Chemical Energy File
advertisement

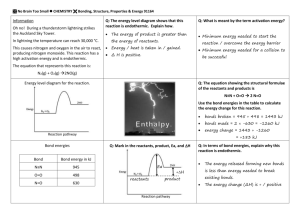
CHEMICAL ENERGY Energy is what makes chemical reactions happen. Some chemical reactions occur when energy is released (often as heat) during the reaction, and this release (“freeing”) of energy drives the reaction forward, like water flowing down a waterfall. Chemical reactions that release energy are called exothermic reactions. Other chemical reactions occur only when energy is absorbed from an outside source, e.g. heat from a Bunsen burner; in this case energy needs to be absorbed during the reaction. Chemical reactions that absorb energy are called endothermic reactions. Check: Spontaneous reactions, i.e., reactions occurring on their own without continuous intervention, are usually also ______________________________________. Check: So where is this chemical energy stored? If chemical energy is released, it is released from where? If chemical energy is absorbed, it is absorbed to where? The answer is ___________________________________________________________. A chemical reaction can be viewed as a process where existing chemical bonds are broken and new, different chemical bonds are formed. This process results in a change in matter (chemistry is defined as the study of matter and its changes). For either endothermic or exothermic chemical reactions, the following is true: Energy must be absorbed in order to break chemical bonds Energy is always released when chemical bonds are formed. All chemical reactions involve the breaking and making of bonds. Check: If it takes more energy to break the existing bonds than is released when the new bonds are formed, then the reaction will be _____________________________. If it takes less energy to break the existing bonds than is released when the new bonds are formed, then the reaction will be _____________________________. Reaction Energy Diagrams The progress of this change in energy during a chemical reaction can be diagrammed to distinguish exothermic and endothermic reactions. The y-axis of the diagram shows “energy content” of the matter and the x-axis shows time (progress of the reaction). Exothermic reaction: 106735318 Endothermic reaction: 1 Thermochemical Equations The energy of a chemical reaction ( also called the heat of reaction) is a quantitative part of the chemical reaction. A chemical equation that includes the heat of reaction is called a “thermochemical equation.” One way to write thermochemical equations is (“Mass+Energy Form”): If a reaction is endothermic, then show the heat of reaction as a reactant (i.e., on the left side of the equation). If a reaction is exothermic, then show the heat of reaction as a product (i.e., on the right side of the equation). The quantity of heat is related to the molar ratios of reactants and products shown by the coefficients of the formulas, but the heat of reaction itself never has a coefficient.. Examples (1) CoCO3(s) + 81.6 kJ CoO(s) + CO2(g) This reaction is ______________________ ( “81.6 kJ are absorbed when 1 mol CoCO3 is heated to produce 1 mol CoO and 1 mol CO2”). (2) C(s) + O2(g) CO2(g) + 393.5 kJ This reaction is ______________________ (“393.5 kJ are released when 1 mol C reacts with 1 mol O2 to produce 1 mol CO2”). The SI unit of energy is the joule (J). Check: 1 kJ = _________J (chemical energy can also be expressed in units of calories (cal), where 1 cal = 4.18 J). Enthalpy Change The “energy content” of matter is called the “enthalpy” and is represented by the symbol, H. The heat of reaction is the difference in enthalpy between the reactants and the products of the reaction and is represented by the symbol, ΔH (spoken as “change in enthalpy,” or “enthalpy change,” or simply “delta H.”). ΔH has the same magnitude as the heat of reaction shown in the thermochemical equations above written in Mass+Energy Form, but the sign can be positive (+) or negative (-) (see “conventions” below). Check: How would you show ΔH in the reaction energy diagrams shown above? So another way to write a thermochemical equations is (“ΔH Form”): Write the chemical equation as usual Show the value of ΔH following the chemical equation in the form: ΔH = +/- value Examples (3) CoCO3(s) CoO(s) + CO2(g) (corresponds to equation (1) above) (4) C(s) + O2(g) CO2(g) (corresponds to equation (2) above) 106735318 ΔH = +81.6 kJ ΔH = -393.5 kJ 2 Conventions for ΔH Convention I: There is an important convention regarding use of the numeric sign on ΔH to show the difference between exothermic and endothermic reactions: The ΔH for an exothermic reaction has a NEGATIVE value, because an exothermic reaction involves the loss (release) of heat by the matter. The ΔH for an endothermic reaction has a POSITIVE value, because an endothermic reaction involves the gain (absorption) of heat by the matter. Convention II: Another important convention involves the units of ΔH. ΔH is often given in units of kJ/mol of one of the reactants irrespective of the coefficient on that reactant in the equation. However, if ΔH is given in just kJ (as in the examples above), then the kJ correspond to the molar ratios shown in the equation AS IT IS WRITTEN. How to handle these two different expressions of units for ΔH is shown in the section below on stoichiometry. Problem 1 Write balanced thermochemical equations for the following chemical reaction USING BOTH FORMS for writing thermochemical equations as described above, and state whether the reaction is exothermic or endothermic: Chlorine gas (Cl2) reacts with sodium bromide (NaBr) to produce sodium chloride (NaCl) and bromine gas (Br2). The heat of reaction is -100.18 kJ. 106735318 3 Estimating ΔH from the Energy of Chemical Bonds The value of ΔH is the amount of