Scale, Proportion, and Quantity - NSTA Learning Center
advertisement

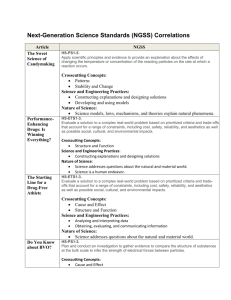
LIVE INTERACTIVE LEARNING @ YOUR DESKTOP NGSS Crosscutting Concepts: Scale, Proportion, and Quantity Presented by: Amy Taylor and Kelly Riedinger March 19, 2013 6:30 p.m. – 8:00 p.m. Eastern time 1 2 http://learningcenter.nsta.org NSTA Learning Center • 10,600+ resources – 3,700+ free! – Add to “My Library” to access later • Community forums • Online advisors to assist you • Tools to plan and document your learning • http://learningcenter.nsta.org 3 Introducing today’s presenters… Ted Willard National Science Teachers Association Amy Taylor University of North Carolina Wilmington Kelly Riedinger University of North Carolina Wilmington 4 Developing the Standards 5 Developing the Standards Assessments Curricula Instruction Teacher Development July 2011 2011-2013 6 Developing the Standards July 2011 7 A Framework for K-12 Science Education Three-Dimensions: View free PDF form The National Academies Press at www.nap.edu 8 Scientific and Engineering Practices Crosscutting Concepts Disciplinary Core Ideas Secure your own copy from www.nsta.org/store Scientific and Engineering Practices 1. Asking questions (for science) and defining problems (for engineering) 2. Developing and using models 3. Planning and carrying out investigations 4. Analyzing and interpreting data 5. Using mathematics and computational thinking 6. Constructing explanations (for science) and designing solutions (for engineering) 7. Engaging in argument from evidence 8. Obtaining, evaluating, and communicating information 9 Crosscutting Concepts 1. Patterns 2. Cause and effect: Mechanism and explanation 3. Scale, proportion, and quantity 4. Systems and system models 5. Energy and matter: Flows, cycles, and conservation 6. Structure and function 7. Stability and change 10 Disciplinary Core Ideas Life Science Physical Science LS1: From Molecules to Organisms: Structures and Processes PS1: Matter and Its Interactions LS2: Ecosystems: Interactions, Energy, and Dynamics LS3: Heredity: Inheritance and Variation of Traits PS2: Motion and Stability: Forces and Interactions PS3: Energy PS4: Waves and Their Applications in Technologies for Information Transfer LS4: Biological Evolution: Unity and Diversity Earth & Space Science Engineering & Technology ESS1: Earth’s Place in the Universe ETS1: Engineering Design ESS2: Earth’s Systems ETS2: Links Among Engineering, Technology, Science, and Society ESS3: Earth and Human Activity 11 Life Science LS1: From Molecules to Organisms: Structures and Processes LS1.A: Structure and Function LS1.B: Growth and Development of Organisms LS1.C: Organization for Matter and Energy Flow in Organisms LS1.D: Information Processing LS2: Ecosystems: Interactions, Energy, and Dynamics LS2.A: Interdependent Relationships in Ecosystems LS2.B: Cycles of Matter and Energy Transfer in Ecosystems LS2.C: Ecosystem Dynamics, Functioning, and Resilience LS2.D: Social Interactions and Group Behavior LS3: Heredity: Inheritance and Variation of Traits LS3.A: Inheritance of Traits LS3.B: Variation of Traits 12 LS4: Biological Evolution: Unity and Diversity LS4.A: Evidence of Common Ancestry and Diversity LS4.B: Natural Selection LS4.C: Adaptation LS4.D: Biodiversity and Humans Earth & Space Science ESS1: Earth’s Place in the Universe ESS1.A: The Universe and Its Stars ESS1.B: Earth and the Solar System ESS1.C: The History of Planet Earth ESS2: Earth’s Systems ESS2.A: Earth Materials and Systems ESS2.B: Plate Tectonics and Large‐Scale System Interactions ESS2.C: The Roles of Water in Earth’s Surface Processes ESS2.D: Weather and Climate ESS2.E: Biogeology ESS3: Earth and Human Activity ESS3.A: Natural Resources ESS3.B: Natural Hazards ESS3.C: Human Impacts on Earth Systems ESS3.D: Global Climate Change Physical Science Engineering & Technology PS1: Matter and Its Interactions PS1.A: Structure and Properties of Matter PS1.B: Chemical Reactions PS1.C: Nuclear Processes ETS1: Engineering Design ETS1.A: Defining and Delimiting an Engineering Problem ETS1.B: Developing Possible Solutions ETS1.C: Optimizing the Design Solution PS2: Motion and Stability: Forces and Interactions PS2.A: Forces and Motion PS2.B: Types of Interactions PS2.C: Stability and Instability in Physical Systems ETS2: Links Among Engineering, Technology, Science, and Society ETS2.A: Interdependence of Science, Engineering, and Technology ETS2.B: Influence of Engineering, Technology, and Science on Society and the Natural World PS3: Energy PS3.A: Definitions of Energy PS3.B: Conservation of