) has been shown to be a powerful tool in detecting metabolites that are similar in elemental composition to the parent. Traditionally, MDF is a two-step process, where full-scan data is acquired first, then MDF analysis is performed post-processing to identify peaks of interest. A second injection is then performed to acquire the MS/MS spectra of these potential metabolites. Parent metabolite quantification The high-speed TOF MS survey scan was used for the dual purpose of parent compound quantification and detection of potential metabolites. Using real-time MDF to increase IDA efficiency, accurate mass product ion spectra were triggered on potential metabolites and used for structural elucidation. Quantitative PK data for buspirone parent compounds were easily processed in MultiQuant™ Software (Figure 3). Introduction Combining quantitative bioanalysis and in vivo metabolite identification (quant/qual) in the same workflow has the potential to significantly increase the productivity of pharmaceutical drug discovery and development. Traditionally, quantitative bioanalysis is performed on samples obtained from pharmacokinetic (PK) studies, while metabolite identification is done in a separate analysis often on two different instruments. Due to sensitivity limitations of traditional LC/MS/MS instruments, studies for metabolite identification are often performed at much higher, non-physiologically relevant doses than for PK studies. Mass spectrometry Using real-time mass defect filtering to trigger IDA, the TripleTOF 5600 system can acquire both MS and MS/MS data in the same run, eliminating the need for a second injection. Multiple mass defect ranges are often required to detect different metabolite classes. The system is capable of applying multiple filters at the same time (Figure 2). The software automatically calculates the mass defects based on the elemental compositions provided and performs intelligent, priority-based MS/MS triggering. This gives priority to peaks matching the mass defect range for MS/MS data acquisition so that both expected and unexpected metabolites can be detected. Key benefits of the TripleTOF 5600 system for quant/qual workflows • Sample analysis was performed on the TripleTOF 5600 system coupled to an Acquity UPLC system. An acetonitrile/water /0.1% formic acid gradient was used on a Waters BEH C18, 2.1×100 mm, 1.7 μm column. The flow rate was 0.6 mL/min, and the column was heated to 50°C. Real-time multiple mass defect filter Sample preparation • • Chromatography LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Figure 1: Automated pharmacokinetic (PK) profiling and metabolite identification using the AB SCIEX TripleTOF® 5600 System with MetabolitePilot™ Software and MultiQuant™ Software packages. www.absciex.com DRUG METABOLISM 59 LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Figure 2: Automated real-time multiple mass defect filter for MS/MS triggering on Phase I and II metabolites and glutathione conjugates in information-dependent acquisition (IDA) mode. Data from multiple buspirone PK time points were processed in batch mode in MetabolitePilot Software (Figure 4). In addition to conventional sample to control comparison, additional detection algorithms such as MDF were applied at the same time. Mass defect filters for multiple phase I and II metabolite as well as cleavage metabolites were automatically calculated based on parent compound structure and used for data processing. Other peak finding strategies can also be used such as isotope pattern filtering and searching for common accurate mass product ions or neutral losses. A confirmation score is automatically calculated for each potential metabolite detected, based on mass accuracy, isotope pattern, mass defect, and similarity of the MS/MS spectrum to that of the parent (Figure 5). A diverse range of buspirone metabolites was successfully detected (Table 1). Due to the high mass accuracy, an unambiguous assignment of elemental composition was achieved. High-resolution, high-mass-accuracy product ion spectra were obtained without the need for a second injection due to the high IDA triggering efficiency of the system. Figure 4: Automated batch processing of multiple pharmacokinetic (PK) time course samples for metabolite identification in MetabolitePilot™ Software. RUO-MKT-01-1583-A Figure 6: Structural elucidation of a mono-hydroxy buspirone metabolite using PeakView® Software is shown. Figure 7: Relative quantification of parent and metabolites of buspirone. Peak area versus time profiles (0-8hr) for buspirone parent and metabolites in rat plasma, generated using MultiQuant™ Software. Structural elucidation Metabolite identification and confirmation using MetabolitePilot™ Software 60 Figure 3: Buspirone was quantified in rat plasma from TOF MS data using MultiQuant™ Software. High mass accuracy for the fragment ions in the product ion spectra is a particularly valuable aid when identifying the site of metabolism, because elemental compositions can be unambiguously assigned to the product ions. The structural elucidation workspace quickly performs this task (Figure 6). Relative quantification of metabolites • • • Once metabolites are identified, it is often very important to estimate their concentrations and kinetic profile relative to the parent. This was easily performed by exporting the accurate masses of the metabolites to MultiQuant™ Software and plotting the areas vs. time (Figure 7). Because the TOF MS scan is completely non-targeted, metabolite information can be queried without the need to reacquire data. This would also aid in identifying metabolites that are disproportionate across different species to address MIST questions. References Conclusions • High-throughput in vivo quant/qual was successfully performed using the AB SCIEX TripleTOF 5600 system combined with MetabolitePilot™ Software and MultiQuant™ Software. The high speed and sensitivity of the system enabled metabolite detection and identification on standard PK plasma samples. Efficient data processing software and the successful application of multiple peak-finding strategies resulted in comprehensive phase I and II metabolite coverage as well as unexpected cleavage metabolites for buspirone. Obtaining high-mass accuracy product ion spectra without compromising speed is invaluable for structure elucidation of metabolites and the identification of the site of metabolism for soft spot optimization. 1 King RC, Gundersdorf R, Fernández-Metzler CL, Rapid Commun Mass Spectrom. 2003; 17(21), 2413-22. 2 Li AC, Alton D, Bryant MS, Shou WZ, Rapid Commun Mass Spectrom. 2005;19(14), 1943-50. 3 Xia YQ, Miller JD, Bakhtiar R, Franklin RB, Liu DQ, Rapid Commun Mass Spectrom. 2003; 17(11), 1137-45. 4 Zhang H, Zhang D, Ray K, J. Mass Spectrom. 2003 Oct;38(10), 1110-2. 5 Zhu M, Ma L, Zhang D, Ray K, Zhao W, Humphreys WG, Skiles G, Sanders M, Zhang H, Drug Metab Dis-pos. 2006 Oct;34(10), 1722-33. 6 Zhang H, Zhu M, Ray KL, Ma L, Zhang D, Rapid Commun Mass Spectrom. 2008 Jul; 22(13):2082-8. Acknowledgements The authors would like to acknowledge Drs. Ragu Ramanathan, Jonathan Josephs, Nirmala Raghavan, Emily Luk, and Yanou Yang for providing in vivo buspirone rat PK samples for analysis; the authors would also like to thank Drs. Gary Impey and Christie Hunter for their critical review. Figure 5: MetabolitePilot™ Software results window displays multiple forms of metabolite data: phase I and II metabolites with proposed elemental composition, mass accuracy, % area, extracted ion chromatograms (XIC) of metabolites, TOF MS and MS/MS spectra, with scoring information, all in one window for easy visualization. DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 61 LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. Figure 2: Automated real-time multiple mass defect filter for MS/MS triggering on Phase I and II metabolites and glutathione conjugates in information-dependent acquisition (IDA) mode. Data from multiple buspirone PK time points were processed in batch mode in MetabolitePilot Software (Figure 4). In addition to conventional sample to control comparison, additional detection algorithms such as MDF were applied at the same time. Mass defect filters for multiple phase I and II metabolite as well as cleavage metabolites were automatically calculated based on parent compound structure and used for data processing. Other peak finding strategies can also be used such as isotope pattern filtering and searching for common accurate mass product ions or neutral losses. A confirmation score is automatically calculated for each potential metabolite detected, based on mass accuracy, isotope pattern, mass defect, and similarity of the MS/MS spectrum to that of the parent (Figure 5). A diverse range of buspirone metabolites was successfully detected (Table 1). Due to the high mass accuracy, an unambiguous assignment of elemental composition was achieved. High-resolution, high-mass-accuracy product ion spectra were obtained without the need for a second injection due to the high IDA triggering efficiency of the system. Figure 4: Automated batch processing of multiple pharmacokinetic (PK) time course samples for metabolite identification in MetabolitePilot™ Software. RUO-MKT-01-1583-A Figure 6: Structural elucidation of a mono-hydroxy buspirone metabolite using PeakView® Software is shown. Figure 7: Relative quantification of parent and metabolites of buspirone. Peak area versus time profiles (0-8hr) for buspirone parent and metabolites in rat plasma, generated using MultiQuant™ Software. Structural elucidation Metabolite identification and confirmation using MetabolitePilot™ Software 60 Figure 3: Buspirone was quantified in rat plasma from TOF MS data using MultiQuant™ Software. High mass accuracy for the fragment ions in the product ion spectra is a particularly valuable aid when identifying the site of metabolism, because elemental compositions can be unambiguously assigned to the product ions. The structural elucidation workspace quickly performs this task (Figure 6). Relative quantification of metabolites • • • Once metabolites are identified, it is often very important to estimate their concentrations and kinetic profile relative to the parent. This was easily performed by exporting the accurate masses of the metabolites to MultiQuant™ Software and plotting the areas vs. time (Figure 7). Because the TOF MS scan is completely non-targeted, metabolite information can be queried without the need to reacquire data. This would also aid in identifying metabolites that are disproportionate across different species to address MIST questions. References Conclusions • High-throughput in vivo quant/qual was successfully performed using the AB SCIEX TripleTOF 5600 system combined with MetabolitePilot™ Software and MultiQuant™ Software. The high speed and sensitivity of the system enabled metabolite detection and identification on standard PK plasma samples. Efficient data processing software and the successful application of multiple peak-finding strategies resulted in comprehensive phase I and II metabolite coverage as well as unexpected cleavage metabolites for buspirone. Obtaining high-mass accuracy product ion spectra without compromising speed is invaluable for structure elucidation of metabolites and the identification of the site of metabolism for soft spot optimization. 1 King RC, Gundersdorf R, Fernández-Metzler CL, Rapid Commun Mass Spectrom. 2003; 17(21), 2413-22. 2 Li AC, Alton D, Bryant MS, Shou WZ, Rapid Commun Mass Spectrom. 2005;19(14), 1943-50. 3 Xia YQ, Miller JD, Bakhtiar R, Franklin RB, Liu DQ, Rapid Commun Mass Spectrom. 2003; 17(11), 1137-45. 4 Zhang H, Zhang D, Ray K, J. Mass Spectrom. 2003 Oct;38(10), 1110-2. 5 Zhu M, Ma L, Zhang D, Ray K, Zhao W, Humphreys WG, Skiles G, Sanders M, Zhang H, Drug Metab Dis-pos. 2006 Oct;34(10), 1722-33. 6 Zhang H, Zhu M, Ray KL, Ma L, Zhang D, Rapid Commun Mass Spectrom. 2008 Jul; 22(13):2082-8. Acknowledgements The authors would like to acknowledge Drs. Ragu Ramanathan, Jonathan Josephs, Nirmala Raghavan, Emily Luk, and Yanou Yang for providing in vivo buspirone rat PK samples for analysis; the authors would also like to thank Drs. Gary Impey and Christie Hunter for their critical review. Figure 5: MetabolitePilot™ Software results window displays multiple forms of metabolite data: phase I and II metabolites with proposed elemental composition, mass accuracy, % area, extracted ion chromatograms (XIC) of metabolites, TOF MS and MS/MS spectra, with scoring information, all in one window for easy visualization. DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 61 Metabolite Identification with the QTRAP® 5500 LC/MS/MS System: Sensitivity, Selectivity, Speed, and Unique Workflows Key scientific challenges of metabolite ID • • Matrix interferences – Extracting or finding metabolites in the matrices from in vitro and especially in vivo metabolic studies often requires both sensitive and specific scan types. Throughput backlogs – High-throughput analyses, driven by efficient data collection and reporting, is or rapid detection and identification of metabolites in drug discovery laboratories. Key benefits of the QTRAP 5500® System and for metabolite ID • • • Sensitivity – The high sensitivity of the QTRAP 5500 system allows for metabolic profiling at physiologically relevant dosing. Specificity – The unique scanning functions of the QTRAP 5500 systems combine high specificity and sensitivity for powerful metabolite ID workflows. Speed – High scan rates allow the instrument to keep up with UHPLC chromatography. Key features of the QTRAP 5500 system and for metabolite ID • • • TripleTrap™ Scanning – these scanning functions unique to the QTRAP systems combine highly specific triple quadrupole scans with very fast and sensitive linear ion trap scans for powerful metabolite ID workflows. Sensitivity – The high sensitivity of the QTRAP 5500 system enables more metabolites to be found than with previous systems. Fast scanning – The highest scanning rates and rapid polarity switching permit the fast data collection necessary to keep pace with UHPLC chromatography. Overview The QTRAP 5500 system reduces the challenges of “identifying metabolites during drug discovery and development. The enhanced speed and sensitivity of the QTRAP 5500 system provides a more efficient workflows for metabolite identification; the predictive multiple reaction monitoring (pMRM) approach and the use of multiple precursor ion and/or neutral loss survey scans in a single analysis, including polarity switching, means the ability to find and identify lower-level metabolites is better than ever before. When used with LightSight® Software, the QTRAP 5500 system serves as a workhorse for metabolite detection and complements accurate mass approaches. QTRAP 5500 system overview The QTRAP 5500 system is a completely redesigned platform. The ion path (Figure 1) consists of a curved collision cell that permits a reduced platform footprint; additional enhancements to instrumentation have lead to multiple performance improvements. The new QJet 2 ion guide is 2.5-times longer (125mm) than the ion guide first implemented in the API 5000™ System. The addition of the QJet ion guide offers improved protection from contamination of the ion optics, but its main function is to efficiently capture and focus ions into the Q0 region. A major advancement in the ion path is the new Linear Accelerator™ Trap Collision Cell, which brings LINAC® Collision Cell technology to the Q3 quadrupole for improved resolution and simplified tuning procedure. Additionally, LINAC technology provides improved ion extraction efficiencies for as much as 100-fold gain in sensitivity for ion trap scan modes. Additionally, there’s been a reduction in ion cooling fragmentation times producing superior MS3 results. Together with new electronics, all of the above changes have resulted in significant performance enhancements in speed, selectivity and sensitivity. Improvements include: Introduction Metabolite identification is central to many of the activities in the discovery and development pipeline. From rapid structural assignment during the discovery process to provide an early perspective on a drug candidate’s metabolically labile sites or soft spot analysis, to a more complete characterization of pharmacokinetic properties to establish dose and toxicity levels, there’s an ever-increasing demand on throughput. The ability to find, identify, and confirm metabolites as fast as possible is critical, for both major and very low-level metabolites. Doing this in a single analysis is a highly sought after goal. The utility of QTRAP system technology with its unique combination of triple quadrupole and linear ion trap (LIT) scan capabilities together with automated approaches to metabolite identification has brought this goal closer to reality. • Increased quadrupole sensitivity (MRM response, and signal-to-noise, product ion and neutral loss) • Enhanced sensitivity 10-100-fold sensitivity improvement in trap modes (EPI MS/MS) • More rapid (up to 4-fold faster) QqQ scanning • Faster polarity switching (50 ms) • Faster (up to 5-fold) LIT scanning (20,000 Da/ s) The enhanced speed and sensitivity of the QTRAP 5500 system means a more efficient workflow for metabolite identification; the (pMRM) approach and the use of multiple precursor ion and/or neutral loss survey scans in a single analysis, including polarity switching, means the ability to find and identify lower-level metabolites is better than ever before. Below, challenging metabolite ID applications that are simplified with the QTRAP 5500 system are discussed. Metabolite ID examples • In vivo analyses • Microdosing studies LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. • Metabolic stability studies • GSH conjugate screening (reactive metabolites) In vivo analyses Detecting metabolites from in vivo samples at clinically relevant concentrations is a critical challenge for drug discovery and development. A wide range of complex matrices (bile, plasma, urine, and fecal extracts) can pose a number of critical issues and challenges during metabolite analysis, including the requirement for high sensitivity to detect low levels of circulating metabolites. Furthermore, these biofluids can cause ion suppression, further decreasing in sensitivity. Additionally, the complexity of background signals from biological matrices increases the complexity of MS and MS/MS data analysis. In general, the selective nature of MRM, precursor, and neutral loss scans provide the highest level of detection compared to any single MS-based methodology. • Shorter fill time capabilities (50 μs) for faster analyses The QTRAP 5500 system brings a number of key advantages to metabolite identification. The addition of the QJet® 2 Ion Guide and improved Q3 LIT increases signal response 9-fold in quadrupole mode and 10- to 100-fold in LIT mode. The overall duty cycle is further improved with trap fill times as short at 50 μs due to linear ion trap sensitivity increases, and polarity switching as low as 50 ms. The system also has enhanced scan rates of up to 20,000 Da/s in LIT mode that are 4-fold faster in quadrupole mode. This greater cycle rate allows for compatibility with UHPLC time scales as well as improved information-dependent acquisition (IDA) coverage provided by a greater number of MS/MS-dependent scans on relevant peaks. This tech note describes several key strategies to identify metabolites in a single analysis. Figure 1: The ion path of the QTRAP® 5500 System. 