energy released or absorbed by a chemical reaction. Since that amount of energy is also the difference between the energy needed to break bonds and the energy needed to make bonds, then we should be able to predict the value of ΔH by calculation. Of course in order to be able to do this, we need to know: How much energy is needed to break one molar quantity of each type of chemical bond shown in the reactants of a chemical reaction (called the “bond energy”) How much energy is released when one molar quantity of each type of new chemical bond is formed in the products of a chemical reaction (this would be the negative of the bond energy) The general formula for calculating ΔH from bond energies is: ΔH = (sum of energies of all reactant bonds) – (sum of energies of all product bonds) Notice that the “-“ sign making the different takes care of the sign of ΔH (convention I above). It is most important to take into consideration the molar quantities of each substance given by the coefficients of the chemical equation as well as the number of each type of bond that shows up in each molecule. E.g., H2O has two O-H bonds in its structure; “2H2O” in a chemical equation would then require you to count a total of 4 O-H bonds (i.e., 2 moles of H2O containing 2 O-H bonds each). The way to calculate the energy of all the bonds for a given molecule in an equation (whether reactant or product) is: (coefficient of the molecule with that bond type) x (number of bonds of that type appearing in the structure of one molecule) x (bond energy of that bond type) The units of this calculation are: mol x (dimensionless ratio) x kJ/mol = kJ These energies must then be calculated for all the different bond types in the molecule and for all molecules in the chemical equation, and then plugged into the equation for ΔH. Yikes!!!!!!! This sounds complicated, but we will only look at relatively simple cases, in particular, only for gases, since it is much more complicated for solids and liquids. Example Problem Estimate ΔH from bond energies for the following reaction: 2H2 (g) + O2 (g) 2H2O (g) bond energies: Solution: H-H 436 kJ / mol O=O 496 kJ / mol O-H 463 kJ / mol ΔH = (2 x 1 x 436) + (1 x 1 x 496) – (2 x 2 x 463) = - 484 kJ Problem 2 Estimate ΔH from bond energies for the reaction: N2 (g) + 3H2 (g) 2NH3 (g) bond energies (in kJ/mol) are: N≡N 944 and N-H 388 106735318 4 Stoichiometry of Thermochemical Equations Using the methods of quantitative chemistry, the maximum theoretical quantity of heat released or absorbed during a reaction can be calculated from the ΔH and the molar ratios of reactants or products involved by using a balanced equation and the given heat of reaction. Example: How much heat would be released by the complete combustion of 365 g of carbon in the presence of excess oxygen as given by equation (2 or 4) above? Solution: The ΔH for this reaction is -393.5 kJ for 1 mol of carbon as written. Step 1: Calculate how many moles of C is given by 365 g of C (365 g C) / (12 g/mol C) = 30.4 mol C. Step 2: Use the molar ratio given by the equation to set up a proportion (30.4 mol C) x (-393.5 kJ/mol C) = - 12 000 kJ Note: In the example of the previous section, we calculated ΔH = - 484 kJ for the reaction: 2H2 (g) + O2 (g) 2H2O (g). This value of ΔH applies to the “equation as written” so we could also write ΔH = - 484 kJ / 2 mol H2. If you were to look up the ΔH for this reaction in a handbook, you would probably find ΔH = - 242 kJ/mol of H2. So you would need to multiply the handbook value x coefficient of H2 in the equation (“2” in our case) to get ΔH = - 484 kJ. Problem 3 1. Refer to the reaction in Problem 1. How much total heat could be released by the reaction of 361 g sodium bromide with excess chlorine gas? Combustion Reactions The chemical reaction shown in equation (2) or (4) in the examples above is itself an example of a “combustion reaction,” which is the reaction of some substance with oxygen. “Combustion” is commonly referred to as “burning,” e.g., of wood, paper, natural gas, etc. The materials we usually think of as “fuels” are often “hydrocarbons,” i.e., their chemical composition consists of the elements carbon and hydrogen, e.g., methane (CH4), butane (C4H10), or gasoline (C8H18). Combustion reactions are always exothermic. The thermochemical equation (skeleton) for the “complete combustion” of a hydrocarbon: hydrocarbon + O2 CO2 + H2O + heat of combustion (kJ) or hydrocarbon + O2 CO2 + H2O , ΔH = - heat of combustion (kJ) Check: What substance does the “complete combustion” of carbon produce? _____________________ What substance does the combustion of hydrogen produce? _______________________ 106735318 5 Exercise 1 Name ____________________________ Date ________ 1. You are planning a steak cookout for your friends. If it requires 9850 kJ of grill heat to cook one steak, how much charcoal would you need in order to cook 12 steaks? Assume charcoal consists of just carbon and all combustion is complete. 2. In the process of making cement, calcium carbonate is heated to produce calcium oxide and carbon dioxide gas according to the following equation: CaCO3(s) CaO(s) + CO2(g) ΔH = +176 kJ How much heat is needed to produce 952 g of CaO from CaCO3? 3. The reaction of nitrogen monoxide gas (NO) gas with oxygen gas (O2) produces nitrogen dioxide gas (NO2). The heat of combustion is -57 kJ/mol NO. What is the total heat change when 1.24 g NO are reacted? 4. By using the heat-measuring method of calorimetry, you determine that the complete combustion of 17.6 g of propane (C3H8) releases 888 kJ of heat. a) What is the heat of combustion in kJ/mol of propane? b) Write a balanced thermochemical equation for the complete combustion of propane. 106735318 6