Energy and Energy Transfer PS3.C: Relationship Between Energy and Forces PS3.D:Energy in Chemical Processes and Everyday Life PS4: Waves and Their Applications in Technologies for Information Transfer PS4.A: Wave Properties PS4.B: Electromagnetic Radiation PS4.C: Information Technologies and Instrumentation Note: In NGSS, the core ideas for Engineering, Technology, and the Application of Science are integrated with the Life Science, Earth & Space Science, and Physical Science core ideas Developing the Standards Assessments Curricula Instruction Teacher Development July 2011 2011-2013 13 Developing the Standards 2011-2013 14 Closer Look at a Performance Expectation MS-PS1 Matter and Its Interactions Students who demonstrate understanding can: MS-PS1-d. Develop molecular models of reactants and products to support the explanation that atoms, and therefore mass, are conserved in a chemical reaction. [Clarification Statement: Models can include physical models and drawings that represent atoms rather than symbols. The focus is on law of conservation of matter.] [Assessment Boundary: The use of atomic masses is not required. Balancing symbolic equations (e.g. N2 + H2 -> NH3) is not required.] The performance expectations above were developed using the following elements from the NRC document A Framework for K-12 Science Education: Science and Engineering Practices Disciplinary Core Ideas Crosscutting Concepts Developing and Using Models Modeling in 6–8 builds on K–5 and progresses to developing, using and revising models to support explanations, describe, test, and predict more abstract phenomena and design systems. • Use and/or develop models to predict, describe, support explanation, and/or collect data to test ideas about phenomena in natural or designed systems, including those representing inputs and outputs, and those at unobservable scales. (MS-PS1-a), (MS-PS1-c), (MS-PS1-d) PS1.B: Chemical Reactions • Substances react chemically in characteristic ways. In a chemical process, the atoms that make up the original substances are regrouped into different molecules, and these new substances have different properties from those of the reactants. (MS-PS1-d), ( MS-PS1-e), (MS-PS1-f) • The total number of each type of atom is conserved, and thus the mass does not change. (MS-PS1-d) Energy and Matter • Matter is conserved because atoms are conserved in physical and chemical processes. (MS-PS1-d) --------------------------------------------Connections to Nature of Science Science Models, Laws, Mechanisms, and Theories Explain Natural Phenomena • Laws are regularities or mathematical descriptions of natural phenomena. (MS-PS1-d) 15 Note: Performance expectations combine practices, core ideas, and crosscutting concepts into a single statement of what is to be assessed. They are not instructional strategies or objectives for a lesson. Closer Look at a Performance Expectation MS-PS1 Matter and Its Interactions Students who demonstrate understanding can: MS-PS1-d. Develop molecular models of reactants and products to support the explanation that atoms, and therefore mass, are conserved in a chemical reaction. [Clarification Statement: Models can include physical models and drawings that represent atoms rather than symbols. The focus is on law of conservation of matter.] [Assessment Boundary: The use of atomic masses is not required. Balancing symbolic equations (e.g. N2 + H2 -> NH3) is not required.] The performance expectations above were developed using the following elements from the NRC document A Framework for K-12 Science Education: Science and Engineering Practices Disciplinary Core Ideas Crosscutting Concepts Developing and Using Models Modeling in 6–8 builds on K–5 and progresses to developing, using and revising models to support explanations, describe, test, and predict more abstract phenomena and design systems. • Use and/or develop models to predict, describe, support explanation, and/or collect data to test ideas about phenomena in natural or designed systems, including those representing inputs and outputs, and those at unobservable scales. (MS-PS1-a), (MS-PS1-c), (MS-PS1-d) PS1.B: Chemical Reactions • Substances react chemically in characteristic ways. In a chemical process, the atoms that make up the original substances are regrouped into different molecules, and these new substances have different properties from those of the reactants. (MS-PS1-d), ( MS-PS1-e), (MS-PS1-f) • The total number of each type of atom is conserved, and thus the mass does not change. (MS-PS1-d) Energy and Matter • Matter is conserved because atoms are