62 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 63 Metabolite Identification with the QTRAP® 5500 LC/MS/MS System: Sensitivity, Selectivity, Speed, and Unique Workflows Key scientific challenges of metabolite ID • • Matrix interferences – Extracting or finding metabolites in the matrices from in vitro and especially in vivo metabolic studies often requires both sensitive and specific scan types. Throughput backlogs – High-throughput analyses, driven by efficient data collection and reporting, is or rapid detection and identification of metabolites in drug discovery laboratories. Key benefits of the QTRAP 5500® System and for metabolite ID • • • Sensitivity – The high sensitivity of the QTRAP 5500 system allows for metabolic profiling at physiologically relevant dosing. Specificity – The unique scanning functions of the QTRAP 5500 systems combine high specificity and sensitivity for powerful metabolite ID workflows. Speed – High scan rates allow the instrument to keep up with UHPLC chromatography. Key features of the QTRAP 5500 system and for metabolite ID • • • TripleTrap™ Scanning – these scanning functions unique to the QTRAP systems combine highly specific triple quadrupole scans with very fast and sensitive linear ion trap scans for powerful metabolite ID workflows. Sensitivity – The high sensitivity of the QTRAP 5500 system enables more metabolites to be found than with previous systems. Fast scanning – The highest scanning rates and rapid polarity switching permit the fast data collection necessary to keep pace with UHPLC chromatography. Overview The QTRAP 5500 system reduces the challenges of “identifying metabolites during drug discovery and development. The enhanced speed and sensitivity of the QTRAP 5500 system provides a more efficient workflows for metabolite identification; the predictive multiple reaction monitoring (pMRM) approach and the use of multiple precursor ion and/or neutral loss survey scans in a single analysis, including polarity switching, means the ability to find and identify lower-level metabolites is better than ever before. When used with LightSight® Software, the QTRAP 5500 system serves as a workhorse for metabolite detection and complements accurate mass approaches. QTRAP 5500 system overview The QTRAP 5500 system is a completely redesigned platform. The ion path (Figure 1) consists of a curved collision cell that permits a reduced platform footprint; additional enhancements to instrumentation have lead to multiple performance improvements. The new QJet 2 ion guide is 2.5-times longer (125mm) than the ion guide first implemented in the API 5000™ System. The addition of the QJet ion guide offers improved protection from contamination of the ion optics, but its main function is to efficiently capture and focus ions into the Q0 region. A major advancement in the ion path is the new Linear Accelerator™ Trap Collision Cell, which brings LINAC® Collision Cell technology to the Q3 quadrupole for improved resolution and simplified tuning procedure. Additionally, LINAC technology provides improved ion extraction efficiencies for as much as 100-fold gain in sensitivity for ion trap scan modes. Additionally, there’s been a reduction in ion cooling fragmentation times producing superior MS3 results. Together with new electronics, all of the above changes have resulted in significant performance enhancements in speed, selectivity and sensitivity. Improvements include: Introduction Metabolite identification is central to many of the activities in the discovery and development pipeline. From rapid structural assignment during the discovery process to provide an early perspective on a drug candidate’s metabolically labile sites or soft spot analysis, to a more complete characterization of pharmacokinetic properties to establish dose and toxicity levels, there’s an ever-increasing demand on throughput. The ability to find, identify, and confirm metabolites as fast as possible is critical, for both major and very low-level metabolites. Doing this in a single analysis is a highly sought after goal. The utility of QTRAP system technology with its unique combination of triple quadrupole and linear ion trap (LIT) scan capabilities together with automated approaches to metabolite identification has brought this goal closer to reality. • Increased quadrupole sensitivity (MRM response, and signal-to-noise, product ion and neutral loss) • Enhanced sensitivity 10-100-fold sensitivity improvement in trap modes (EPI MS/MS) • More rapid (up to 4-fold faster) QqQ scanning • Faster polarity switching (50 ms) • Faster (up to 5-fold) LIT scanning (20,000 Da/ s) The enhanced speed and sensitivity of the QTRAP 5500 system means a more efficient workflow for metabolite identification; the (pMRM) approach and the use of multiple precursor ion and/or neutral loss survey scans in a single analysis, including polarity switching, means the ability to find and identify lower-level metabolites is better than ever before. Below, challenging metabolite ID applications that are simplified with the QTRAP 5500 system are discussed. Metabolite ID examples • In vivo analyses • Microdosing studies LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. • Metabolic stability studies • GSH conjugate screening (reactive metabolites) In vivo analyses Detecting metabolites from in vivo samples at clinically relevant concentrations is a critical challenge for drug discovery and development. A wide range of complex matrices (bile, plasma, urine, and fecal extracts) can pose a number of critical issues and challenges during metabolite analysis, including the requirement for high sensitivity to detect low levels of circulating metabolites. Furthermore, these biofluids can cause ion suppression, further decreasing in sensitivity. Additionally, the complexity of background signals from biological matrices increases the complexity of MS and MS/MS data analysis. In general, the selective nature of MRM, precursor, and neutral loss scans provide the highest level of detection compared to any single MS-based methodology. • Shorter fill time capabilities (50 μs) for faster analyses The QTRAP 5500 system brings a number of key advantages to metabolite identification. The addition of the QJet® 2 Ion Guide and improved Q3 LIT increases signal response 9-fold in quadrupole mode and 10- to 100-fold in LIT mode. The overall duty cycle is further improved with trap fill times as short at 50 μs due to linear ion trap sensitivity increases, and polarity switching as low as 50 ms. The system also has enhanced scan rates of up to 20,000 Da/s in LIT mode that are 4-fold faster in quadrupole mode. This greater cycle rate allows for compatibility with UHPLC time scales as well as improved information-dependent acquisition (IDA) coverage provided by a greater number of MS/MS-dependent scans on relevant peaks. This tech note describes several key strategies to identify metabolites in a single analysis. Figure 1: The ion path of the QTRAP® 5500 System. 62 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 63 In this work, a set of verapamil in vitro hepatocyte metabolites were spiked into bile matrix at a low level (2.5 μM) to mimic the in vivo sample matrix. LightSight® Software 2.1 has allowed for a new novel survey mode called pMRM (predictive MRM). This new type of IDA experiment can be created automatically using LightSight Software 2.1 and loaded directly onto the QTRAP system for rapid analysis. The advantage of pMRM is the improved signal-to-noise ratio, IDA selectivity, and efficiency compared to single MS strategies. The QTRAP 5500 system with pMRM survey mode as well as dual precursor and neutral loss scan mode was evaluated against a two injection mass defect filter (MDF) high-resolution experiment “Breakthrough in vivo metabolites analysis using the QTRAP 5500 system and Predictive MRM”. Using the targeted pMRM approach, 158 transitions were created by LightSight Software 2.1 to search for phase I and II metabolites. A total of 1.5 sec was the total duty cycle for the method that included automated collection of MS/MS at 20,000 amu/sec. When employing the multiple precursor ion and neutral loss approach, the overall duty cycle was 2.2 sec. Both workflows had cycle times well-suited for high-throughput chromatographic analyses. To summarize, even at higher-throughput chromatographic conditions (by a factor of 2), the QTRAP 5500 system found and identified with confirmatory MS/MS, 7-fold more metabolites using a single injection workflow than the high-resolution approach. With the addition of a second injection, the QTRAP 5500 system still managed to top the high-resolution experiments, finding 2-fold more metabolites (Figure 2). Microdosing studies Many new chemical entities fail in phase I clinical research studies due to poor pharmacokinetics. Until phase I studies are completed, human pharmacokinetics must be predicted from animal models. Microdosing is an approach that may make human clinical research studies more effective, predictable, and quicker. The microdosing strategy allows for early assessment of human pharmacokinetics and the bioavailability of a drug candidate. Microdosing studies are designed to evaluate pharmacokinetics or imaging of specific targets without inducing pharmacologic effects. According to the FDA guidelines, a microdose is defined as less than 1/100th of a test substance dose calculated (based on animal data) to yield a pharmacologic effect, with the maximum dose being ≤100 micrograms.1 Identifying unique and major human metabolites early in the drug development process allows for the timely assessment of potential safety issues. The current technique for evaluating microdosing studies is accelerator mass spectrometry (AMS). AMS is an ultra-sensitive technique where the lower limit of quantitation is in the low fg/mL range. However, this technique has many drawbacks including the high cost of routine use due to the required synthesis of radio-labeled compounds. It has been shown2-4 that conventional liquid chromatography/tandem mass 64 RUO-MKT-01-1583-A QTRAP 5500 system advantages: LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. • Smaller, bench-top-sized footprint • Made for routine use • Easy-to-use • Radio-labeling not required • High throughput • Increased QqQ and LIT sensitivity Metabolic stability studies Figure 2: Tabulation of all found metabolites, in either single or double injection workflows. The QTRAP® 5500 System shows far greater detection efficiency than high-resolution MDF two-injection workflows. spectrometry (LC/MS/MS) has sufficient sensitivity to support quantification of parent drug in human microdosing studies. Comprehensive detection and confirmation of metabolites can be achieved at microdosing levels in a single injection using LC/MS/MS. In this work, metabolites from microdoses of omeprazole were detected and confirmed in rat plasma using a QTRAP 5500 system. In this study, three jugular vein-cannulated Sprague-Dawley rats were dosed via oral gavages at 1.67 μg/kg of omeprazole. Plasma was taken at 20, 40, 60, 80, and 120 min from each rat. A pMRM method was generated with 129 MRM transitions with a pause time of 5 msec and dwell time of 5 msec per transition. MS/MS data was collected automatically via IDA at a scan rate of 10,000 Da/sec. The overall duty cycle time for this method was approximately 2.6 sec. This application is discussed in detail in a technical note titled, “Detection and Confirmation of Metabolites at Microdosing Levels Using the New QTRAP 5500 LC/MS/MS System.” Figures 3 and 4 highlight how improved sensitivity of the QTRAP 5500 system has bolstered for this sensitivity-challenged application area. To summarize, the sensitivity and speed improvements in the QTRAP 5500 system have made it the ideal tool for microdosing studies, in particular for finding and identifying metabolites at microdosing levels. This system and workflow has significant advantages over accelerator mass spectrometry (AMS) technologies. While AMS has been traditionally associated with microdosing studies because the high sensitivity levels achieved, there are some drawbacks to the technology, which are reviewed below: • Very hardware-intense technology that requires highly qualified staff to keep it running. • Preparation of radio-labeled compounds is time consuming and expensive. • This technique and it can only evaluate a very limited number of analytes at once. • The turnaround time from sample preparation to acquisition to results is very slow. The metabolic stability of a drug candidate impacts its bioavailability and half-life in vivo and should be evaluated early in the drug discovery and development process. In vitro experiments using liver microsomes or hepatocytes provide valuable insights into metabolic stability and aid in the selection of drug candidates with favorable pharmacokinetic properties. The identity of the metabolites formed during parent drug metabolism is also important. Collecting both qualitative (metabolite ID) and quantitative (metabolic stability, PK, pharmacodynamic) data at the same time can improve the efficiency within the drug discovery and development process so that critical data can be obtained faster. Figure 3: A minor metabolite (tri-oxidation + glucuronidation) of omeprazole was detected and confirmed using LightSight® Software. Figure 5 shows hundreds of predicted metabolites that were monitored using MRM transitions, and the metabolic stability of the parent drug was also monitored by following an MRM transition over a series of time points. The quantitative results required to determine the half-life while at the same time monitor the presence of both major and minor metabolites were monitored at the same time with confirmatory MS/MS all in the same automated experiment. GSH-conjugate screening Screening for reactive metabolites of drug candidates using GSH trapping is an important part of early safety assessment in pharmaceutical discovery and development. Due to the toxicity of reactive metabolites, detection of these species even at trace levels can be relevant in the optimal design of a therapeutic drug. Current low- and high-resolution single-MS-based detection often offers insufficient sensitivity for the detection and confirmation of GSH-conjugates. The QTRAP 5500 system is the first LC/MS system that can combine the most powerful modes of GSH metabolite detection into a single injection workflow with sufficient sensitivity to ensure accurate results. A series of groundbreaking workflows with optimized of scan speeds, quadrupole MS/MS sensitivity, rapid positive/negative switching, and unprecedented LIT MS/MS confirmation sensitivity will be presented in this application overview. Drug-induced idiosyncratic hepatotoxicity is a concern to pharmaceutical companies, especially because these drugs are often missed in pre-clinical safety assessments and clinical research studies, probably due to the low level of reactive Figure 4: MS/MS of a minor metabolite (tri-oxidation + glucuronidation) of omeprazole is displayed; m/z 394 is a diagnostic fragment for this metabolite. metabolite formation. Reactive metabolites are capable of covalent modification of proteins or nucleic acids through nucleophilic substitution. These types of reactions may contribute to idiosyncratic hepatotoxicity due to the interruption of certain cellular processes. The process and causes of idiosyncratic drug hepatotoxicity is not fully understood. However, the formation of reactive metabolites appears to be associated with various toxicological events. The presence of GSH-conjugated metabolites is an indication of the formation of reactive metabolites and is thus critical to identify and monitor in metabolite profiling studies within the drug discovery and development process. None of these issues are apparent with the QTRAP 5500 system. DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 65 In this work, a set of verapamil in vitro hepatocyte metabolites were spiked into bile matrix at a low level (2.5 μM) to mimic the in vivo sample matrix. LightSight® Software 2.1 has allowed for a new novel survey mode called pMRM (predictive MRM). This new type of IDA experiment can be created automatically using LightSight Software 2.1 and loaded directly onto the QTRAP system for rapid analysis. The advantage of pMRM is the improved signal-to-noise ratio, IDA selectivity, and efficiency compared to single MS strategies. The QTRAP 5500 system with pMRM survey mode as well as dual precursor and neutral loss scan mode was evaluated against a two injection mass defect filter (MDF) high-resolution experiment “Breakthrough in vivo metabolites analysis using the QTRAP 5500 system and Predictive MRM”. Using the targeted pMRM approach, 158 transitions were created by LightSight Software 2.1 to search for phase I and II metabolites. A total of 1.5 sec was the total duty cycle for the method that included automated collection of MS/MS at 20,000 amu/sec. When employing the multiple precursor ion and neutral loss approach, the overall duty cycle was 2.2 sec. Both workflows had cycle times well-suited for high-throughput chromatographic analyses. To summarize, even at higher-throughput chromatographic conditions (by a factor of 2), the QTRAP 5500 system found and identified with confirmatory MS/MS, 7-fold more metabolites using a single injection workflow than the high-resolution approach. With the addition of a second injection, the QTRAP 5500 system still managed to top the high-resolution experiments, finding 2-fold more metabolites (Figure 2). Microdosing studies Many new chemical entities fail in phase I clinical research studies due to poor pharmacokinetics. Until phase I studies are completed, human pharmacokinetics must be predicted from animal models. Microdosing is an approach that may make human clinical research studies more effective, predictable, and quicker. The microdosing strategy allows for early assessment of human pharmacokinetics and the bioavailability of a drug candidate. Microdosing studies are designed to evaluate pharmacokinetics or imaging of specific targets without inducing pharmacologic effects. According to the FDA guidelines, a microdose is defined as less than 1/100th of a test substance dose calculated (based on animal data) to yield a pharmacologic effect, with the maximum dose being ≤100 micrograms.1 Identifying unique and major human metabolites early in the drug development process allows for the timely assessment of potential safety issues. The current technique for evaluating microdosing studies is accelerator mass spectrometry (AMS). AMS is an ultra-sensitive technique where the lower limit of quantitation is in the low fg/mL range. However, this technique has many drawbacks including the high cost of routine use due to the required synthesis of radio-labeled compounds. It has been shown2-4 that conventional liquid chromatography/tandem mass 64 RUO-MKT-01-1583-A QTRAP 5500 system advantages: LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. • Smaller, bench-top-sized footprint • Made for routine use • Easy-to-use • Radio-labeling not required • High throughput • Increased QqQ and LIT sensitivity Metabolic stability studies Figure 2: Tabulation of all found metabolites, in either single or double injection workflows. The QTRAP® 5500 System shows far greater detection efficiency than high-resolution MDF two-injection workflows. spectrometry (LC/MS/MS) has sufficient sensitivity to support quantification of parent drug in human microdosing studies. Comprehensive detection and confirmation of metabolites can be achieved at microdosing levels in a single injection using LC/MS/MS. In this work, metabolites from microdoses of omeprazole were detected and confirmed in rat plasma using a QTRAP 5500 system. In this study, three jugular vein-cannulated Sprague-Dawley rats were dosed via oral gavages at 1.67 μg/kg of omeprazole. Plasma was taken at 20, 40, 60, 80, and 120 min from each rat. A pMRM method was generated with 129 MRM transitions with a pause time of 5 msec and dwell time of 5 msec per transition. MS/MS