Supplement Section (doc 30K)
advertisement

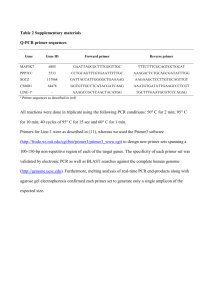
Manuscript number LLO6-LEU-0810 " Telomerase Expression in B-Cell Chronic Lymphocytic Leukemia Predicts Survival and Delineates Subgroups of Patients with the Same IgVH Mutation Status and Different Outcome" Data Supplements Primers, probes and standard curve construction for the quantification of hTERT transcripts To quantify the mRNA of hTERT, two sets of primers were designed. The forward primer AT1 (5'CGGAAGAGTGTCTGGAGCAA-3') and the reverse primer AT2 (5'- GGATGAAGCGGAGTCTGGA-3') bind to nucleotide sequences nt 1784-1803 and nt 1904-1926, respectively, (Genbank accession number: AF015950), located upstream of the RT motif 1 of the hTERT gene. The forward primer FL1 (5'-TGTACTTTGTCAAGGTGGATGTGA-3') and the reverse primer FL2 (5'-GCTGGAGGTCTGTCAAGGTAGAG-3') bind to nucleotide sequences located within the α and β splicing sites, respectively (nt 2172-2195 and nt 2344-2366 ; Genbank accession number: AF015950) (Figure 1A). While amplification with AT1 and AT2 primers allowed the detection of all hTERT transcripts, amplification with FL1 and FL2 primers recognized only mRNA containing the α and β sites and encoding the functional hTERT protein (15). The fluorogenic probes AT (FAM 5'-TTGCAAAGCATTGGAATCAGACAGCAC-3' TAMRA) and FL (FAM 5'-CGCGTACGACACCAT-3' MGBNFQ), which recognize sequences located within the products amplified by AT1/AT2 and FL1/FL2 primer pairs, respectively, were synthesized by PE Applied Biosystems. The standard reference curve to quantitate hTERT-AT and hTERT-FL transcripts was performed using serial 5-fold dilutions of the hTERT amplicon we constructed as follows: RNA from BL41 cells, strongly expressing the hTERT gene, was extracted and retrotranscribed as described above, and cDNA was amplified using primers F (5'ATGTCACGGAGACCACGTTT-3') and R (5'-AGAGCAGCGTGGAGAGGAT-3') which bind to nucleotide sequences located upstream and downnstream, respectively, of regions recognized by AT1/AT2 and FL1/FL2 primer pairs (Figure 1A). Amplification was performed in a 50 μl mixture containing 1μl Taq Gold (5 U/μl; Applied Biosystems), 5 μl Buffer 10X (500 mM KCl, 100mM Tris-HCl, pH 8.3), 3μl MgCl2 (25mM), 200μM dNTP, and 35 pmol of each F and R primer, and carried out for 35 cycles, each consisting of 30 sec at 95°C, 30 sec at 60°C, and 30 sec at and 72°C, in a thermal cycler (9600 Perkin Elmer). The amplicon of 840 bp was run on an agarose gel, purified by using NucleoSpin Extract kit (M-Medical) according to the manufacturer's instructions, and quantified by spectrophotometer. Real-time PCR for quantification of hTERT transcripts All PCR reactions were performed using an ABI prism 7700 Sequence Detection System (PE Applied Biosystems). Each PCR was performed in 50 μl of mixture containing 25 μl 2X TaqMan Universal (PE Applied Biosystems), 100nM of fluorogenic probe, 300nM of each primer, and 10 μl of appropriately diluted cDNA samples (standard curve points and experimental samples). After 2 min at 50°C to allow the uracil N-glycosylase to act, and a denaturation step of 10 min at 95°C, 45 cycles were run each consisting of 30 sec at 95°C, 30 sec at 60°C, and 30 sec at and 72°C. Each sample was run in triplicate; a threshold cycle (Ct) value for each triplicate was calculated by determining the point at which the fluorescence exceeded a threshold limit (10-fold the standard deviation of baseline), and the mean values of the replicates, plotted against the standard hTERT reference curve, was used to calculate the copies of hTERT transcripts in the sample (Figure 1B). To normalize hTERT transcripts for the amount and quality of RNA, 10 μl cDNA of each sample was amplified under the above conditions, by using the GAPDH Kit (PE Applied Biosystems). The mean Ct values of the two replicates, plotted against the standard GAPDH reference curve, were used to calculate the copies of GAPDH in the sample. Values were then normalized for 106 copies of GAPDH. Figure supplement . Real-time PCR to quantify hTERT transcripts. (A) Primer pair AT1 and AT2 bind to nucleotide sequences located upstream of the RT motif 1 on the hTERT gene and allow amplification of all hTERT transcripts (hTERT-AT). Primer pair FL1 and FL2 bind to nucleotide sequences located within the α and β splicing sites, respectively, and allow the amplification of the full-length mRNA encoding the functional hTERT protein (hTERT-FL). Primer pair F and R were employed to obtain the amplicon FR containing both regions recognized by either the AT1/AT2 or FL1/FL2 primer pairs. (B) The reference standard curves for quantification of hTERT-AT and hTERT-FL transcripts were constructed by five-fold serial dilution of the amplicon FR amplified with the hTERT-AT and hTERT-FL primer pairs, respectively.