conserved in physical and chemical processes. (MS-PS1-d) --------------------------------------------Connections to Nature of Science Science Models, Laws, Mechanisms, and Theories Explain Natural Phenomena • Laws are regularities or mathematical descriptions of natural phenomena. (MS-PS1-d) 16 Note: Performance expectations combine practices, core ideas, and crosscutting concepts into a single statement of what is to be assessed. They are not instructional strategies or objectives for a lesson. Closer Look at a Performance Expectation MS-PS1 Matter and Its Interactions Students who demonstrate understanding can: MS-PS1-d. Develop molecular models of reactants and products to support the explanation that atoms, and therefore mass, are conserved in a chemical reaction. [Clarification Statement: Models can include physical models and drawings that represent atoms rather than symbols. The focus is on law of conservation of matter.] [Assessment Boundary: The use of atomic masses is not required. Balancing symbolic equations (e.g. N2 + H2 -> NH3) is not required.] The performance expectations above were developed using the following elements from the NRC document A Framework for K-12 Science Education: Science and Engineering Practices Disciplinary Core Ideas Crosscutting Concepts Developing and Using Models Modeling in 6–8 builds on K–5 and progresses to developing, using and revising models to support explanations, describe, test, and predict more abstract phenomena and design systems. • Use and/or develop models to predict, describe, support explanation, and/or collect data to test ideas about phenomena in natural or designed systems, including those representing inputs and outputs, and those at unobservable scales. (MS-PS1-a), (MS-PS1-c), (MS-PS1-d) PS1.B: Chemical Reactions • Substances react chemically in characteristic ways. In a chemical process, the atoms that make up the original substances are regrouped into different molecules, and these new substances have different properties from those of the reactants. (MS-PS1-d), ( MS-PS1-e), (MS-PS1-f) • The total number of each type of atom is conserved, and thus the mass does not change. (MS-PS1-d) Energy and Matter • Matter is conserved because atoms are conserved in physical and chemical processes. (MS-PS1-d) --------------------------------------------Connections to Nature of Science Science Models, Laws, Mechanisms, and Theories Explain Natural Phenomena • Laws are regularities or mathematical descriptions of natural phenomena. (MS-PS1-d) 17 Note: Performance expectations combine practices, core ideas, and crosscutting concepts into a single statement of what is to be assessed. They are not instructional strategies or objectives for a lesson. Closer Look at a Performance Expectation MS-PS1 Matter and Its Interactions Students who demonstrate understanding can: MS-PS1-d. Develop molecular models of reactants and products to support the explanation that atoms, and therefore mass, are conserved in a chemical reaction. [Clarification Statement: Models can include physical models and drawings that represent atoms rather than symbols. The focus is on law of conservation of matter.] [Assessment Boundary: The use of atomic masses is not required. Balancing symbolic equations (e.g. N2 + H2 -> NH3) is not required.] The performance expectations above were developed using the following elements from the NRC document A Framework for K-12 Science Education: Science and Engineering Practices Disciplinary Core Ideas Crosscutting Concepts Developing and Using Models Modeling in 6–8 builds on K–5 and progresses to developing, using and revising models to support explanations, describe, test, and predict more abstract phenomena and design systems. • Use and/or develop models to predict, describe, support explanation, and/or collect data to test ideas about phenomena in natural or designed systems, including those representing inputs and outputs, and those at unobservable scales. (MS-PS1-a), (MS-PS1-c), (MS-PS1-d) PS1.B: Chemical Reactions • Substances react chemically in characteristic ways. In a chemical process, the atoms that make up the original substances are regrouped into different molecules, and these new substances have different properties from those of the reactants. (MS-PS1-d), ( MS-PS1-e), (MS-PS1-f) • The total number of each type of atom is conserved, and thus the mass does not change. (MS-PS1-d) Energy and Matter • Matter is conserved because atoms are conserved in physical and chemical processes. (MS-PS1-d) --------------------------------------------Connections to Nature of