data was collected automatically via IDA at a scan rate of 10,000 Da/sec. The overall duty cycle time for this method was approximately 2.6 sec. This application is discussed in detail in a technical note titled, “Detection and Confirmation of Metabolites at Microdosing Levels Using the New QTRAP 5500 LC/MS/MS System.” Figures 3 and 4 highlight how improved sensitivity of the QTRAP 5500 system has bolstered for this sensitivity-challenged application area. To summarize, the sensitivity and speed improvements in the QTRAP 5500 system have made it the ideal tool for microdosing studies, in particular for finding and identifying metabolites at microdosing levels. This system and workflow has significant advantages over accelerator mass spectrometry (AMS) technologies. While AMS has been traditionally associated with microdosing studies because the high sensitivity levels achieved, there are some drawbacks to the technology, which are reviewed below: • Very hardware-intense technology that requires highly qualified staff to keep it running. • Preparation of radio-labeled compounds is time consuming and expensive. • This technique and it can only evaluate a very limited number of analytes at once. • The turnaround time from sample preparation to acquisition to results is very slow. The metabolic stability of a drug candidate impacts its bioavailability and half-life in vivo and should be evaluated early in the drug discovery and development process. In vitro experiments using liver microsomes or hepatocytes provide valuable insights into metabolic stability and aid in the selection of drug candidates with favorable pharmacokinetic properties. The identity of the metabolites formed during parent drug metabolism is also important. Collecting both qualitative (metabolite ID) and quantitative (metabolic stability, PK, pharmacodynamic) data at the same time can improve the efficiency within the drug discovery and development process so that critical data can be obtained faster. Figure 3: A minor metabolite (tri-oxidation + glucuronidation) of omeprazole was detected and confirmed using LightSight® Software. Figure 5 shows hundreds of predicted metabolites that were monitored using MRM transitions, and the metabolic stability of the parent drug was also monitored by following an MRM transition over a series of time points. The quantitative results required to determine the half-life while at the same time monitor the presence of both major and minor metabolites were monitored at the same time with confirmatory MS/MS all in the same automated experiment. GSH-conjugate screening Screening for reactive metabolites of drug candidates using GSH trapping is an important part of early safety assessment in pharmaceutical discovery and development. Due to the toxicity of reactive metabolites, detection of these species even at trace levels can be relevant in the optimal design of a therapeutic drug. Current low- and high-resolution single-MS-based detection often offers insufficient sensitivity for the detection and confirmation of GSH-conjugates. The QTRAP 5500 system is the first LC/MS system that can combine the most powerful modes of GSH metabolite detection into a single injection workflow with sufficient sensitivity to ensure accurate results. A series of groundbreaking workflows with optimized of scan speeds, quadrupole MS/MS sensitivity, rapid positive/negative switching, and unprecedented LIT MS/MS confirmation sensitivity will be presented in this application overview. Drug-induced idiosyncratic hepatotoxicity is a concern to pharmaceutical companies, especially because these drugs are often missed in pre-clinical safety assessments and clinical research studies, probably due to the low level of reactive Figure 4: MS/MS of a minor metabolite (tri-oxidation + glucuronidation) of omeprazole is displayed; m/z 394 is a diagnostic fragment for this metabolite. metabolite formation. Reactive metabolites are capable of covalent modification of proteins or nucleic acids through nucleophilic substitution. These types of reactions may contribute to idiosyncratic hepatotoxicity due to the interruption of certain cellular processes. The process and causes of idiosyncratic drug hepatotoxicity is not fully understood. However, the formation of reactive metabolites appears to be associated with various toxicological events. The presence of GSH-conjugated metabolites is an indication of the formation of reactive metabolites and is thus critical to identify and monitor in metabolite profiling studies within the drug discovery and development process. None of these issues are apparent with the QTRAP 5500 system. DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 65 Here, data was acquired at the fastest quadrupole scan rate of 2,000 Da/sec for the precursor and neutral loss experiments. The LIT experiments were acquired at a scan rate of 20,000 Da/sec with a fixed fill time of 35 msec and Q0 trapping to increase duty cycle. A pos/neg switching time of 50 ms was used for the two requisite polarity changes needed for this experiment. All data was processed in LightSight Software 2.1 and the ACD Fragmenter/Specmanager™ to find and assign major metabolites. Figure 6 shows the XIC trace for precursor ion m/z 272 (-) and neutral loss m/z 129 (+) IDA, and the major GSH metabolites are shaded in red. A more detailed discussion can be found in reference 5. The QTRAP 5500 system showed a significant improvement in the sensitive detection and confirmation of GSH metabolites during single injection experiment with high-throughput chromatographic separation, a proces amenable to early discovery screening. Furthermore, the improved sensitivity in LIT mode on the QTRAP 5500 system provides superior fragmentation patterns and spectrum confirmation over those achieved on less sensitive accurate mass instruments. The additional dynamic range allows for a more complete picture of GSH metabolism to be developed for a drug of interest. Summary Highlights The QTRAP 5500 system provides unique workflow possibilities as well as enhanced speed, sensitivity, and selectivity. The hybrid technology combines quadrupole scan functions for finding potential metabolites with high-sensitivity LIT scans (for identifying and confirming metabolites. The pMRM approach screens for hundreds of potential metabolites and triggers the collection of full-scan MS/MS permitting metabolite identification in a single analysis. Multiple precursor ion and/or neutral loss combinations, together with polarity switching, allows for significantly better workflow strategies for reactive metabolite identification in a single analysis. With improvements in speed and sensitivity, much better peak definition is achieved at lower levels, leading to better identification, even at very high chromatographic throughput. Next-generation metabolite prediction using LightSight Software is a perfect complement to instrument capabilities, enabling even novice metabolite investigators to achieve complete results easily. • • • • • • • • References 1 2 66 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com Food and Drug Administration. Guidance for Industry, investigators, and reviewers – exploratory IND studies. January 2006. S.K. Balani, et al., “Evaluation of Microdosing to Assess Pharmacokinetic Linearity in Rats using Liquid Chromatography-Tandem Mass Spectrometry,” Drug Metab and Dis-pos. 2006; 34: 384-388. 3 M. McLean, et al., “Accelerating Drug Development: Methodology to Support First-in-Man Pharmacokinetic Studies by the Using of Drug Candidate Microdosing,” Drug Dev Res. 2007; 68: 14-22. 4 J. Ni, et al., “Microdosing Assessment to Evaluate Pharmacokinetics and Drug Metabolism in Rats using Liquid Chromatography-Tandem Mass Spectrometry,” Pharm Res. 2008. 4 Figure 5: Nefazodone was incubated in rat liver microsomes for 0, 5, 15, and 30 min. The half-life was determined by monitoring the parent drug as well as major and minor metabolites in the same analysis; metabolites were detected, identified and confirmed together in the same run. A) Parent drug B) metabolic stability of parent drug, C) oxidative metabolite, D) MSMS of oxidative metabolite. Improved quadrupole sensitivity (MRM response and signal-to-noise ratios, parent ion and neutral loss) Improved sensitivity in trap modes (10-100-fold enhancement in EPI scans) Faster QqQ scanning (4-fold) Faster polarity switching (50 msec) Faster LIT scanning (20,000 amu/sec) Shorter fill-time capabilities (50 μsec). Unique workflows ideal for metabolite detection • pMRM • Multiple full-scan survey possibilities (precursor ion, neutral loss, polarity switching) Qualitative and quantitative information together LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. “GSH Conjugate Screening with the QTRAP 5500 LC/MS/MS system.” Figure 6: The extracted ion chromatogram (XIC) trace of +/- neutral loss/parent ion information-dependent acquisition of diclofenac enables the analysis of GSH metabolites GSH analysis, using a short high pressure gradient. www.absciex.com DRUG METABOLISM 67 Here, data was acquired at the fastest quadrupole scan rate of 2,000 Da/sec for the precursor and neutral loss experiments. The LIT experiments were acquired at a scan rate of 20,000 Da/sec with a fixed fill time of 35 msec and Q0 trapping to increase duty cycle. A pos/neg switching time of 50 ms was used for the two requisite polarity changes needed for this experiment. All data was processed in LightSight Software 2.1 and the ACD Fragmenter/Specmanager™ to find and assign major metabolites. Figure 6 shows the XIC trace for precursor ion m/z 272 (-) and neutral loss m/z 129 (+) IDA, and the major GSH metabolites are shaded in red. A more detailed discussion can be found in reference 5. The QTRAP 5500 system showed a significant improvement in the sensitive detection and confirmation of GSH metabolites during single injection experiment with high-throughput chromatographic separation, a proces amenable to early discovery screening. Furthermore, the improved sensitivity in LIT mode on the QTRAP 5500 system provides superior fragmentation patterns and spectrum confirmation over those achieved on less sensitive accurate mass instruments. The additional dynamic range allows for a more complete picture of GSH metabolism to be developed for a drug of interest. Summary Highlights The QTRAP 5500 system provides unique workflow possibilities as well as enhanced speed, sensitivity, and selectivity. The hybrid technology combines quadrupole scan functions for finding potential metabolites with high-sensitivity LIT scans (for identifying and confirming metabolites. The pMRM approach screens for hundreds of potential metabolites and triggers the collection of full-scan MS/MS permitting metabolite identification in a single analysis. Multiple precursor ion and/or neutral loss combinations, together with polarity switching, allows for significantly better workflow strategies for reactive metabolite identification in a single analysis. With improvements in speed and sensitivity, much better peak definition is achieved at lower levels, leading to better identification, even at very high chromatographic throughput. Next-generation metabolite prediction using LightSight Software is a perfect complement to instrument capabilities, enabling even novice metabolite investigators to achieve complete results easily. • • • • • • • • References 1 2 66 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com Food and Drug Administration. Guidance for Industry, investigators, and reviewers – exploratory IND studies. January 2006. S.K. Balani, et al., “Evaluation of Microdosing to Assess Pharmacokinetic Linearity in Rats using Liquid Chromatography-Tandem Mass Spectrometry,” Drug Metab and Dis-pos. 2006; 34: 384-388. 3 M. McLean, et al., “Accelerating Drug Development: Methodology to Support First-in-Man Pharmacokinetic Studies by the Using of Drug Candidate Microdosing,” Drug Dev Res. 2007; 68: 14-22. 4 J. Ni, et al., “Microdosing Assessment to Evaluate Pharmacokinetics and Drug Metabolism in Rats using Liquid Chromatography-Tandem Mass Spectrometry,” Pharm Res. 2008. 4 Figure 5: Nefazodone was incubated in rat liver microsomes for 0, 5, 15, and 30 min. The half-life was determined by monitoring the parent drug as well as major and minor metabolites in the same analysis; metabolites were detected, identified and confirmed together in the same run. A) Parent drug B) metabolic stability of parent drug, C) oxidative metabolite, D) MSMS of oxidative metabolite. Improved quadrupole sensitivity (MRM response and signal-to-noise ratios, parent ion and neutral loss) Improved sensitivity in trap modes (10-100-fold enhancement in EPI scans) Faster QqQ scanning (4-fold) Faster polarity switching (50 msec) Faster LIT scanning (20,000 amu/sec) Shorter fill-time capabilities (50 μsec). Unique workflows ideal for metabolite detection • pMRM • Multiple full-scan survey possibilities (precursor ion, neutral loss, polarity switching) Qualitative and quantitative information together LATE STAGE DISCOVERY LATE STAGE DISCOVERY For Research Use Only. Not for use in diagnostic procedures. “GSH Conjugate Screening with the QTRAP 5500 LC/MS/MS system.” Figure 6: The extracted ion chromatogram (XIC) trace of +/- neutral loss/parent ion information-dependent acquisition of diclofenac enables the analysis of GSH metabolites GSH analysis, using a short high pressure gradient. www.absciex.com DRUG METABOLISM 67 Simultaneous Metabolite Identification and Quantitation Using UV Data Integration and LightSight® Software 2.2 Experimental conditions Alek N. Dooley, Carmai Seto, Hesham Ghobarah, and Elliott B. Jones Bromocriptine eluted when the gradient was approximately 60% solution A, to ensure the separation of all metabolites, whether of increased or decreased polarity. A sample (5 μL) was injected onto a Phenonomenex HPLC column (50 × 2 mm). Prior to mass spectrometric analysis, the LC eluant was directed through a Shimadzu SDP20AV dual wavelength detector set to 302 nm. Predictive multiple reaction monitoring with informationdependent acquisition (pMRM-IDA) methods were created using LightSight Software 2.2 and run on a 4000 QTRAP system, a hybrid triple quadrupole/linear ion trap mass spectrometer. With prior knowledge of the parent drug and its fragmentation pathway, theoretical MRM transitions for metabolites can be determined based on a particular biotransformation set. The pMRM experiments were built using the phase I comprehensive biotransformation set provided by the LightSight Software. A total of 166 MRM transitions were included in the pMRM survey scan linked to a single enhanced product ion (EPI) dependent scan. The EPI experiment was acquired at a scan rate of 4,000 Da/sec with dynamic fill time turned on. Structural elucidation of the MS/MS fragments was achieved with the assistance of the ACD/Labs MS Fragmenter software package. AB SCIEX, Redwood City, California and Concord, Ontario, Canada Key scientific challenges in quant/qual workflows • • Accurate quantification – The mass spectrometric response for the same molar concentration can vary greatly by compound. Quantifying a compound using either MS or UV data can be quite useful to avoid over- or underestimating the amount of a metabolite. Accurate assignment of the metabolic site – Most workflows require the use of multiple software packages to identify, elucidate, and quantify the metabolites. Key benefits of the LightSight Software 2.2 for quant/qual workflows ® • • Ease-of-use – LightSight Software can be used to process qualitative and quantitative data simultaneously. Increased throughput – Integrated tools for structural elucidation rapidly facilitate assigning sites of metabolism. Key features of the LightSight Software 2.2 for quant/qual workflows • • One comprehensive software package – Quantitative and qualitative data are easily and automatically generated using LightSight Software. Correlation – The software provides peak areas from both the analog (UV, diode array, or radioactivity) detector and the mass spectrometric data. Overview Integration of UV and MS data is highly desirable for biotransformation studies. For example, in vitro metabolic stability studies can provide qualitative metabolism data and kinetics for the parent compound. Relative quantitation of metabolites detected by integrating UV data is a very useful workflow; However manual processing and correlation of both data streams is time-consuming. Here, qualitative and quantitative data are collected by acquiring both mass spectrometric and UV data in the same experiment, and then both data types are processed and correlated automatically in LightSight Software 2.2. Bromocriptine was incubated at 50 μM in rabbit liver microsomes using an NADPH regenerating system under oxidative conditions at 37 °C. After 1 hr, the incubation was quenched with an equal volume of acetonitrile. The supernatant was then diluted 1:5 with water prior to injection, giving a final bromocriptine concentration of 5 μM in the control. Results and discussion Introduction Drug metabolism studies often estimate the relative abundances of metabolites alongside the elucidation of metabolite structures and the assignment of sites of metabolism. While MS-based detection is a powerful technique for metabolite identification and structure elucidation, small changes in metabolite structure and functional groups can often lead to major differences in ionization efficiency and fragmentation. Therefore, estimating metabolite concentrations based on the MS signal alone can be difficult due to the absence of a reference standards for quantitation. By comparison, UV absorbance is considered to provide a more uniform response and allows for estimation of the relative abundances of metabolites. HIGH RESOLUTION ACCURATE MASS DEFINITIVE METABOLITE ID For Research Use Only. Not for use in diagnostic procedures. Figure 1 shows the molecular structure of bromocriptine with sites of fragmentation that occur during MS/MS analysis indicated along with the nominal mass of the fragment. The m/z 346 fragment ion was chosen as the Q3 mass for the pMRM method. Compounds with higher λmax, above the typical solvent noise that exists from 100 to 150 nm, typically yield excellent quality data that correlates nicely with MRM data. Bromocriptine, with a λmax of 302 nm, exhibits a maximum UV absorbance that has few interferences with competing background. Figure 1: Structure and fragmentation of bromocriptine with the m/z ratio for each fragment shown at the site of cleavage. If there is complementary analog data (e.g. PDA, UV, ADC) available, LightSight Software will correlate the MS data to the analog data and vice versa. The table potential metabolites contains detected metabolites and both data types to ensure that all potential metabolites are found even if a metabolite exhibits only one type of signal. Using the LightSight Software, a total of 14 metabolites were detected and identified, and, of those 14, 11 were oxidative metabolites of bromocriptine. A summary of the MS and UV data for these metabolites is given Table 1. Figure 2 shows the correlation between the MS data and the UV trace for bromocriptine and its metabolites prepared using LightSight Software. The metabolites included single, di-, and tri-oxidations of bromocriptine. The proposed structures of some metabolites are given in Figure 3. Figure 4 shows the comparison between a tri-oxidation metabolite of bromocriptine and the parent bromocriptine MS/MS spectra. The pMRM transition that corresponds to the triple-oxidation metabolite shows four clear chromatographic peaks, the presence of which were further confirmed by the UV trace (Figure 5). Integration of MS and analog data processing is highly desirable because drug metabolism scientists can then perform structure elucidation based on MS/MS data, estimate concentration based on UV data, and then generate one integrated report. This technical note describes the use of LightSight Software 2.2 to study the in vitro metabolism of bromocriptine using a 4000 QTRAP® LC/MS/MS System in combination with integrated UV detection, data processing, and reporting. Table 1: Metabolites were identified using LightSight® Software sorted by retention time. 68 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 69 Simultaneous Metabolite Identification and Quantitation Using UV Data Integration and LightSight® Software 2.2 Experimental conditions Alek N. Dooley, Carmai Seto, Hesham Ghobarah, and Elliott B. Jones Bromocriptine eluted when the gradient was approximately 60% solution A, to ensure the separation of all metabolites, whether of increased or decreased polarity. A sample (5 μL) was injected onto a Phenonomenex HPLC column (50 × 2 mm). Prior to mass spectrometric analysis, the LC eluant was directed through a Shimadzu SDP20AV dual wavelength detector set to 302 nm. Predictive multiple reaction monitoring with informationdependent acquisition (pMRM-IDA) methods were created using LightSight Software 2.2 and run on a 4000 QTRAP system, a hybrid triple quadrupole/linear ion trap mass spectrometer. With prior knowledge of the parent drug and its fragmentation pathway, theoretical MRM transitions for metabolites can be determined based on a particular biotransformation set. The pMRM experiments were built using the phase I comprehensive biotransformation set provided by the LightSight Software. A total of 166 MRM transitions were included in the pMRM survey scan linked to a single enhanced product ion (EPI) dependent scan. The EPI experiment was acquired at a scan rate of 4,000 Da/sec with dynamic fill time turned on. Structural elucidation of the MS/MS fragments was achieved with the assistance of the ACD/Labs MS Fragmenter software package. AB SCIEX, Redwood City, California and Concord, Ontario, Canada Key scientific challenges in quant/qual workflows • • Accurate quantification – The mass spectrometric response for the same molar concentration can vary greatly by compound. Quantifying a compound using either MS or UV data can be quite useful to avoid over- or underestimating the amount of a metabolite. Accurate assignment of the metabolic site – Most workflows require the use of multiple software packages to identify, elucidate, and quantify the metabolites. Key benefits of the LightSight Software 2.2 for quant/qual workflows ® • • Ease-of-use – LightSight Software can be used to process qualitative and quantitative data simultaneously. Increased throughput – Integrated tools for structural elucidation rapidly facilitate assigning sites of metabolism. Key features of the LightSight Software 2.2 for quant/qual workflows • • One comprehensive software package – Quantitative and qualitative data are easily and automatically generated using LightSight Software. Correlation – The software provides peak areas from both the analog (UV, diode array, or radioactivity) detector and the mass spectrometric data. Overview Integration of UV and MS data is highly desirable for biotransformation studies. For example, in vitro metabolic stability studies can provide qualitative metabolism data and kinetics for the parent compound. Relative quantitation of metabolites detected by integrating UV data is a very useful workflow; However manual processing and correlation of both data streams is time-consuming. Here, qualitative and quantitative data are collected by acquiring both mass spectrometric and UV data in the same experiment, and then both data types are processed and correlated automatically in LightSight Software 2.2. Bromocriptine was incubated at 50 μM in rabbit liver microsomes using an NADPH regenerating system under oxidative conditions at 37 °C. After 1 hr, the incubation was quenched with an equal volume of acetonitrile. The supernatant was then diluted 1:5 with water prior to injection, giving a final bromocriptine concentration of 5 μM in the control. Results and discussion Introduction Drug metabolism studies often estimate the relative abundances of metabolites alongside the elucidation of metabolite structures and the assignment of sites of metabolism. While MS-based detection is a powerful technique for metabolite identification and structure elucidation, small changes in metabolite structure and functional groups can often lead to major differences in ionization efficiency and fragmentation. Therefore, estimating metabolite concentrations based on the MS signal alone can be difficult due to the absence of a reference standards for quantitation. By comparison, UV absorbance is considered to provide a more uniform response and allows for estimation of the relative abundances of metabolites. HIGH RESOLUTION ACCURATE MASS DEFINITIVE METABOLITE ID For Research Use Only. Not for use in diagnostic procedures. Figure 1 shows the molecular structure of bromocriptine with sites of fragmentation that occur during MS/MS analysis indicated along with the nominal mass of the fragment. The m/z 346 fragment ion was chosen as the Q3 mass for the pMRM method. Compounds with higher λmax, above the typical solvent noise that exists from 100 to 150 nm, typically yield excellent quality data that correlates nicely with MRM data. Bromocriptine, with a λmax of 302 nm, exhibits a maximum UV absorbance that has few interferences with competing background. Figure 1: Structure and fragmentation of bromocriptine with the m/z ratio for each fragment shown at the site of cleavage. If there is complementary analog data (e.g. PDA, UV, ADC) available, LightSight Software will correlate the MS data to the analog data and vice versa. The table potential metabolites contains detected metabolites and both data types to ensure that all potential metabolites are found even if a metabolite exhibits only one type of signal. Using the LightSight Software, a total of 14 metabolites were detected and identified, and, of those 14, 11 were oxidative metabolites of bromocriptine. A summary of the MS and UV data for these metabolites is given Table 1. Figure 2 shows the correlation between the MS data and the UV trace for bromocriptine and its metabolites prepared using LightSight Software. The metabolites included single, di-, and tri-oxidations of bromocriptine. The proposed structures of some metabolites are given in Figure 3. Figure 4 shows the comparison between a tri-oxidation metabolite of bromocriptine and the parent bromocriptine MS/MS spectra. The pMRM transition that corresponds to the triple-oxidation metabolite shows four clear chromatographic peaks, the presence of which were further confirmed by the UV trace (Figure 5). Integration of MS and analog data processing is highly desirable because drug metabolism scientists can then perform structure elucidation based on MS/MS data, estimate concentration based on UV data, and then generate one integrated report. This technical note describes the use of LightSight Software 2.2 to study the in vitro metabolism of bromocriptine using a 4000 QTRAP® LC/MS/MS System in combination with integrated UV detection, data processing, and reporting. Table 1: Metabolites were identified using LightSight® Software sorted by retention time. 68 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 69 HIGH RESOLUTION ACCURATE MASS DEFINITIVE METABOLITE ID For Research Use Only. Not for use in diagnostic procedures. Figure 4: Proposed structures are shown for metabolites of bromocriptine detected in the microsomal incubation. Conclusions Figure 2: UV and MS data were correlated in the Processing Workspace of LightSight® Software 2.2. LightSight Software 2.2 efficiently identifies, confirms, and quantitates metabolites using both MS and analog data. LightSight Software provides both structure elucidation based on the MS/MS data and estimated concentration based on UV in a single analysis. Figure 5: The extracted ion chromatogram (XIC) for the di-oxidation metabolites of bromocriptine (top) and the UV chromatogram (bottom). Figure 3: Comparison of parent and metabolite MS/MS spectra. The MS/MS spectrum for bromocriptine (bottom panel) and a di-oxidation metabolite reveals that all the fragments at or below m/z 426.1 are common between both spectra, indicating that the sites of metabolism do not occur on these portions of the parent drug. 70 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 71 HIGH RESOLUTION ACCURATE MASS DEFINITIVE METABOLITE ID For Research Use Only. Not for use in diagnostic procedures. Figure 4: Proposed structures are shown for metabolites of bromocriptine detected in the microsomal incubation. Conclusions Figure 2: UV and MS data were correlated in the Processing Workspace of LightSight® Software 2.2. LightSight Software 2.2 efficiently identifies, confirms, and quantitates metabolites using both MS and analog data. LightSight Software provides both structure elucidation based on the MS/MS data and estimated concentration based on UV in a single analysis. Figure 5: The extracted ion chromatogram (XIC) for the di-oxidation metabolites of bromocriptine (top) and the UV chromatogram (bottom). Figure 3: Comparison of parent and metabolite MS/MS spectra. The MS/MS spectrum for bromocriptine (bottom panel) and a di-oxidation metabolite reveals that all the fragments at or below m/z 426.1 are common between both spectra, indicating that the sites of metabolism do not occur on these portions of the parent drug. 70 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 71 Differential Mobility Spectrometry for Quantitative and Qualitative Applications in Pharmaceutical Workflows Using SelexION™ Differential Mobility Separation Technology to detect metabolites with better selectivity in complex mixtures on the AB SCIEX TripleTOF® 5600+ LC/MS/MS System James A. Ferguson, PhD. AB SCIEX, 500 Old Connecticut Path, Framingham, MA 01701 Key scientific challenges of metabolite assays • • • • • Materials and methods There are many sources of interfering compounds that can complicate the isolation of metabolites during a pharmacokinetics study. Some interfering compounds come from mobile phases and columns, some from the sample matrix and, depending on the chromatography, some isobars can behave like interferences. Early ADME studies require high throughput and faster chromatography, which can, in turn, could lead to coeluting peaks, especially for very similar compounds such as the positional isomers that can occur from oxidative metabolites or multiple demethylations (e.g. verapamil). The goal of this study was to show separation of these structural isomers that cannot be separated on any existing mass spectrometer. DMS separations were demonstrated for three oxidation metabolites of carbamazepine and two demethylation metabolites of verapamil (Figure 1). Sample preparation The data showing the separation of structural isomers (not shown), indicate that the majority of the compounds moved further into “negative COV space” as the separation voltages (SV) increased (see Figure 2 for a schematic and operational overview of the DMS cell). Nefazodone moved to positive COV space during DMS transmission optimization runs. Because of this additional level of separation provided by DMS, the carbamazepine samples incubated with rat liver microsomes were assessed for metabolites with improved signal-to-noise ratios. The incubations were analyzed while ramping COV; when optimal DMS conditions were found, the samples were re-analyzed using the best SV/COV combination. The DMS-based chromatogram was nearly identical to the extracted ion chromatograms of known metabolites and showed higher signal for some metabolites. This preliminary work shows some promise for finding metabolites in complex matrices. Sensitivity – in vivo studies, due to the biological matrices involved, can negatively impact sensitivity and selectivity. Limited quantitation range – Lowered sensitivity combined with often poor selectivity can compromise the desired lower limits of quantitation (LLOQ). Limitations in selectivity – In both in vivo and in vitro studies, matrix ions can be the most abundant ions in the spectra. Pulling out relevant metabolites rom the background noise can be a challenge on some systems. Key benefits of differential mobility separation (DMS) for metabolite assays • Introduction Enhanced LLOQs – For cases where background noise limits the LOQ, DMS provides an additional level of selectivity, orthogonal to the mass spectrometer and LC system. Better sensitivity – Selectivity gains from DMS permit less involved sample preparations and/or faster gradients, allowing for overall improvements in sensitivity and throughput due to more efficient extractions and better recovery. Selectivity improvements overcome sensitivity losses – DMS is often accompanied by a loss in absolute sensitivity, but the gains in selectivity improve the potential for real gains in LLOQ. Key features of SelexION™ Differential Mobility Separation Technology for metabolite assays • • • • 72 Separation of diverse species reduces baseline noise – SelexION technology separates isobaric species (true iso-bars in this tech note), and charge state interferences to reduce background levels and achieve better selectivity and LOQs – while retaining compatibility with UHPLC speeds. Simple installation and maintenance – DMS is truly orthogonal to LC and MSMS; installs in minutes with no tools required and no need to break vacuum. Device maintenance is minimal and very straightforward. Shortened assay times – SelexION technology can potentially reduce chromatographic run times. Efficient separation process – Planar geometry results in short residence times, high speeds, and minimal diffusion losses for maximum sensitivity and UHPLC compatibility. RUO-MKT-01-1583-A Synthetic oxidative metabolites of carbamazepine (CBZ) and two verapamil demethylations (Figure 1) from Toronto Research Chemicals were used to illustrate DMS separation of isobaric metabolites (structural isomers). To test whether DMS could minimize background, nefazodone was incubated at 10 µM with rat liver microsomes. The samples were then quenched with ACN, lyophilized, and reconstituted in 5% ACN/H2O for analysis. Chromatography Pumps and autosampler: Prominence series from Shimadzu Column: Waters Acquity BEH C18 (2.1 × 100 mm), 1.7 µm Column temp.: 40 °C Injection: 5 μL Flow rate: Programmed (see gradients below) Mobile phase: A) 0.1% formic acid B) Acetonitrile with 0.1% formic acid HIGH RESOLUTION ACCURATE MASS DEFINITIVE METABOLITE ID For Research Use Only. Not for use in diagnostic procedures. Gradient for the quantitative study: Time (min) %A %B Flow (mL/min) 0.00 97.5 2.5 0.50 0.25 97.5 2.5 — 1.00 20.0 80.0 0.65 1.10 2.5 97.5 — 1.35 2.5 97.5 — 1.40 97.5 2.5 0.65 1.60 97.5 2.5 0.50 2.00 97.5 2.5 END Gradient for the qualitative (metabolite ID) study: Time (min) %A %B • • Furthering selectivity with chemical modifiers – Introducing chemical modifiers to the SelexION device cell allows for amplification of the separation capacity and adds a new dimension of selectivity. Ruggedness – SelexION technology provides ruggedness and stability to enable high performance quantitative bioanalysis. DRUG METABOLISM www.absciex.com Figure 1: Structural Isomers used in this study are shown here. A = 2-hydroxycarbamazepine, B = 3-hydroxycarbamazepine, C = 10,11 carbamazepine epoxide; all metabolites have the molecular formula C15H12N2O2. D = norverapamil, and E = p-O-desmethylverapamil; both metabolites have the formula C26H36N2O4. www.absciex.com Flow (mL/min) 0.00 97.5 2.5 0.50 0.50 97.5 2.5 — 9.50 20.0 80.0 0.65 10.00 2.5 97.5 — 10.50 2.5 97.5 — 11.00 97.5 2.5 0.65 11.25 97.5 2.5 0.50 12.00 97.5 2.5 END DRUG METABOLISM 73 Differential Mobility Spectrometry for Quantitative and Qualitative Applications in Pharmaceutical Workflows Using SelexION™ Differential Mobility Separation Technology to detect metabolites with better selectivity in complex mixtures on the AB SCIEX TripleTOF® 5600+ LC/MS/MS System James A. Ferguson, PhD. AB SCIEX, 500 Old Connecticut Path, Framingham, MA 01701 Key scientific challenges of metabolite assays • • • • • Materials and methods There are many sources of interfering compounds that can complicate the isolation of metabolites during a pharmacokinetics study. Some interfering compounds come from mobile phases and columns, some from the sample matrix and, depending on the chromatography, some isobars can behave like interferences. Early ADME studies require high throughput and faster chromatography, which can, in turn, could lead to coeluting peaks, especially for very similar compounds such as the positional isomers that can occur from oxidative metabolites or multiple demethylations (e.g. verapamil). The goal of this study was to show separation of these structural isomers that cannot be separated on any existing mass spectrometer. DMS separations were demonstrated for three oxidation metabolites of carbamazepine and two demethylation metabolites of verapamil (Figure 1). Sample preparation The data showing the separation of structural isomers (not shown), indicate that the majority of the compounds moved further into “negative COV space” as the separation voltages (SV) increased (see Figure 2 for a schematic and operational overview of the DMS cell). Nefazodone moved to positive COV space during DMS transmission optimization runs. Because of this additional level of separation provided by DMS, the carbamazepine samples incubated with rat liver microsomes were assessed for metabolites with improved signal-to-noise ratios. The incubations were analyzed while ramping COV; when optimal DMS conditions were found, the samples were re-analyzed using the best SV/COV combination. The DMS-based chromatogram was nearly identical to the extracted ion chromatograms of known metabolites and showed higher signal for some metabolites. This preliminary work shows some promise for finding metabolites in complex matrices. Sensitivity – in vivo studies, due to the biological matrices involved, can negatively impact sensitivity and selectivity. Limited quantitation range – Lowered sensitivity combined with often poor selectivity can compromise the desired lower limits of quantitation (LLOQ). Limitations in selectivity – In both in vivo and in vitro studies, matrix ions can be the most abundant ions in the spectra. Pulling out relevant metabolites rom the background noise can be a challenge on some systems. Key benefits of differential mobility separation (DMS) for metabolite assays • Introduction Enhanced LLOQs – For cases where background noise limits the LOQ, DMS provides an additional level of selectivity, orthogonal to the mass spectrometer and LC system. Better sensitivity – Selectivity gains from DMS permit less involved sample preparations and/or faster gradients, allowing for overall improvements in sensitivity and throughput due to more efficient extractions and better recovery. Selectivity improvements overcome sensitivity losses – DMS is often accompanied by a loss in absolute sensitivity, but the gains in selectivity improve the potential for real gains in LLOQ. Key features of SelexION™ Differential Mobility Separation Technology for metabolite assays • • • • 72 Separation of diverse species reduces baseline noise – SelexION technology separates isobaric species (true iso-bars in this tech note), and charge state interferences to reduce background levels and achieve better selectivity and LOQs – while retaining compatibility with UHPLC speeds. Simple installation and maintenance – DMS is truly orthogonal to LC and MSMS; installs in minutes with no tools required and no need to break vacuum. Device maintenance is minimal and very straightforward. Shortened assay times – SelexION technology