Science Science Models, Laws, Mechanisms, and Theories Explain Natural Phenomena • Laws are regularities or mathematical descriptions of natural phenomena. (MS-PS1-d) 18 Note: Performance expectations combine practices, core ideas, and crosscutting concepts into a single statement of what is to be assessed. They are not instructional strategies or objectives for a lesson. Scale, Proportion, and Quantity: A Crosscutting Concept Amy Taylor 19 Kelly Riedinger Who are we? Amy Taylor • • • Associate Professor of science education in the Elementary, Middle Level, Literacy Department at University of North Carolina Wilmington Prior work includes high school teaching in biology and environmental science, graduate research with teachers’ and students’ understanding of scale and nanotechnology Current work (past 5 years) supporting teachers and students in scientific practices Kelly Riedinger • • • 20 Assistant Professor of science education in the Elementary, Middle Level, Literacy Department at University of North Carolina Wilmington Prior work includes middle and high school teaching oceanography, physical science, and earth science as well as teaching in informal science settings (PreK‐8) Current work (past 2 years) includes learning in informal science education settings and preservice teacher preparation Caveats We are not authors of the framework so we have no special insight into the decisions made by the committee. We can use our expertise having worked with teachers and students to help you think about types of scale and how you can engage your students in scaling, proportions, and quantity. 21 Why we find scale interesting • Experiences as a former high school science teacher • New emerging technologies have enabled scientists to observe the extreme scales from the atomic and cosmic sciences • Scale is common in both science and everyday life and impacts all disciplines of science • When we asked scientists to indicate how important scale was to their work, responses included: – – – – 22 ‘‘I can’t operate without a sense of scale.’’ ‘‘Scale is an integral part of what I do.’’ Scale is ‘‘extremely important.’’ ‘‘I think it would be impossible for me to practice without the concept of scale.’’ Overview • What is scale? • Scale, Proportion, and Quantity as a crosscutting concept • Why scale is important? • Approaches to teaching? – Vignettes to illustrate and highlight essential features • Resources • Discussion 23 POLL: What do you first think of when you hear the word scale? 24 POLL: How comfortable are you with the topic of scale and teaching this concept to K‐12 students? Novice Limited Adequate Expert I have no understanding of this concept. I need to learn more about scale before I can teach this topic to students. I have some understanding of scale and I’m ready to try teaching the concept, but I’d like more information and ideas for learning activities. I have an in‐depth understanding of scale and I’m ready to implement learning activities with students. 25 Scale, Proportion, and Quantity The word scale has multiple definitions: Scale can be a device to weigh objects Can cover a fish or a butterfly We scale a wall by climbing Refer to measurement scales such as pH, temperature, or Richter – In science, when we talk about scale we are referring to the properties of an object that can change as size is increased or decreased, and behavior that changes as a result. – – – – 26 Scale, Proportion, and Quantity • Scale is described in terms of range & magnitude. • Three commonly used types of scales in science: Ordinal Interval Logarithmic Enhanced Fujita Scale Saffir‐Simpson scale 27 Kelvin Celsius Richter pH Other Types of Scales Mass Brightness Nano Current Parsecs Architectural Microscopic 28 Geologic Time Decibel scales Light years Voltage Mercali Map scales Temperature Crosscutting Concepts In NGSS Crosscutting concepts bridge boundaries across the various sub‐disciplines of science and engineering. The crosscutting concepts provide students with an organizational framework for making sense of and connecting knowledge across the various science disciplines. 29 Crosscutting Concept: Scale, Proportion, and Quantity The concept of scale, proportion, and quantity spans disciplines in science and engineering. It concerns the sizes of things and the mathematical relationships between elements. Related to this concept, it is important for students to understand what is relevant at different measures and to recognize how changes in scale, proportion, or quantity affect a system’s structure and function. 