can potentially reduce chromatographic run times. Efficient separation process – Planar geometry results in short residence times, high speeds, and minimal diffusion losses for maximum sensitivity and UHPLC compatibility. RUO-MKT-01-1583-A Synthetic oxidative metabolites of carbamazepine (CBZ) and two verapamil demethylations (Figure 1) from Toronto Research Chemicals were used to illustrate DMS separation of isobaric metabolites (structural isomers). To test whether DMS could minimize background, nefazodone was incubated at 10 µM with rat liver microsomes. The samples were then quenched with ACN, lyophilized, and reconstituted in 5% ACN/H2O for analysis. Chromatography Pumps and autosampler: Prominence series from Shimadzu Column: Waters Acquity BEH C18 (2.1 × 100 mm), 1.7 µm Column temp.: 40 °C Injection: 5 μL Flow rate: Programmed (see gradients below) Mobile phase: A) 0.1% formic acid B) Acetonitrile with 0.1% formic acid HIGH RESOLUTION ACCURATE MASS DEFINITIVE METABOLITE ID For Research Use Only. Not for use in diagnostic procedures. Gradient for the quantitative study: Time (min) %A %B Flow (mL/min) 0.00 97.5 2.5 0.50 0.25 97.5 2.5 — 1.00 20.0 80.0 0.65 1.10 2.5 97.5 — 1.35 2.5 97.5 — 1.40 97.5 2.5 0.65 1.60 97.5 2.5 0.50 2.00 97.5 2.5 END Gradient for the qualitative (metabolite ID) study: Time (min) %A %B • • Furthering selectivity with chemical modifiers – Introducing chemical modifiers to the SelexION device cell allows for amplification of the separation capacity and adds a new dimension of selectivity. Ruggedness – SelexION technology provides ruggedness and stability to enable high performance quantitative bioanalysis. DRUG METABOLISM www.absciex.com Figure 1: Structural Isomers used in this study are shown here. A = 2-hydroxycarbamazepine, B = 3-hydroxycarbamazepine, C = 10,11 carbamazepine epoxide; all metabolites have the molecular formula C15H12N2O2. D = norverapamil, and E = p-O-desmethylverapamil; both metabolites have the formula C26H36N2O4. www.absciex.com Flow (mL/min) 0.00 97.5 2.5 0.50 0.50 97.5 2.5 — 9.50 20.0 80.0 0.65 10.00 2.5 97.5 — 10.50 2.5 97.5 — 11.00 97.5 2.5 0.65 11.25 97.5 2.5 0.50 12.00 97.5 2.5 END DRUG METABOLISM 73 Using DMS to minimize background for qualitative studies Figure 2: SelexION™ Technology schematic. SelexION technology uses a planar geometry to which the term differential mobility spectrometry or DMS can be applied. The separation voltage (SV) radially displaces ions towards one or the other electrode depending upon their high and low field mobility characteristics. The compensation voltage (COV) restores the trajectory of ions from a given compound, allowing them to transit through the DMS device and enter the mass spectrometer. SelexION device settings Results and discussion The SelexION technology device installs onto the AB SCIEX TripleTOF® 5600+ LC/MS/MS System in a few minutes without venting the system and without tools. For the best performance, the system can be equilibrated for 20–30 min at the desired temperature. This work was done on a single, experimental instrument model. Quantitative analysis Within the device, ions are separated by differential mobility due to an individual molecule’s characteristic size and shape. An optimized combination of separation voltage (SV) and compensation voltage (COV) is used to separate the selected analyte from interferences. These parameters can be optimized very simply as part of instrument tuning. Optimization of the SV and COV voltages is performed by constant tee-infusion of the analyte into an LC flow that corresponds roughly to the eluent composition for each particular analyte while ramping COV. (a parameter that can be influenced by mobile phase and source conditions) and then ramping again at another SV. (Figure 3 shows an example of this.) The optimal combination of separation and compensation voltages gives the best separation while maintaining maximum peak intensity. Somtimes SV and COVcannot be optimized to achieve separation as was the case for the metabolites of carbamazepine. When this happens, the SelexION device has one more tool to use – the addition of a chemical modifier to the gas stream. For these compounds, isopropanol resulted in SV/COV combinations that provided optimal separation. Data acquisition and processing Sample data were acquired with the Analyst® TF Software 1.6 and processed using PeakView® Software 2.0. 74 Figure 3: Optimizing the SelexION™ Technology Device. The three traces show the ramping of the COV with a fixed SV of 3000 V. It was possible to minimize but not completely eliminate contributions from the other isomers at the voltages indicated by the bars with colors approximating the colors of the traces for each analyte. The addition of modifier gave better separation. RUO-MKT-01-1583-A While working on quantitative data using positional isomers (data not shown), it was found that the majority of the compounds moved further into “negative COV space” with higher SV. However, nefazodone (NEF) moved to positive COV space. To test this proof-of-concept, samples incubated with rat liver microsomes were analyzed in two modes: 1) with the DMS optimized to transmit NEF and 2) in the transparent mode to simulate normal MS data acquisition. Figure 8 shows the chromatograms from the transparent and optimized modes. Figure 9 shows the XICs of the top metabolites found when operating in transparent mode and the TIC from the optimized mode showing nearly identical chromatograms. Table 1 lists the top metabolites of nefazodone found by each technique. Figure 5: Transmission of verapamil (VERA) metabolites. XICs for norverapamil and p-O-desmethylverapamil are shown are shown using a method optimized for p-O-desmethylverapamil. As in Figure 4, the upper trace has the extracted ion chromatograms (XICs) overlaid, and the lower is the 3-D view. The transmission of norverapamil is essentially the same as that of the blank under optimized conditions for the other desmethyl isomer. In this study, the separating power of the SelexION technology device was tested using three synthetic structural isomers of oxidized carbamazepine (CBZ) and two isomers of demethylated verapamil (VERA). For these molecules, only a system with resolving power beyond that of other current chromatography systems could be used in order to resolve the extremely small mass differences arising from the differences in their free energies of formation). The SV and COV space was mapped for the three CBZ oxides and for the two desmethyl VERA compounds. Then SV/COV combinations were chosen to minimize the transmission of other isomers. While good chromatography (which potentially limits throughput) can separate the isomers, the system was run using a very fast gradient to maintain efficiency, making the isomers co-elute. For some compounds, it might be possible to take advantage of the fragmentation patterns of the isomers by monitoring different high-resolultion MRM (MRMHR) transitions to distinguish between the isomers, but the CBZ isomers had nearly identical fragmentation pattern differences, so separating CBZ isomers using the collision cell was not possible. Figure 4: Transmission of the three carbamazepin (CBZ) metabolites. The traces are for the three isomers with molecular formulae of C12H12N2O using a method optimized for 2-OH CBZ. The upper trace shows all the extracted ion chromatograms (XICs) overlaid and the bottom trace shows them in a pseudo-3D view. There is a small (~ 5%) transmission of 3-OH CBZ (red trace) when the system is optimized for 2-OH CBZ. Individual compounds and mixtures of the isomers of hydroxycarbamazepine (OH-CBZ) and desmethylverapamil were run under the optimized DMS conditions for each individual analyte using a short gradient to simulate co-elution. Figure 4 shows the transmission of the each OH-CBZs when run under conditions optimal for the 2-OH-CBZ. Figure 5 shows both verapamil metabolites under conditions optimal for the transmission of p-O-desmethylverapamil. HIGH RESOLUTION ACCURATE MASS DEFINITIVE METABOLITE ID For Research Use Only. Not for use in diagnostic procedures. Figure 6: Calibration curve for 2-OH CBZ from 1–100 ng/mL. Samples with a mixture of all three CBZ oxidations were also run at 1 and 100 ng/mL for each oxide and shown in the table as quality control (QC) samples. If the other oxides were essentially removed from the chromatogram by the differential mobility spectrometry (DMS), the % accuracies should be about the same for the standards and the QCs (which is, indeed, the case). Calibration curves for 2-OH CBZ and CBZ epoxide are shown in Figure 6 and Figure 7. DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 75 Using DMS to minimize background for qualitative studies Figure 2: SelexION™ Technology schematic. SelexION technology uses a planar geometry to which the term differential mobility spectrometry or DMS can be applied. The separation voltage (SV) radially displaces ions towards one or the other electrode depending upon their high and low field mobility characteristics. The compensation voltage (COV) restores the trajectory of ions from a given compound, allowing them to transit through the DMS device and enter the mass spectrometer. SelexION device settings Results and discussion The SelexION technology device installs onto the AB SCIEX TripleTOF® 5600+ LC/MS/MS System in a few minutes without venting the system and without tools. For the best performance, the system can be equilibrated for 20–30 min at the desired temperature. This work was done on a single, experimental instrument model. Quantitative analysis Within the device, ions are separated by differential mobility due to an individual molecule’s characteristic size and shape. An optimized combination of separation voltage (SV) and compensation voltage (COV) is used to separate the selected analyte from interferences. These parameters can be optimized very simply as part of instrument tuning. Optimization of the SV and COV voltages is performed by constant tee-infusion of the analyte into an LC flow that corresponds roughly to the eluent composition for each particular analyte while ramping COV. (a parameter that can be influenced by mobile phase and source conditions) and then ramping again at another SV. (Figure 3 shows an example of this.) The optimal combination of separation and compensation voltages gives the best separation while maintaining maximum peak intensity. Somtimes SV and COVcannot be optimized to achieve separation as was the case for the metabolites of carbamazepine. When this happens, the SelexION device has one more tool to use – the addition of a chemical modifier to the gas stream. For these compounds, isopropanol resulted in SV/COV combinations that provided optimal separation. Data acquisition and processing Sample data were acquired with the Analyst® TF Software 1.6 and processed using PeakView® Software 2.0. 74 Figure 3: Optimizing the SelexION™ Technology Device. The three traces show the ramping of the COV with a fixed SV of 3000 V. It was possible to minimize but not completely eliminate contributions from the other isomers at the voltages indicated by the bars with colors approximating the colors of the traces for each analyte. The addition of modifier gave better separation. RUO-MKT-01-1583-A While working on quantitative data using positional isomers (data not shown), it was found that the majority of the compounds moved further into “negative COV space” with higher SV. However, nefazodone (NEF) moved to positive COV space. To test this proof-of-concept, samples incubated with rat liver microsomes were analyzed in two modes: 1) with the DMS optimized to transmit NEF and 2) in the transparent mode to simulate normal MS data acquisition. Figure 8 shows the chromatograms from the transparent and optimized modes. Figure 9 shows the XICs of the top metabolites found when operating in transparent mode and the TIC from the optimized mode showing nearly identical chromatograms. Table 1 lists the top metabolites of nefazodone found by each technique. Figure 5: Transmission of verapamil (VERA) metabolites. XICs for norverapamil and p-O-desmethylverapamil are shown are shown using a method optimized for p-O-desmethylverapamil. As in Figure 4, the upper trace has the extracted ion chromatograms (XICs) overlaid, and the lower is the 3-D view. The transmission of norverapamil is essentially the same as that of the blank under optimized conditions for the other desmethyl isomer. In this study, the separating power of the SelexION technology device was tested using three synthetic structural isomers of oxidized carbamazepine (CBZ) and two isomers of demethylated verapamil (VERA). For these molecules, only a system with resolving power beyond that of other current chromatography systems could be used in order to resolve the extremely small mass differences arising from the differences in their free energies of formation). The SV and COV space was mapped for the three CBZ oxides and for the two desmethyl VERA compounds. Then SV/COV combinations were chosen to minimize the transmission of other isomers. While good chromatography (which potentially limits throughput) can separate the isomers, the system was run using a very fast gradient to maintain efficiency, making the isomers co-elute. For some compounds, it might be possible to take advantage of the fragmentation patterns of the isomers by monitoring different high-resolultion MRM (MRMHR) transitions to distinguish between the isomers, but the CBZ isomers had nearly identical fragmentation pattern differences, so separating CBZ isomers using the collision cell was not possible. Figure 4: Transmission of the three carbamazepin (CBZ) metabolites. The traces are for the three isomers with molecular formulae of C12H12N2O using a method optimized for 2-OH CBZ. The upper trace shows all the extracted ion chromatograms (XICs) overlaid and the bottom trace shows them in a pseudo-3D view. There is a small (~ 5%) transmission of 3-OH CBZ (red trace) when the system is optimized for 2-OH CBZ. Individual compounds and mixtures of the isomers of hydroxycarbamazepine (OH-CBZ) and desmethylverapamil were run under the optimized DMS conditions for each individual analyte using a short gradient to simulate co-elution. Figure 4 shows the transmission of the each OH-CBZs when run under conditions optimal for the 2-OH-CBZ. Figure 5 shows both verapamil metabolites under conditions optimal for the transmission of p-O-desmethylverapamil. HIGH RESOLUTION ACCURATE MASS DEFINITIVE METABOLITE ID For Research Use Only. Not for use in diagnostic procedures. Figure 6: Calibration curve for 2-OH CBZ from 1–100 ng/mL. Samples with a mixture of all three CBZ oxidations were also run at 1 and 100 ng/mL for each oxide and shown in the table as quality control (QC) samples. If the other oxides were essentially removed from the chromatogram by the differential mobility spectrometry (DMS), the % accuracies should be about the same for the standards and the QCs (which is, indeed, the case). Calibration curves for 2-OH CBZ and CBZ epoxide are shown in Figure 6 and Figure 7. DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 75 Conclusions This preliminary work was completed on a prototype DMS instrument, and there are certainly more analytes and more replicates for each analyte that must be investigated. The results seem to indicate that the SelexION technology device can, at least for some compounds, be used to differentiate between structural isomers that even a high-resolution mass spectrometer cannot resolve. Table1: Top metabolites of nefazodone found. Metabolites above are approximately 1% of total area in descending order by relative area. The table on the left lists metabolites found when the samples were analyzed on a system without differential mobility spectrometry (DMS). The table on the right shows metabolites found when the same sample was anlyzed with DMS. The data for % area for each metabolite in the two tables are quite similar. Figure 7: Calibration curve for carbamazepine (CBZ) epoxide from 1–100 ng/mL. Recovery of the QC samples (see figure 6 caption) again shows that DMS gives enough separation from the other two oxides. Similarly, the data from the nefazodone in vitro incubations indicate that the use of DMS should be further investigated for lowering the background noise when running more complicated in vivo samples. For instance, more experiments are needed to see if any metabolites were overlooked when the SelexION technology device on the system. It is certain that DMS can provide useful orthogonal selectivity during the quantification of structural isomers. The true gain in sensitivity as a function of absolute sensitivity and selectivity will be analyte-dependant and will also be influenced by chromatographic conditions and sample preparation techniques. References 1 Schneider BB, Covey TR, Coy SL, Krylov VE, Nazarov EG, Anal.Chem. 2010; 82: 1867-1880. Schneider BB, Covey TR, Coy SL, Krylov VE, Nazarov EG, ThOC am 08:50, Proceedings of 58nd ASMS Conference on Mass Spectrometry and Allied Topics, Salt Lake City, May 23-27 2010. 2 HIGH RESOLUTION ACCURATE MASS DEFINITIVE METABOLITE ID For Research Use Only. Not for use in diagnostic procedures. Figure 9: Overlaid extracted ion chromatograms (XIC) and total ion chromatograms (TIC). The two traces above are the XICs of the major metabolites (intensities above ~1% of total area) with DMS in transparent mode (blue trace), and the TIC (unextracted) DMS optimized for nefazodone (NEF) (pink trace) zoomed in to the area where the metabolites are eluting. The two chromatograms look remarkably similar. It is interesting to note that in addition to how clean the chromatogram is with DMS optimized, it is also higher in intensity, which is possibly due to less interference from co-eluting peaks. Figure 8: Overlaid TICs. The two traces above are the total ion chromatogram (TIC) from operating with differential mobility spectrometry (DMS) in transparent mode (blue trace), and DMS optimized for nefazodone (pink trace). It is clear that the background is much lower in the pink trace, with DMS optimized for this analyte. 76 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 77 Conclusions This preliminary work was completed on a prototype DMS instrument, and there are certainly more analytes and more replicates for each analyte that must be investigated. The results seem to indicate that the SelexION technology device can, at least for some compounds, be used to differentiate between structural isomers that even a high-resolution mass spectrometer cannot resolve. Table1: Top metabolites of nefazodone found. Metabolites above are approximately 1% of total area in descending order by relative area. The table on the left lists metabolites found when the samples were analyzed on a system without differential mobility spectrometry (DMS). The table on the right shows metabolites found when the same sample was anlyzed with DMS. The data for % area for each metabolite in the two tables are quite similar. Figure 7: Calibration curve for carbamazepine (CBZ) epoxide from 1–100 ng/mL. Recovery of the QC samples (see figure 6 caption) again shows that DMS gives enough separation from the other two oxides. Similarly, the data from the nefazodone in vitro incubations indicate that the use of DMS should be further investigated for lowering the background noise when running more complicated in vivo samples. For instance, more experiments are needed to see if any metabolites were overlooked when the SelexION technology device on the system. It is certain that DMS can provide useful orthogonal selectivity during the quantification of structural isomers. The true gain in sensitivity as a function of absolute sensitivity and selectivity will be analyte-dependant and will also be influenced by chromatographic conditions and sample preparation techniques. References 1 Schneider BB, Covey TR, Coy SL, Krylov VE, Nazarov EG, Anal.Chem. 2010; 82: 1867-1880. Schneider BB, Covey TR, Coy SL, Krylov VE, Nazarov EG, ThOC am 08:50, Proceedings of 58nd ASMS Conference on Mass Spectrometry and Allied Topics, Salt Lake City, May 23-27 2010. 