30 Examples in Physical Science • Atomic Scale • Energy Transfer at different scales • The structure of matter at the atomic and sub‐atomic scales helps to explain a system’s larger scale structures, properties, Scale, and functions Proportion, • Radioactive decay, proportions of isotopes and Quantity • Relationship among different types of quantities can be represented by proportions and ratios (e.g., velocity as a ratio of distance traveled versus time) • Multiple phenomena (e.g., motion, light, sound, electrical and magnetic fields) occur at the macroscopic scale 31 Examples in Life Science Scale, Proportion, and Quantity 32 • Living things are made of cells that can be observed at different scales • Surface area and cell transfer • Living organisms vary in size and scale (e.g., cells whales) • Lifespans vary • Life processes occur at different time scales Examples in Earth Science • The geologic time scale depicts the relative times of events in Earth’s history • Scale models are used to represent phenomena too large or small to observe (e.g., Earth‐Sun‐Moon models) Scale, • Analyses of rock strata and the fossil record provide only relative Proportion, dates, not an absolute scale • Geologists use relative positions to estimate dates and Quantity • Relative distances of the sun and other stars from one other • Relationship between distance of stars and their apparent brightness • Topographic maps use scale to represent relief and surface features 33 Scale, Proportion, and Quantity in the Next Generation Science Standards Physical Science Elementary School 34 Relative scales allow objects to be compared and described (e.g., bigger and smaller; hotter and colder; faster and slower). 2‐PS1‐d Standard units are used to measure and describe physical quantities such as weight, time, temperature, and volume. 5‐PS1‐c Earth Science Natural objects and observable phenomena exist from the very small to the immensely large. 5‐ ESS1‐a Scale, Proportion, and Quantity in the Next Generation Science Standards Middle School Physical Science Life Science Earth Science Proportional relationships (e.g. speed as the ratio of distance traveled to time taken) among different types of quantities provide information about the magnitude of properties and processes. MS‐PS2‐b Phenomena that can be observed at one scale may not be observable at another scale. MS‐LS1‐a Time, space, and energy phenomena can be observed at various scales using models to study systems that are too large or too small. MS‐ESS1‐c, MS‐ESS1‐e, MS‐ESS1‐f, MS‐ESS1‐g Scientific relationships can be represented through the use of algebraic expressions and equations. MS‐PS2‐b 35 Time, space, and energy phenomena can be observed at various scales using models to study systems that are too large or too small. MS‐LS2‐g Scale, Proportion, and Quantity in the Next Generation Science Standards High School 36 Life Science Earth Science The significance of a phenomenon is dependent on the scale, proportion, and quantity at which it occurs. HS‐LS2‐a Patterns observable at one scale may not be observable or exist at other scales. HS‐ESS1‐a, HS‐ESS1‐I Using the concept of orders of magnitude allows one to understand how a model at one scale relates to a model at another scale. HS‐LS2‐b Algebraic thinking is used to examine scientific data and predict the effect of a change in one variable on another (e.g. linear growth vs. exponential growth). HS‐ESS1‐g Algebraic thinking is used to examine scientific data and predict the effect of a change in one variable on another (e.g., linear growth vs. exponential growth). HS‐LS3‐d The significance of a phenomenon is dependent on the scale, proportion, and quantity at which it occurs. HS‐ESS2‐a Using the concept of orders of magnitude allows one to understand how a model at one scale relates to a model at another scale. HS‐ESS2‐f Scale, Proportion, and Quantity: Progression K‐2: Measurement; Counting, compare quantities, order quantities; Use of scale models, diagrams, and maps 3‐5: Measurement with standard units; Understanding that with natural objects scales range from very small to immensely large; Construct and interpret data models and graphs MS: Estimation; Powers of 10 scales; Use algebraic thinking and equations; Recognize the function of a system may change with scale and that phenomena observable at one scale may not be observable at another scale HS: Move back and forth between models at various scales; Understand that the significance of a phenomenon is dependent on the scale at which it occurs; Use more complex algebraic thinking and statistical relationships 37 The Next Generation Science Standards and Scale The Framework Identifies 8 Science & Engineering Practices Asking questions and defining Using mathematics and problems computational thinking Developing and using models Developing explanations and designing solutions Planning and carrying out Engaging in argument from investigations evidence Analyzing and interpreting data 38 Obtaining, evaluating, and communicating information The Next Generation Science Standards and Scale The Framework Identifies 8 Science & Engineering Practices Asking questions and defining Using mathematics and problems computational thinking Developing and using models Developing explanations and designing solutions Planning and carrying out Engaging in argument from investigations evidence Analyzing and interpreting data 39 Obtaining, evaluating, and communicating information The Next Generation Science Standards and Scale The Framework Identifies 8 Science & Engineering Practices Asking questions and defining Using mathematics and problems computational thinking Developing and using models Developing explanations and designing solutions Planning and carrying out Engaging in argument from investigations evidence Analyzing and interpreting data 40 Obtaining, evaluating, and communicating information The Next Generation Science Standards and Scale The Framework Identifies 8 Science & Engineering Practices Asking questions and defining Using mathematics and problems computational thinking Developing and using models Developing explanations and designing solutions Planning and carrying out Engaging in argument from investigations evidence Analyzing and interpreting data 41 Obtaining, evaluating, and communicating information The Next Generation Science Standards and Scale The Framework Identifies 8 Science & Engineering Practices Asking questions and defining Using mathematics and problems computational thinking Developing and using models Developing explanations and designing solutions Planning and carrying out Engaging in argument from investigations evidence Analyzing and interpreting data 42 Obtaining, evaluating, and communicating information Quick Write Prompts • What are some examples of ways you have used scale, proportion, and quantity in your classroom? [Type your responses in the Chat.] 43 Time to Chat • Any other questions? 44 Science Teaching Examples: Measurement • Tools of measurement – Physical properties (e.g., meter stick, graduated cylinder, balance, electronic scale) – Weather data tools (e.g., barometer, thermometer, rain gauge, wind vane) – Oceanography tools (e.g., current cross, secchi disk, salinometer, pH water test kit) 45 Science Teaching Examples: Scale • Types of scales (e.g., Geologic time scale, Fujita tornado scale, pH scale) • Relative scales (e.g., bigger vs. smaller, colder vs. warmer) • Scaled maps, models, diagrams – Topographic maps – Earth‐Sun‐Moon models – Dinosaur models – Ocean floor topography 46 Science Teaching Examples: Quantity • Counting quantities (e.g., bacteria, leaves on a branch, number of flowering buds) • Comparisons of counting • Ordering quantities • Creating, analyzing and interpreting graphs 48 POLL Which item would be in the “middle” if you were to arrange them from smallest to largest? A. Width of football field B. School bus C. Thickness of a penny D. Diameter of a human hair E. Length of an adult’s shoe 49 POLL Which item would be in the “middle” if you were to arrange them from smallest to largest? A. Distance from Earth to International Space Station B. Diameter of Earth C. Distance you could walk in 10 minutes D. Distance from Earth to Sun E. Distance from Earth to Moon 50 POLL Which item would be in the “middle” if you were to arrange them from smallest to largest? 1. Diameter of DNA strand 2. Diameter of a proton 3. Size of a hydrogen atom 4. Diameter of typical cell 5. Size of a typical small molecule 51 POLL Can you assign the actual size to the item? Size of a typical small molecule: A. 10 ‐12 B. 10 ‐5 C. 10 ‐15 D. 10 ‐10 E. 10 ‐9 52 Why is it important? • Fascination with the size and scale of things • What research says… – How people understand scale in terms of: • Learning of scale • Powers of Ten • Measurement and estimation • Use of scale in work/school 53 Students’ Thinking… A little girl was riding in an airplane and while the plane was taking off she turned around to her parents and said: “When do we get small?” 54 As teachers…do we care if students are off by a factor of… – 10? – 100? – 1000? – 1,000,000? 