2 HIGH RESOLUTION ACCURATE MASS DEFINITIVE METABOLITE ID For Research Use Only. Not for use in diagnostic procedures. Figure 9: Overlaid extracted ion chromatograms (XIC) and total ion chromatograms (TIC). The two traces above are the XICs of the major metabolites (intensities above ~1% of total area) with DMS in transparent mode (blue trace), and the TIC (unextracted) DMS optimized for nefazodone (NEF) (pink trace) zoomed in to the area where the metabolites are eluting. The two chromatograms look remarkably similar. It is interesting to note that in addition to how clean the chromatogram is with DMS optimized, it is also higher in intensity, which is possibly due to less interference from co-eluting peaks. Figure 8: Overlaid TICs. The two traces above are the total ion chromatogram (TIC) from operating with differential mobility spectrometry (DMS) in transparent mode (blue trace), and DMS optimized for nefazodone (pink trace). It is clear that the background is much lower in the pink trace, with DMS optimized for this analyte. 76 RUO-MKT-01-1583-A DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 77 Solving Bottlenecks in Metabolite Identification Using TripleTOF® Systems and MetabolitePilot™ Software Introduction High performance solution with innovative capabilities and integrated software to improve efficiency Key scientific challenges of metabolite ID workflows • • • Productivity – Increasing productivity in drug discovery and development continues to be a primary goal in pharmaceutical research. Analysis at relevant doses – For many compounds, ADME studies have been completed at a high, non-therapeutic concentration to monitor the metabolism process. This can sometimes lead to false conclusions because some metabolic pathways are concentration-dependent for some compounds. High performance software – In the past, the work of finding, confirming, and quantifying metabolites has required the use of multiple software packages. MetabolitePilot™ Software can perform all three functions in one software package. Key benefits of the TripleTOF® 5600 System and MetabolitePilot Software for metabolite ID workflows • • • • • • 78 Innovative instrument design – combining the proven high performance triple quadrupole front end with the Accelerator TOF™ Analyzer, a state of the art accurate mass analyzer with unprecedented sensitivity, speed, resolution, and mass accuracy. Smarter acquisition – The introduction of real-time mass defect triggering allows the TripleTOF 5600 system to automatically acquire MS/MS on low-level metabolites. Now, the TripleTOF 5600 system can trigger on ions with mass defects related to the parent drug at intensities lower than other compounds, allowing the proverbial needle to be pulled from its haystack. Rapid scanning speeds – The TripleTOF 5600 system can obtain up to 100 high-resolution MS/MS spectra per second, which accommodates fast chromatography while still obtaining comprehensive MS and MS/ MS data and without sacrificing resolution. High sensitivity – The TripleTOF 5600 system is sensitive enough to detect metabolites at low substrate concentrations in vitro and from in vivo PK samples. A comprehensive metabolite ID package – MetabolitePilot Software is an integrated, feature-rich, and easy-to-use software package that combines multiple peak-finding strategies, structural assignment algorithms, MS/MS fragment interpretation modules, and multi-sample correlation in a single software package. Combined quant/qual workflows – The excellent detector dynamic range for quant in full-scan mode and the high mass accuracy in MS and MS/MS modes enable comprehensive metabolite identification and quantification methods. RUO-MKT-01-1583-A High-resolution accurate mass LC/MS/MS has become the technique of choice for metabolite identification. During the discovery stage, high MS and MS/MS scan speeds and a powerful full-scan capacity make high-resolution mass spectrometry well-suited to the detection the of major metabolites for lead optimization. During the development stage, powerful features such as mass-defect-triggered information-dependent acquisition (IDA) and high MS/MS mass accuracy in-depth structure elucidation and the characterization of in vivo metabolites, even in complex matrices such as bile. As high resolution instruments have become increasingly powerful, manual processing and interpretation of large amounts of data pose a major challenge. Integrated software to detect metabolites, perform structure assignment and MS/MS interpretation, and compare metabolism across multiple samples is essential to realize the benefits of high-resolution data. Figure 1: AB SCIEX TripleTOF® 5600 System ion path. Proven front-end technology coupled to a state-of-the-art Accelerator TOF™ Analyzer has resulted in unparalleled performance for both quant and qual methods. The AB SCIEX TripleTOF 4600 and 5600+ systems combined with MetabolitePilot Software offer an intuitive, fully-integrated hardware and software solution that sets the standard for speed, sensitivity, and ease-of-use. This technical note provide an overview of the key benefits of the TripleTOF system’s technology for metabolite ID discovery and development. The TripleTOF systems have revolutionized this workflow by implementing mass defects as a real-time IDA criterion for triggering MS/MS in the same run. Multiple mass defect ranges can be specified in the IDA criteria (Figure 3). Due to its high acquisition speed, the system is able to perform the mass defect filtering on each TOF scan immediately after it is acquired. Advanced technology enables true quant/qual without compromises Key features of the TripleTOF 5600 system and MetabolitePilot Software for metabolite ID workflows • • • • Resolution and mass accuracy regardless of scan speed – Instruments employing orbital trapping require slower scanning to obtain higher resolution. The TripleTOF systems maintain approximately 30K resolution regardless of analysis speed. This high-performance, especially in MS/ MS mode, provides unambiguous elemental composition assignment and identification of the site of metabolism. MetabolitePilot Software allows batch processing for higher throughput and more efficient use of the chemist’s time. Calibration stability and linearity – Traditional time-of-flight (TOF) instruments typically require internal calibration to maintain mass accuracy, resulting in lowered signals for analytes and a non-linear quantitative response. The TripleTOF systems have been designed to overcome both of these limitations. Resolution and mass accuracy even at low m/z – Traditional TOF instruments provided good mass accuracy and resolution at high m/z. The high detector speed of the TripleTOF systems allow for sufficient resolution at low m/z, providing unambiguous formulae assigments using accurate mass, as well as the isotope and fragmention pattern. DRUG METABOLISM www.absciex.com DEFINITIVE METABOLITE ID DEFINITIVE METABOLITE ID For Research Use Only. Not for use in diagnostic procedures. The AB SCIEX TripleTOF systems combine the best attributes of triple quadrupoles and accurate mass analyzers in a single instrument (Figure 1). This results in the linearity and sensitivity of a high performance triple quadrupole for quantitation integrated with the speed, high mass accuracy (1-2 ppm or less), and high resolution (30K) of an accurate mass instrument for qualitative analysis. As a result, it is possible to acquire information-rich data with highresolution TOF MS and MS/MS data in IDA mode on a UHPLC time scale (Figure 2). This is achieved without sacrificing resolution for speed, or sensitivity, and mass accuracy for linearity. The end result is that both high-resolution MS and MS/MS data is acquired in a single injection, even for complex in vivo samples and for low-level metabolites (Table 1). This single-step method not only improves throughput but also makes data analysis and review faster and simpler, with all the data available to the analyst for data interpretation. Real-time multiple mass defect-triggered IDA (mMDF-IDA) Mass defect filtering is a powerful accurate mass technique to detect phase I and II metabolites that are related by elemental composition to the parent compound. This filtering mode is particularly beneficial in complex, in vivo samples. The traditional approach involves applying a mass defect filter the data after acquisition to identify potential metabolite peaks. A second injection is then performed to obtain the MS/MS spectra of these peaks. The data is combined to allow a proper evaluation of each peak. This approach requires two injections and two data processing stages, which can be time-consuming. www.absciex.com Figure 2: Broad coverage for phase I metabolites. An overlay of accurate mass extracted ion chromatograms (XIC’s) for imipramine incubation at 5 min is shown. A wide range of imipramine phase I metabolites were easily detected using a completely generic time-of-flight mass spectrometry TOF-MS informationdependent acquisition (IDA) method in under 2.5 min. DRUG METABOLISM 79 Solving Bottlenecks in Metabolite Identification Using TripleTOF® Systems and MetabolitePilot™ Software Introduction High performance solution with innovative capabilities and integrated software to improve efficiency Key scientific challenges of metabolite ID workflows • • • Productivity – Increasing productivity in drug discovery and development continues to be a primary goal in pharmaceutical research. Analysis at relevant doses – For many compounds, ADME studies have been completed at a high, non-therapeutic concentration to monitor the metabolism process. This can sometimes lead to false conclusions because some metabolic pathways are concentration-dependent for some compounds. High performance software – In the past, the work of finding, confirming, and quantifying metabolites has required the use of multiple software packages. MetabolitePilot™ Software can perform all three functions in one software package. Key benefits of the TripleTOF® 5600 System and MetabolitePilot Software for metabolite ID workflows • • • • • • 78 Innovative instrument design – combining the proven high performance triple quadrupole front end with the Accelerator TOF™ Analyzer, a state of the art accurate mass analyzer with unprecedented sensitivity, speed, resolution, and mass accuracy. Smarter acquisition – The introduction of real-time mass defect triggering allows the TripleTOF 5600 system to automatically acquire MS/MS on low-level metabolites. Now, the TripleTOF 5600 system can trigger on ions with mass defects related to the parent drug at intensities lower than other compounds, allowing the proverbial needle to be pulled from its haystack. Rapid scanning speeds – The TripleTOF 5600 system can obtain up to 100 high-resolution MS/MS spectra per second, which accommodates fast chromatography while still obtaining comprehensive MS and MS/ MS data and without sacrificing resolution. High sensitivity – The TripleTOF 5600 system is sensitive enough to detect metabolites at low substrate concentrations in vitro and from in vivo PK samples. A comprehensive metabolite ID package – MetabolitePilot Software is an integrated, feature-rich, and easy-to-use software package that combines multiple peak-finding strategies, structural assignment algorithms, MS/MS fragment interpretation modules, and multi-sample correlation in a single software package. Combined quant/qual workflows – The excellent detector dynamic range for quant in full-scan mode and the high mass accuracy in MS and MS/MS modes enable comprehensive metabolite identification and quantification methods. RUO-MKT-01-1583-A High-resolution accurate mass LC/MS/MS has become the technique of choice for metabolite identification. During the discovery stage, high MS and MS/MS scan speeds and a powerful full-scan capacity make high-resolution mass spectrometry well-suited to the detection the of major metabolites for lead optimization. During the development stage, powerful features such as mass-defect-triggered information-dependent acquisition (IDA) and high MS/MS mass accuracy in-depth structure elucidation and the characterization of in vivo metabolites, even in complex matrices such as bile. As high resolution instruments have become increasingly powerful, manual processing and interpretation of large amounts of data pose a major challenge. Integrated software to detect metabolites, perform structure assignment and MS/MS interpretation, and compare metabolism across multiple samples is essential to realize the benefits of high-resolution data. Figure 1: AB SCIEX TripleTOF® 5600 System ion path. Proven front-end technology coupled to a state-of-the-art Accelerator TOF™ Analyzer has resulted in unparalleled performance for both quant and qual methods. The AB SCIEX TripleTOF 4600 and 5600+ systems combined with MetabolitePilot Software offer an intuitive, fully-integrated hardware and software solution that sets the standard for speed, sensitivity, and ease-of-use. This technical note provide an overview of the key benefits of the TripleTOF system’s technology for metabolite ID discovery and development. The TripleTOF systems have revolutionized this workflow by implementing mass defects as a real-time IDA criterion for triggering MS/MS in the same run. Multiple mass defect ranges can be specified in the IDA criteria (Figure 3). Due to its high acquisition speed, the system is able to perform the mass defect filtering on each TOF scan immediately after it is acquired. Advanced technology enables true quant/qual without compromises Key features of the TripleTOF 5600 system and MetabolitePilot Software for metabolite ID workflows • • • • Resolution and mass accuracy regardless of scan speed – Instruments employing orbital trapping require slower scanning to obtain higher resolution. The TripleTOF systems maintain approximately 30K resolution regardless of analysis speed. This high-performance, especially in MS/ MS mode, provides unambiguous elemental composition assignment and identification of the site of metabolism. MetabolitePilot Software allows batch processing for higher throughput and more efficient use of the chemist’s time. Calibration stability and linearity – Traditional time-of-flight (TOF) instruments typically require internal calibration to maintain mass accuracy, resulting in lowered signals for analytes and a non-linear quantitative response. The TripleTOF systems have been designed to overcome both of these limitations. Resolution and mass accuracy even at low m/z – Traditional TOF instruments provided good mass accuracy and resolution at high m/z. The high detector speed of the TripleTOF systems allow for sufficient resolution at low m/z, providing unambiguous formulae assigments using accurate mass, as well as the isotope and fragmention pattern. DRUG METABOLISM www.absciex.com DEFINITIVE METABOLITE ID DEFINITIVE METABOLITE ID For Research Use Only. Not for use in diagnostic procedures. The AB SCIEX TripleTOF systems combine the best attributes of triple quadrupoles and accurate mass analyzers in a single instrument (Figure 1). This results in the linearity and sensitivity of a high performance triple quadrupole for quantitation integrated with the speed, high mass accuracy (1-2 ppm or less), and high resolution (30K) of an accurate mass instrument for qualitative analysis. As a result, it is possible to acquire information-rich data with highresolution TOF MS and MS/MS data in IDA mode on a UHPLC time scale (Figure 2). This is achieved without sacrificing resolution for speed, or sensitivity, and mass accuracy for linearity. The end result is that both high-resolution MS and MS/MS data is acquired in a single injection, even for complex in vivo samples and for low-level metabolites (Table 1). This single-step method not only improves throughput but also makes data analysis and review faster and simpler, with all the data available to the analyst for data interpretation. Real-time multiple mass defect-triggered IDA (mMDF-IDA) Mass defect filtering is a powerful accurate mass technique to detect phase I and II metabolites that are related by elemental composition to the parent compound. This filtering mode is particularly beneficial in complex, in vivo samples. The traditional approach involves applying a mass defect filter the data after acquisition to identify potential metabolite peaks. A second injection is then performed to obtain the MS/MS spectra of these peaks. The data is combined to allow a proper evaluation of each peak. This approach requires two injections and two data processing stages, which can be time-consuming. www.absciex.com Figure 2: Broad coverage for phase I metabolites. An overlay of accurate mass extracted ion chromatograms (XIC’s) for imipramine incubation at 5 min is shown. A wide range of imipramine phase I metabolites were easily detected using a completely generic time-of-flight mass spectrometry TOF-MS informationdependent acquisition (IDA) method in under 2.5 min. DRUG METABOLISM 79 Information-Dependent Acquisition (IDA) Method Metabolites detected by processing full-scan data Peak Intensity alone Dynamic Background Subtraction (DBS) mMDF with DBS 31 31 31 Automatically acquired MS/MS Spectra 17 27 31 Triggering Success Rate 53% 87% 100% DEFINITIVE METABOLITE ID DEFINITIVE METABOLITE ID For Research Use Only. Not for use in diagnostic procedures. Table 1: Diclofenac metabolites detected in bile using multiple mass defect filtering (mMDF)-information-dependent acquisition IDA. MS/MS spectra of all the metabolites present were acquired successfully in a single injection analysis.1 Figure 3: Real-time multiple mass defect triggered information-dependent acquisition (IDA). Mass defect ranges can be selected and programmed in the Analyst® Software method editor as IDA criteria for MS/MS selection in real time. Integrated MS/MS fragment interpretation MS/MS fragment interpretation and assignment to a proposed metabolite structure is arguably the single most labor-intensive and time-consuming task when identifying metabolites. Typically separate software packages were needed for data interpretation which required switching between two programs. Other software packages often do not provide integrated storage and reporting of results, proposed structures, or MS/MS fragment assignments that support proposed structures. MetabolitePilot Software has a powerful MS/MS fragment interpretation function integrated directly in the results workspace. When reviewing potential metabolites, the scientist can edit structures and perform automatic fragment assignment (Figure 4). Both the structure and fragment assignments are saved in the results table and are directly accessible in data review. Integrated reports can be generated automatically. Fragment assignments are automatically highlighted on the structure when the fragment is selected. The software also automatically shows possible neutral losses from the selected fragment to show its relationship to other product ions in the MS/MS spectrum. A score is assigned to each fragment assignment to aid in data review. The spectrum can be automatically annotated with fragment structures and elemental compositions (Figure 4). Due to the high resolution and mass accuracy of the TripleTOF systems in MS/MS mode, unambiguous elemental compositions of product ions can be calculated by the software. This integrated functionality allows the scientist to quickly evaluate different structure proposals in an interactive and userfriendly manner. The structure and the entire fragment assignment file are automatically saved in the results workspace (Figure 5). Figure 6: Correlation workspace. Integrated, multi-sample correlation enables comparison of metabolites across a time course study from within MetabolitePilot™ Software. In addition to automatic plots of selected metabolites, chromatographic and MS/MS data was also overlaid. Multi-sample correlation With increased emphasis on MIST guidelines, correlation of metabolism across multiple samples and estimation of relative amounts has become a critical part of data interpretation. For example, the metabolic profile needs to be compared across multiple species to spot a disproportionate metabolite as early as possible. In vitro – in vivo correlation and the study of metabolite kinetics across a time course also require multi-sample correlation. Traditional software is limited to working with a single sample/ control pair at a time, requiring that the data be exported for further correlation. 