55 A Sense of Scale Understanding Scale Teachers Students • Most accurate in their • More difficulty with sizes knowledge of human scale outside the human scale • Being able to directly • Found small scales more experience objects and difficult to conceptualize than distances influenced by large scales concepts of size and scale • Aware of very small and large • Teachers hold more accurate objects but lacked accurate concepts of large scale than knowledge of the exact sizes, as small scale well as their relative sizes 56 Body Rulers • Study examined the impact of teaching students to use their bodies as rough measurement tools • Results showed that teaching students to use body rulers for estimation had a significant • influence on their estimation accuracy • Proportional reasoning was significantly correlated with students’ measurements 57 Hopefully giving them a lifelong tool that they could use to make linear measurements and estimations Powers of Ten • Study that examined the impact of the film Powers of Ten on middle school students’ understanding of ‘‘size and scale’’ • Students’ proportional reasoning ability was found to be positively correlated with their accuracy of ordering objects and assigning them with correct size labels 58 (Eames Office, 2009) 59 (Eames Office, 2009) 60 (Eames Office, 2009) 61 (Eames Office, 2009) 62 (Eames Office, 2009) 63 (Eames Office, 2009) 64 (Eames Office, 2009) 65 (Eames Office, 2009) 66 (Eames Office, 2009) 67 (Eames Office, 2009) 68 (Eames Office, 2009) 69 (Eames Office, 2009) 70 (Eames Office, 2009) . 71 (Eames Office, 2009) Over 50 Scientists & Community Professionals Used scale to… – Design buildings – Discover ancient cultures – Track hurricanes – Design equipment – Create sculpture – Build a home – Survey a stream 72 • Repeatedly these individuals said, ‘scale is my job’, ‘scale is in everything I do’, ‘it is essential to my job’, and ‘scale is critical.’ • Across disciplines, understanding the sizes of things and scale is essential to understanding phenomena and processes. • To be effective in their job, they needed to be able to move from small‐scale to large‐scale flexibly. Time to Chat • What questions do you have? • How has the content presented so far influenced your thinking about teaching scale to students? 73 Implications For Teaching Examples of lessons using scale Grade Life Science Earth Science 3‐5 Cartesian Diver Lab 6‐8 9‐12 74 Physical Science Topographic Maps Sea Floor Mapping Cell Size Sea Floor Mapping Sample Activity: Elementary School Cartesian Diver • Demonstrate ratios of density and pressure • How it works: – Squeezing the bottle increases the pressure and compresses the air in the diver (represented through dropper, ketchup packet, etc.). – This increases the density of the diver, thus changing the buoyancy and causing it to sink. 75 Sample Activity: Middle School Creating Topographic Maps • Use Play‐doh® to create a landscape. • Measure and mark off 2cm sections from the base of the landscape to the top. • Use fishing line to cut a layer for each of the marked sections. Place each section on paper and trace around. Repeat with remaining marks. USGS Activity • Have students note and compare the landforms to their created maps. Help http://vulcan.wr.usgs.gov/Outreach them to make connections between this /Publications/GIP19/chapter_three activity and topographic maps. _play‐dough_topo.pdf • As an extension, use real topographic maps and have students create the landforms using their Play‐doh®. 76 Sample Activity: Middle/High School Mapping the Ocean Floor NOAA Activity http://csc.noaa.gov/psc/seamedia/ Lessons/G5U4L3%20Seafloor%20Pr ofiling.pdf 77 Atomic Force Microscope Similar technique of “determining topography” at the nanoscale! 78 Sample Activity: High School Cell Size and Surface Area Extreme Science Activity 79 • Obtain three agar/potato cubes: – 1 cm3, 2 cm3, 3 cm3 • Place the cubes in the beaker and pour in enough diffusion medium to cover them and soak for 20 minutes. • Cut the cubes in half and examine and compare their inside appearance. • Measure the depth of the colored zones for each cube in mm and record data. Effects of Scale Parameters Case I Case III Case V Case VII Case IX Length 1 3 5 7 9 Face area 1 9 25 49 81 Surface area 6 54 150 294 486 Volume 1 27 125 343 729 Area/Volume ratio 6 2 1.2 0.86 0.67 Volume: Length x Width x Height Surface Area: Length x Width x 6 (# of faces of cube) 80 Examples of the impact of increasing surface area include: • Why we chew food before swallowing (more surface area leads to faster digestion in the stomach) • Villi in intestines and alveoli lungs • Why elephant ears are so large (more surface area leads to faster cooling rates) • Decreasing surface area helps an animal retain body heat, such as when a dog curls up outside on a cold day • Volume of single‐celled organisms is restricted by the need for metabolites to reach interior of the cell solely by diffusion As scales change, surface area to volume relationships have significant influences on physical, chemical, geological, and biological processes and phenomena. 81 POLL: How comfortable are you with the topic of scale and teaching this concept to K‐12 students? Novice Limited Adequate Expert I have no understanding of this concept. I need to learn more about scale before I can teach this topic to students. I have some understanding of scale and I’m ready to try teaching the concept, but I’d like more information and ideas for learning activities. I have an in‐depth understanding of scale and I’m ready to implement learning activities with students. 82 Suggestions for Teaching Scale Take time to emphasize sizes and scales Verbalize reasoning across scales Teach students to estimate (body rulers and pacing) Teach measurement and various units The Powers of Ten video works! Teach them benchmark sizes and how to reason with benchmarks • Encourage curiosity and scale thinking across disciplines • Awareness of emerging field of nanotechnology • • • • • • 83 Other Scale Resources Eames Office Website. Powers of Ten Film at http://www.powersof10.com/film Jones, M.G., Taylor A., & Falvo, M. (2009). Extreme Science. Arlington VA: NSTA Press, 356 pages. Jones, M.G., Falvo, M., Taylor, A., & Broadwell, B. (2007). Nanoscale Science. Arlington VA: NSTA Press, 155 pages. Nanoscale Science Education: http://www.ncsu.edu/project/scienceEd/ Taylor, A., Jones, M.G., & Pearl, T.P. (2008). Bumpy, sticky, and shaky: Nanoscale science and the curriculum. Science Scope, 31(7), 28‐35. 84 ACKNOWLEDGEMENT This material is based upon work supported by the NSF under Grants No. 0411656, and 0507151 All research based on collaboration with M. Gail Jones, Professor of Science Education from North Carolina State University 85 Contact Information Amy Taylor taylorar@uncw.edu 86 Kelly Riedinger riedinger@uncw.edu NSTA Resources on NGSS www.nsta.org 87 NSTA Resources on NGSS www.nsta.org/ngss 88 Community Forums 89 NSTA Print Resources NSTA Reader’s Guide to the Framework 90 NSTA Journal Articles about the Framework and the Standards NSTA National Conference The place to be to learn about San Antonio, Texas April 11-14 91 Web Seminars on Crosscutting Concepts Feb. 19: Patterns March 5: Cause and effect: Mechanism and explanation March 19: Scale, proportion, and quantity April 16: Systems and system models April 30: Energy and matter: Flows, cycles, and conservation May 14: Structure and function May 28: Stability and change All sessions will take place from 6:30-8:00 p.m. Eastern time on Tuesdays 92 Web Seminars on NGSS Archives of past programs Fall 2012 Scientific and Engineering Practices (series of 8) Winter/Spring 2013 Second Draft of NGSS Engineering in NGSS NGSS in the Elementary Grades Connecting NGSS with Common Core Math and ELA Crosscutting Concepts series http://learningcenter.nsta.org/products/symposia_seminars/ NGSS/webseminar.aspx 93 on NGSS Moving Toward NGSS: Using Formative Assessment to Link Instruction and Learning Members: $179; Non-members $199 Live web seminars on April 18, 25, May 2 Presenter: Page Keeley Moving Toward NGSS: Visualizing K-8 Engineering Education Members: $179; Non-members $199 Live web seminars on May 16, 23, 30 Presenters: Christine Cunningham and Martha Davis Register at: learningcenter.nsta.org/ngss 94 Thanks to today’s presenters! Ted Willard National Science Teachers Association Amy Taylor University of North Carolina Wilmington Kelly Riedinger University of North Carolina Wilmington 95 Thank you to the sponsor of today’s web seminar: This web seminar contains information about programs, products, and services offered by third parties, as well as links to third-party websites. The presence of a listing or such information does not constitute an endorsement by NSTA of a particular company or organization, or its programs, products, or services. 96 National Science Teachers Association David Evans, Ph.D., Executive Director Zipporah Miller, Associate Executive Director, Conferences and Programs NSTA Web Seminar Team Al Byers, Ph.D., Assistant Executive Director, e-Learning and Government Partnerships Brynn Slate, Manager, Web Seminars, Online Short Courses, and Symposia Jeff Layman, Technical Coordinator, Web Seminars, SciGuides, and Help Desk 97