80 RUO-MKT-01-1583-A Figure 5: Results workspace. Metabolite structures are an integral part of the results table and are displayed automatically when a metabolite is selected (highlighted in yellow). DRUG METABOLISM www.absciex.com Conclusions • • Figure 6 illustrates how an entire time course can be easily evaluated for metabolites formed during an imipramine microsomal incubation. The data is automatically processed in batch mode to generate results tables for all time points. The results for all samples are then selected for correlation. The software automatically tabulates the abundance of each metabolite across all samples and overlays all chromatographic, MS, and MS/MS data as shown for a dioxidation metabolite. • Plots of area counts are generated by selecting one or more metabolites. The user can easily toggle between linear plots and bar graphs (Figure 7), which is more useful when comparing metabolism across multiple species. Automatic summary tables can also be generated showing the area counts or presence or absence of each metabolite across all samples. Correlation reports are automatically generated, and all tables and charts can be easily copied and pasted from the workspace for preparing presentations. www.absciex.com The TripleTOF systems with MetabolitePilot Software significantly improve bottlenecks that develop during metabolite identification due to the time-consuming and complex nature of structure elucidation. Real time multiple mass defect triggered IDA is a powerful method which enables single injection acquisition of MS/MS data, even in complex samples and for low-abundance metabolites. Integrated MS/MS fragment interpretation improves the efficiency of data analysis by performing fragment assignments on a proposed structure in a single integrated workspace. Automatic correlation of UV, MS, and MS/MS data in a single results workspace provides for faster and easier data review. Multiple sample correlation goes beyond traditional single sample/ control processing to enable comparison of metabolites across multiple samples easily and without needing to have manual data handled manipulation in a separate software program. • MetabolitePilot Software can easily correlate metabolites across multiple samples, automatically generate plots, and overlay chromatograms, MS, and MS/MS data from multiple samples all within a single workspace. Both MS and UV responses can be used as well as other types of analog data such as beta-RAM. A relative response factor can be applied for each metabolite individually to correct the MS response for different metabolites based on the UV signal. Figure 4: Integrated MS/MS Interpretation. It is simple to toggle between the results and interpretation workspaces. Within the results workspace, structure editing and automatic fragment assignment can be performed for a proposed metabolite. Fragments are assigned and highlighted on the chemical structure for the selected ion (highlighted in yellow). Figure 7: Flexible data visualization. The user can quickly switch between line plots and bar graphs of the selected metabolites for time course studies and inter-species comparison. • References 1 Jie Xing, Kerong Zhang, Minghshe Zhu, et al, WOG PM 2:30, The 59th ASMS Conference on Mass Spectrometry and Allied Topics, June 5 – 9, 2011, Denver, CO. Rapid Metabolite Identification using MetabolitePilot™ Software”, AB SCIEX Technical Note, Publication 2490211- 02. 2“ 3 “Simultaneous Pharmacokinetic Profiling and Automated Metabolite Identification using the AB SCIEX TripleTOF® 5600 System and MetabolitePilot™ Software”, AB SCIEX Technical Note, Publication 1270210-01. 4 “Solving Bottlenecks in Metabolite ID Data Analysis With MetabolitePilot™ Software”, AB SCIEX Technical Note, Publication 3610211-01. 5 “Breakthrough Productivity for ADME Studies Using the AB SCIEX TripleTOF® 5600 System”, AB SCIEX Technical Note, Publication 0480110-01. 6 R. J. Mortishire-Smith et al, “Evaluation of Chemically Intelligent Acquisition and Processing Tools as a Platform for Discovery Metabolite Identification,” TP 222,The 59th ASMS Conference on Mass Spectrometry and Allied Topics, June 5 – 9, 2011, Denver, CO. 7 To download a trial version of MetabolitePilot™ Software, please visit: http://www.absciex.com/ Products/Software/MetabolitePilot-Software. DRUG METABOLISM 81 Information-Dependent Acquisition (IDA) Method Metabolites detected by processing full-scan data Peak Intensity alone Dynamic Background Subtraction (DBS) mMDF with DBS 31 31 31 Automatically acquired MS/MS Spectra 17 27 31 Triggering Success Rate 53% 87% 100% DEFINITIVE METABOLITE ID DEFINITIVE METABOLITE ID For Research Use Only. Not for use in diagnostic procedures. Table 1: Diclofenac metabolites detected in bile using multiple mass defect filtering (mMDF)-information-dependent acquisition IDA. MS/MS spectra of all the metabolites present were acquired successfully in a single injection analysis.1 Figure 3: Real-time multiple mass defect triggered information-dependent acquisition (IDA). Mass defect ranges can be selected and programmed in the Analyst® Software method editor as IDA criteria for MS/MS selection in real time. Integrated MS/MS fragment interpretation MS/MS fragment interpretation and assignment to a proposed metabolite structure is arguably the single most labor-intensive and time-consuming task when identifying metabolites. Typically separate software packages were needed for data interpretation which required switching between two programs. Other software packages often do not provide integrated storage and reporting of results, proposed structures, or MS/MS fragment assignments that support proposed structures. MetabolitePilot Software has a powerful MS/MS fragment interpretation function integrated directly in the results workspace. When reviewing potential metabolites, the scientist can edit structures and perform automatic fragment assignment (Figure 4). Both the structure and fragment assignments are saved in the results table and are directly accessible in data review. Integrated reports can be generated automatically. Fragment assignments are automatically highlighted on the structure when the fragment is selected. The software also automatically shows possible neutral losses from the selected fragment to show its relationship to other product ions in the MS/MS spectrum. A score is assigned to each fragment assignment to aid in data review. The spectrum can be automatically annotated with fragment structures and elemental compositions (Figure 4). Due to the high resolution and mass accuracy of the TripleTOF systems in MS/MS mode, unambiguous elemental compositions of product ions can be calculated by the software. This integrated functionality allows the scientist to quickly evaluate different structure proposals in an interactive and userfriendly manner. The structure and the entire fragment assignment file are automatically saved in the results workspace (Figure 5). Figure 6: Correlation workspace. Integrated, multi-sample correlation enables comparison of metabolites across a time course study from within MetabolitePilot™ Software. In addition to automatic plots of selected metabolites, chromatographic and MS/MS data was also overlaid. Multi-sample correlation With increased emphasis on MIST guidelines, correlation of metabolism across multiple samples and estimation of relative amounts has become a critical part of data interpretation. For example, the metabolic profile needs to be compared across multiple species to spot a disproportionate metabolite as early as possible. In vitro – in vivo correlation and the study of metabolite kinetics across a time course also require multi-sample correlation. Traditional software is limited to working with a single sample/ control pair at a time, requiring that the data be exported for further correlation. 80 RUO-MKT-01-1583-A Figure 5: Results workspace. Metabolite structures are an integral part of the results table and are displayed automatically when a metabolite is selected (highlighted in yellow). DRUG METABOLISM www.absciex.com Conclusions • • Figure 6 illustrates how an entire time course can be easily evaluated for metabolites formed during an imipramine microsomal incubation. The data is automatically processed in batch mode to generate results tables for all time points. The results for all samples are then selected for correlation. The software automatically tabulates the abundance of each metabolite across all samples and overlays all chromatographic, MS, and MS/MS data as shown for a dioxidation metabolite. • Plots of area counts are generated by selecting one or more metabolites. The user can easily toggle between linear plots and bar graphs (Figure 7), which is more useful when comparing metabolism across multiple species. Automatic summary tables can also be generated showing the area counts or presence or absence of each metabolite across all samples. Correlation reports are automatically generated, and all tables and charts can be easily copied and pasted from the workspace for preparing presentations. www.absciex.com The TripleTOF systems with MetabolitePilot Software significantly improve bottlenecks that develop during metabolite identification due to the time-consuming and complex nature of structure elucidation. Real time multiple mass defect triggered IDA is a powerful method which enables single injection acquisition of MS/MS data, even in complex samples and for low-abundance metabolites. Integrated MS/MS fragment interpretation improves the efficiency of data analysis by performing fragment assignments on a proposed structure in a single integrated workspace. Automatic correlation of UV, MS, and MS/MS data in a single results workspace provides for faster and easier data review. Multiple sample correlation goes beyond traditional single sample/ control processing to enable comparison of metabolites across multiple samples easily and without needing to have manual data handled manipulation in a separate software program. • MetabolitePilot Software can easily correlate metabolites across multiple samples, automatically generate plots, and overlay chromatograms, MS, and MS/MS data from multiple samples all within a single workspace. Both MS and UV responses can be used as well as other types of analog data such as beta-RAM. A relative response factor can be applied for each metabolite individually to correct the MS response for different metabolites based on the UV signal. Figure 4: Integrated MS/MS Interpretation. It is simple to toggle between the results and interpretation workspaces. Within the results workspace, structure editing and automatic fragment assignment can be performed for a proposed metabolite. Fragments are assigned and highlighted on the chemical structure for the selected ion (highlighted in yellow). Figure 7: Flexible data visualization. The user can quickly switch between line plots and bar graphs of the selected metabolites for time course studies and inter-species comparison. • References 1 Jie Xing, Kerong Zhang, Minghshe Zhu, et al, WOG PM 2:30, The 59th ASMS Conference on Mass Spectrometry and Allied Topics, June 5 – 9, 2011, Denver, CO. Rapid Metabolite Identification using MetabolitePilot™ Software”, AB SCIEX Technical Note, Publication 2490211- 02. 2“ 3 “Simultaneous Pharmacokinetic Profiling and Automated Metabolite Identification using the AB SCIEX TripleTOF® 5600 System and MetabolitePilot™ Software”, AB SCIEX Technical Note, Publication 1270210-01. 4 “Solving Bottlenecks in Metabolite ID Data Analysis With MetabolitePilot™ Software”, AB SCIEX Technical Note, Publication 3610211-01. 5 “Breakthrough Productivity for ADME Studies Using the AB SCIEX TripleTOF® 5600 System”, AB SCIEX Technical Note, Publication 0480110-01. 6 R. J. Mortishire-Smith et al, “Evaluation of Chemically Intelligent Acquisition and Processing Tools as a Platform for Discovery Metabolite Identification,” TP 222,The 59th ASMS Conference on Mass Spectrometry and Allied Topics, June 5 – 9, 2011, Denver, CO. 7 To download a trial version of MetabolitePilot™ Software, please visit: http://www.absciex.com/ Products/Software/MetabolitePilot-Software. DRUG METABOLISM 81 Removing Bottlenecks in Metabolite ID Data Analysis with MetabolitePilot™ Software New multi-sample correlation and integrated MS/MS interpretation improve efficiency of data analysis Hesham Ghobarah, Alina Dindyal-Popescu, Hai Ying AB SCIEX, Toronto, Ontario Key scientific challenges of metabolite ID workflows • • • Productivity – Increasing productivity in drug discovery and development continues to be a primary goal in pharmaceutical research. Analysis at relevant doses – For many compounds, ADME studies have been completed at a high, non-therapeutic concentration to monitor the metabolism process. This can sometimes lead to false conclusions because some metabolic pathways are concentration-dependent for some compounds. • • • • • • 82 Innovative instrument design – combining the proven high performance triple quadrupole front end with the Accelerator TOF™ Analyzer, a state of the art accurate mass analyzer with unprecedented sensitivity, speed, resolution, and mass accuracy. Smarter acquisition – The introduction of real-time mass defect triggering allows the TripleTOF 5600 system to automatically acquire MS/MS on low-level metabolites. Now, the TripleTOF 5600 system can trigger on ions with mass defects related to the parent drug at intensities lower than other compounds, allowing the proverbial needle to be pulled from its haystack. Rapid scanning speeds – The TripleTOF 5600 system can obtain up to 100 high-resolution MS/MS spectra per second, which accommodates fast chromatography while still obtaining comprehensive MS and MS/ MS data and without sacrificing resolution. High sensitivity – The TripleTOF 5600 system is sensitive enough to detect metabolites at low substrate concentrations in vitro and from in vivo PK samples. A comprehensive metabolite ID package – MetabolitePilot Software is an integrated, feature-rich, and easy-to-use software package that combines multiple peak-finding strategies, structural assignment algorithms, MS/MS fragment interpretation modules, and multi-sample correlation in a single software package. Combined quant/qual workflows – The excellent detector dynamic range for quant in full-scan mode and the high mass accuracy in MS and MS/MS modes enable comprehensive metabolite identification and quantification methods. RUO-MKT-01-1583-A correlation. Traditional software is limited to working with a single sample/control pair at a time. MetabolitePilot Software can easily perform correlation of metabolites across multiple samples, automatically generate plots, and overlay chromatograms, MS, and MS/MS data from multiple samples all within a single workspace. Mass Spectrometry – A generic method was used for data acquisition. The information-dependent acquisition (IDA) method consisted of a time-of-flight mass spectrometry (TOF MS) survey scan followed by two TOF MS/MS scans. The mass range was m/z 100–1000 for both MS and MS/MS. The Dynamic Background Subtraction™ Algorithm was applied for IDA criteria, and a collision energy of 35 eV with a spread of ±10 eV was used for the MS/MS scans. External mass calibration was performed automatically using the calibrant delivery system (CDS). Data processing was performed using MetabolitePilot Software 1.5. Figure 1 demonstrates how an entire time course can be easily evaluated for metabolites arising from a microsome incubation of imipramine. The data is automatically processed in batch mode to generate results tables for all time points. The results for all samples are then selected for correlation. The software automatically tabulates the abundance of each metabolite across all samples, and overlays all chromatographic, MS, and MS/MS data, as shown for a dioxidation metabolite. Multi-sample correlation High performance software – In the past, the work of finding, confirming, and quantifying metabolites has required the use of multiple software packages. MetabolitePilot™ Software can perform all three functions in one software package. Key benefits of the TripleTOF® 600 System and MetabolitePilot Software for metabolite ID workflows Chromatography – Sample analysis was performed on the AB SCIEX TripleTOF® 5600 System coupled with a Shimadzu Prominence UFLC-XR HPLC system. A generic acetonitrile/water/0.1% formic acid gradient was used on a Phenomenex Synergi Polar-RP column 2.5 µm, (2 × 50 mm). Total run time was 3.7 min. Injection volume was 10 µL. Key features of the TripleTOF 5600 system and MetabolitePilot Software for metabolite ID workflows • • • • With increased emphasis on MIST guidelines, correlation of metabolism across multiple samples and estimation of relative amounts of metabolites has become a critical part of data interpretation. For example, the metabolic profile needs to be compared across multiple species to spot a disproportionate metabolite as early as possible. In vitro – in vivo correlation and the study of metabolite kinetics also require multisample DEFINITIVE METABOLITE ID DEFINITIVE METABOLITE ID For Research Use Only. Not for use in diagnostic procedures. Both MS and UV responses can be used as well as other types of analog data such as beta-RAM. A relative response factor can be applied for each metabolite individually to correct the MS response for different metabolites based on the corresponding UV signal. Plots of area counts are generated by selecting one or more metabolites. The user can easily toggle between linear plots and bar graphs (Figure 2), which is more useful when comparing metabolism across multiple species. Automatic summary tables can also be generated showing the area counts or presence/ absence of each metabolite across all samples (Figure 3). Resolution and mass accuracy regardless of scan speed – Instruments employing orbital trapping require slower scanning to obtain higher resolution. The TripleTOF systems maintain approximately 30K resolution regardless of analysis speed. This high-performance, especially in MS/MS mode, provides unambiguous elemental composition assignment and identification of the site of metabolism. MetabolitePilot Software allows batch processing for higher throughput and more efficient use of the chemist’s time. Calibration stability and linearity – Traditional time-of-flight (TOF) instruments typically require internal calibration to maintain mass accuracy, resulting in lowered signals for analytes and a non-linear quantitative response. The TripleTOF systems have been designed to overcome both of these limitations. Resolution and mass accuracy even at low m/z – Traditional TOF instruments provided good mass accuracy and resolution at high m/z. The high detector speed of the TripleTOF systems allow for sufficient resolution at low m/z, providing unambiguous formulae assigments using accurate mass, as well as the isotope and fragmention pattern. Experimental Sample Preparation – Haloperidol and Imipramine were incubated in rat liver microsomes at an initial substrate concentration of 1 µM. Standard high-throughput incubation conditions were used with time points of 0, 5, 15, and 30 min. Protein concentration was 1 mg/mL, and NADPH was present at 4 mM. The reaction was quenched using an equal volume of acetonitrile and then diluted 1:1 with water prior to analysis. The final substrate concentration at t = 0 was 0.25 µM. DRUG METABOLISM www.absciex.com Figure 1: Correlation workspace. Integrated, multi-sample correlation enables comparison of metabolites across a time course from within MetabolitePilot™ Software. In addition to automatic plots of selected metabolites, chromatographic and MS/MS data are also overlaid. www.absciex.com DRUG METABOLISM 83 Removing Bottlenecks in Metabolite ID Data Analysis with MetabolitePilot™ Software New multi-sample correlation and integrated MS/MS interpretation improve efficiency of data analysis Hesham Ghobarah, Alina Dindyal-Popescu, Hai Ying AB SCIEX, Toronto, Ontario Key scientific challenges of metabolite ID workflows • • • Productivity – Increasing productivity in drug discovery and development continues to be a primary goal in pharmaceutical research. Analysis at relevant doses – For many compounds, ADME studies have been completed at a high, non-therapeutic concentration to monitor the metabolism process. This can sometimes lead to false conclusions because some metabolic pathways are concentration-dependent for some compounds. • • • • • • 82 Innovative instrument design – combining the proven high performance triple quadrupole front end with the Accelerator TOF™ Analyzer, a state of the art accurate mass analyzer with unprecedented sensitivity, speed, resolution, and mass accuracy. Smarter acquisition – The introduction of real-time mass defect triggering allows the TripleTOF 5600 system to automatically acquire MS/MS on low-level metabolites. Now, the TripleTOF 5600 system can trigger on ions with mass defects related to the parent drug at intensities lower than other compounds, allowing the proverbial needle to be pulled from its haystack. Rapid scanning speeds – The TripleTOF 5600 system can obtain up to 100 high-resolution MS/MS spectra per second, which accommodates fast chromatography while still obtaining comprehensive MS and MS/ MS data and without sacrificing resolution. High sensitivity – The TripleTOF 5600 system is sensitive enough to detect metabolites at low substrate concentrations in vitro and from in vivo PK samples. A comprehensive metabolite ID package – MetabolitePilot Software is an integrated, feature-rich, and easy-to-use software package that combines multiple peak-finding strategies, structural assignment algorithms, MS/MS fragment interpretation modules, and multi-sample correlation in a single software package. Combined quant/qual workflows – The excellent detector dynamic range for quant in full-scan mode and the high mass accuracy in MS and MS/MS modes enable comprehensive metabolite identification and quantification methods. RUO-MKT-01-1583-A correlation. Traditional software is limited to working with a single sample/control pair at a time. MetabolitePilot Software can easily perform correlation of metabolites across multiple samples, automatically generate plots, and overlay chromatograms, MS, and MS/MS data from multiple samples all within a single workspace. Mass Spectrometry – A generic method was used for data acquisition. The information-dependent acquisition (IDA) method consisted of a time-of-flight mass spectrometry (TOF MS) survey scan followed by two TOF MS/MS scans. The mass range was m/z 100–1000 for both MS and MS/MS. The Dynamic Background Subtraction™ Algorithm was applied for IDA criteria, and a collision energy of 35 eV with a spread of ±10 eV was used for the MS/MS scans. External mass calibration was performed automatically using the calibrant delivery system (CDS). Data processing was performed using MetabolitePilot Software 1.5. Figure 1 demonstrates how an entire time course can be easily evaluated for metabolites arising from a microsome incubation of imipramine. The data is automatically processed in batch mode to generate results tables for all time points. The results for all samples are then selected for correlation. The software automatically tabulates the abundance of each metabolite across all samples, and overlays all chromatographic, MS, and MS/MS data, as shown for a dioxidation metabolite. Multi-sample correlation High performance software – In the past, the work of finding, confirming, and quantifying metabolites has required the use of multiple software packages. MetabolitePilot™ Software can perform all three functions in one software package. Key benefits of the TripleTOF® 600 System and MetabolitePilot Software for metabolite ID workflows Chromatography – Sample analysis was performed on the AB SCIEX TripleTOF® 5600 System coupled with a Shimadzu Prominence UFLC-XR HPLC system. A generic acetonitrile/water/0.1% formic acid gradient was used on a Phenomenex Synergi Polar-RP column 2.5 µm, (2 × 50 mm). Total run time was 3.7 min. Injection volume was 10 µL. Key features of the TripleTOF 5600 system and MetabolitePilot Software for metabolite ID workflows • • • • With increased emphasis on MIST guidelines, correlation of metabolism across multiple samples and estimation of relative amounts of metabolites has become a critical part of data interpretation. For example, the metabolic profile needs to be compared across multiple species to spot a disproportionate metabolite as early as possible. In vitro – in vivo correlation and the study of metabolite kinetics also require multisample DEFINITIVE METABOLITE ID DEFINITIVE METABOLITE ID For Research Use Only. Not for use in diagnostic procedures. Both MS and UV responses can be used as well as other types of analog data such as beta-RAM. A relative response factor can be applied for each metabolite individually to correct the MS response for different metabolites based on the corresponding UV signal. Plots of area counts are generated by selecting one or more metabolites. The user can easily toggle between linear plots and bar graphs (Figure 2), which is more useful when comparing metabolism across multiple species. Automatic summary tables can also be generated showing the area counts or presence/ absence of each metabolite across all samples (Figure 3). Resolution and mass accuracy regardless of scan speed – Instruments employing orbital trapping require slower scanning to obtain higher resolution. The TripleTOF systems maintain approximately 30K resolution regardless of analysis speed. This high-performance, especially in MS/MS mode, provides unambiguous elemental composition assignment and identification of the site of metabolism. MetabolitePilot Software allows batch processing for higher throughput and more efficient use of the chemist’s time. Calibration stability and linearity – Traditional time-of-flight (TOF) instruments typically require internal calibration to maintain mass accuracy, resulting in lowered signals for analytes and a non-linear quantitative response. The TripleTOF systems have been designed to overcome both of these limitations. Resolution and mass accuracy even at low m/z – Traditional TOF instruments provided good mass accuracy and resolution at high m/z. The high detector speed of the TripleTOF systems allow for sufficient resolution at low m/z, providing unambiguous formulae assigments using accurate mass, as well as the isotope and fragmention pattern. Experimental Sample Preparation – Haloperidol and Imipramine were incubated in rat liver microsomes at an initial substrate concentration of 1 µM. Standard high-throughput incubation conditions were used with time points of 0, 5, 15, and 30 min. Protein concentration was 1 mg/mL, and NADPH was present at 4 mM. The reaction was quenched using an equal volume of acetonitrile and then diluted 1:1 with water prior to analysis. The final substrate concentration at t = 0 was 0.25 µM. DRUG METABOLISM www.absciex.com Figure 1: Correlation workspace. Integrated, multi-sample correlation enables comparison of metabolites across a time course from within MetabolitePilot™ Software. In addition to automatic plots of selected metabolites, chromatographic and MS/MS data are also overlaid. www.absciex.com DRUG METABOLISM 83 accuracy of the TripleTOF 5600 system in MS/MS mode, unambiguous elemental compositions of all product ions can be calculated by the software. Integrated MS/MS fragment interpretation This integrated functionality allows the scientist to quickly evaluate different structure proposals in an interactive and user-friendly manner. The structure and the entire fragment assignment are automatically saved in the results workspace as illustrated in Figure 5. MS/MS fragment interpretation and assignment to a proposed metabolite structure is arguably the single most labor-intensive and time-consuming task when identifying metabolites data interpretation. Typically separate software packages were needed for data interpretation which required switching between two separate programs. Other software programs often do not provide integrated storage and reporting of results, proposed structures, and MS/MS fragment assignments that support the proposed structure. Figure 2: Flexible data visualization. The user can quickly switch between line plots and bar graphs of the selected metabolites for time course results and inter-species comparison. Figure 3: Summary tables. Concise summary tables of all selected metabolites across multiple samples are automatically generated in MetabolitePilot™ Software. The user can display presence or absence (top) and the actual area counts of MS or UV data (bottom). 84 Correlation reports are automatically generated, and all tables and charts can be easily copied and pasted from the workspace for preparing presentations. RUO-MKT-01-1583-A Figure 4: Integrated MS/MS interpretation. Directly within the results workspace, structure editing and automatic fragment assignment can be performed for a proposed metabolite. Fragments are assigned and highlighted on the chemical structure. MetabolitePilot Software has a powerful MS/MS fragment interpretation function integrated directly in the results workspace (Figure 4). When reviewing potential metabolites, the scientist can edit structures and perform automatic fragment assignment. Both the structure and fragment assignments are saved in the results table and are directly accessible in data review. Integrated reports can be generated automatically. Fragment assignments are automatically highlighted on the structure when the fragment is selected. The software also automatically shows possible neutral losses from the selected fragment to show its relationship to other product ions in the MS/MS spectrum. A score is assigned to each fragment assignment to aid in data review. The MS/MS spectrum can be automatically annotated with fragment structures and elemental compositions (Figure 4). Due to the high resolution and mass Conclusions New advanced functions in MetabolitePilot Software 1.5 help increase the efficiency of data analysis and interpretation. Multiple sample correlation goes beyond traditional single sample/control processing to enable comparison of metabolites across multiple samples easily and without manual data manipulation in separate software. Integrated MS/MS fragment interpretation improves the efficiency of the most time-consuming aspect of metabolite data analysis by performing fragment assignments on a proposed structure in a single, integrated workspace throughput sample preparation and fast chromatography. • • • DEFINITIVE METABOLITE ID DEFINITIVE METABOLITE ID For Research Use Only. Not for use in diagnostic procedures. References Rapid Metabolite Identification Using MetabolitePilot™ Software”, AB SCIEX Technical Note, Publication 2490211- 01. 1“ 2 R. J. Mortishire-Smith et al, “Evaluation of Chemically Intelligent Acquisition and Processing Tools as a Platform for Discovery Metabolite Identification,” TP 222,The 59th ASMS Conference on Mass Spectrometry and Allied Topics, June 5 – 9, 2011, Denver, CO. 3 To download a trial version of MetabolitePilot™ software, please visit: http://www.absciex. com/Products/Software/MetabolitePilot-Software Figure 5: Results workspace. Metabolite structures are an integral part of the results table and are displayed automatically when a metabolite is selected. DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 85 accuracy of the TripleTOF 5600 system in MS/MS mode, unambiguous elemental compositions of all product ions can be calculated by the software. Integrated MS/MS fragment interpretation This integrated functionality allows the scientist to quickly evaluate different structure proposals in an interactive and user-friendly manner. The structure and the entire fragment assignment are automatically saved in the results workspace as illustrated in Figure 5. MS/MS fragment interpretation and assignment to a proposed metabolite structure is arguably the single most labor-intensive and time-consuming task when identifying metabolites data interpretation. Typically separate software packages were needed for data interpretation which required switching between two separate programs. Other software programs often do not provide integrated storage and reporting of results, proposed structures, and MS/MS fragment assignments that support the proposed structure. Figure 2: Flexible data visualization. The user can quickly switch between line plots and bar graphs of the selected metabolites for time course results and inter-species comparison. Figure 3: Summary tables. Concise summary tables of all selected metabolites across multiple samples are automatically generated in MetabolitePilot™ Software. The user can display presence or absence (top) and the actual area counts of MS or UV data (bottom). 84 Correlation reports are automatically generated, and all tables and charts can be easily copied and pasted from the workspace for preparing presentations. RUO-MKT-01-1583-A Figure 4: Integrated MS/MS interpretation. Directly within the results workspace, structure editing and automatic fragment assignment can be performed for a proposed metabolite. Fragments are assigned and highlighted on the chemical structure. MetabolitePilot Software has a powerful MS/MS fragment interpretation function integrated directly in the results workspace (Figure 4). When reviewing potential metabolites, the scientist can edit structures and perform automatic fragment assignment. Both the structure and fragment assignments are saved in the results table and are directly accessible in data review. Integrated reports can be generated automatically. Fragment assignments are automatically highlighted on the structure when the fragment is selected. The software also automatically shows possible neutral losses from the selected fragment to show its relationship to other product ions in the MS/MS spectrum. A score is assigned to each fragment assignment to aid in data review. The MS/MS spectrum can be automatically annotated with fragment structures and elemental compositions (Figure 4). Due to the high resolution and mass Conclusions New advanced functions in MetabolitePilot Software 1.5 help increase the efficiency of data analysis and interpretation. Multiple sample correlation goes beyond traditional single sample/control processing to enable comparison of metabolites across multiple samples easily and without manual data manipulation in separate software. Integrated MS/MS fragment interpretation improves the efficiency of the most time-consuming aspect of metabolite data analysis by performing fragment assignments on a proposed structure in a single, integrated workspace throughput sample preparation and fast chromatography. • • • DEFINITIVE METABOLITE ID DEFINITIVE METABOLITE ID For Research Use Only. Not for use in diagnostic procedures. References Rapid Metabolite Identification Using MetabolitePilot™ Software”, AB SCIEX Technical Note, Publication 2490211- 01. 1“ 2 R. J. Mortishire-Smith et al, “Evaluation of Chemically Intelligent Acquisition and Processing Tools as a Platform for Discovery Metabolite Identification,” TP 222,The 59th ASMS Conference on Mass Spectrometry and Allied Topics, June 5 – 9, 2011, Denver, CO. 3 To download a trial version of MetabolitePilot™ software, please visit: http://www.absciex. com/Products/Software/MetabolitePilot-Software Figure 5: Results workspace. Metabolite structures are an integral part of the results table and are displayed automatically when a metabolite is selected. DRUG METABOLISM www.absciex.com www.absciex.com DRUG METABOLISM 85 Your success is our success We take it personally AB SCIEX internal expertise As an AB SCIEX customer you have access to an excellent customer support organization. Wherever you are, we’re there with you as a trusted partner to answer questions, provide solutions, PHARMA BUSINESS UNIT Suma Ramagiri and maximize lab productivity. Gary Impey Joseph Fox Debadeep Bhattacharya RESEARCH APPLICATIONS Yves LeBlanc Larry Campbell RESEARCH HARDWARE AND SOFTWARE Bruce Thomson Our customer support organization has access to the latest product updates, software revisions, methods and repair procedures to make sure that you stay on top of your game. When you have questions, we have answers. Learn more at www.absciex.com/customersupport, or locate your local account representative at www.absciex.com/contactus Bruce Collings Igor V Chernushevich Bradley Schneider Tom Covey APPLICATION SUPPORT Elliot Jones Jeff Miller Keith Goodman Carmai Seto Anoop Kumar Kerong Zhang Vicki Gallant For Research Use Only. Not for use in diagnostic procedures. © 2014 AB SCIEX. The trademarks mentioned herein are the property of AB Sciex Pte. Ltd. or their respective owners. AB SCIEX™ is being used under license. 9870414-01 07/2014 RUO-MKT-01-1583-A Headquarters 500 Old Connecticut Path | Framingham, MA 01701 USA Phone 508-383-7700 www.absciex.com International Sales For our office locations please call the division headquarters or refer to our website at www.absciex.com/offices Your success is our success We take it personally AB SCIEX internal expertise As an AB SCIEX customer you have access to an excellent customer support organization. Wherever you are, we’re there with you as a trusted partner to answer questions, provide solutions, PHARMA BUSINESS UNIT Suma Ramagiri and maximize lab productivity. Gary Impey Joseph Fox Debadeep Bhattacharya RESEARCH APPLICATIONS Yves LeBlanc Larry Campbell RESEARCH HARDWARE AND SOFTWARE Bruce Thomson Our customer support organization has access to the latest product updates, software revisions, methods and repair procedures to make sure that you stay on top of your game. When you have questions, we have answers. Learn more at www.absciex.com/customersupport, or locate your local account representative at www.absciex.com/contactus Bruce Collings Igor V Chernushevich Bradley Schneider Tom Covey APPLICATION SUPPORT Elliot Jones Jeff Miller Keith Goodman Carmai Seto Anoop Kumar Kerong Zhang Vicki Gallant For Research Use Only. Not for use in diagnostic procedures. © 2014 AB SCIEX. The trademarks mentioned herein are the property of AB Sciex Pte. Ltd. or their respective owners. AB SCIEX™ is being used under license. 9870414-01 07/2014 RUO-MKT-01-1583-A Headquarters 500 Old Connecticut Path | Framingham, MA 01701 USA Phone 508-383-7700 www.absciex.com International Sales For our office locations please call the division headquarters or refer to our website at www.absciex.com/offices