World Journal of Engineering SYNTHESIS, characterization and in
advertisement
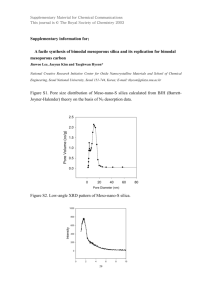
World Journal of Engineering SYNTHESIS, CHARACTERIZATION AND IN VITRO BIOACTIVITY OF SOL-GEL DERIVED MESOPOROUS BIOACTIVE GLASS Yu-Ren Wu, Lay Gaik Teoh*, Pei-Hsing Huang and Wei Haw Wu Department of Mechanical Engineering, National Pingtung University of Science and Technology, Neipu, Pingtung 91201, Taiwan. Introduction temperature. Mesoporous bioactive materials are characterized as having high specific surface areas and pore volume, and uniform channel diameters [1-4] have received much interest for applications in bone tissue regeneration and drug delivery [3]. Meanwhile, mesoporous bioactive materials exhibit better in vitro bone-forming bioactivity compared to conventional sol-gel derived bioactive glasses [3, 5]. Therefore, it is speculated that the pore size, pore volume and pore structure of the bioactive materials play important roles in their properties. In this work we demonstrate the synthesis of mesoporous SiO2-CaO-P2O5 bioactive glass with a high specific surface area and pore volume produced by a sol-gel process with Ca, P and Si sources, and with a combination of surfactant templating by using a triblock copolymer as the template. The in vitro bioactivity of the mesoporous SiO2-CaO-P2O5 was also investigated, demonstrated by formation of apatite nanocrystals after soaking the mesoporous SiO2-CaO-P2O5 in SBF. Results and Discussion The TGA curve at the heating rate of 5 °C/min for the as-synthesized bioactive glass sample synthesized with a triblock copolymer as the template is depicted in Fig. 1. The TGA result suggests a two-step process: the desorption of ethanol solution at about 80 °C, the subsequent loss of the triblock copolymer between 100 and 210 °C. However, the residual triblock copolymer is incompletely removed from the as-synthesized bioactive glass upon calcination at 210 °C. Therefore, the calcination temperature was fixed at 600 °C in the later experimental design to completely remove this. Experimental In a typical synthesis, 2 g of F108 (Aldrich) was dissolved in 25 ml of ethanol. Afterward tetraethyl orthosilicate (TEOS, Acros), calcium nitrate (Ca(NO3)2.4H2O, Aldrich), and triethyl phosphate (TEP, Aldrich) were used as SiO2, CaO, and P2O5 sources, respectively, and were added under vigorous stirring for 1 h. The sol was introduced into a petri dish at room temperature. The gel was aged for 2 days, and then the dried gel was calcined at 600 °C for 3 h to obtain the final calcined mesoporous bioactive glass. The mesostructure of SiO2-CaO-P2O5 obtained was then investigated by thermogravimetric analysis (SETSYS Evolution TGA), N2 adsorption-desorption isotherm measurements (Micromeritics, ASAP 2010), powder X-ray diffraction (Rigaku, D/max-Ⅳ, using Cu-Kα radiation), and transmission electron microscopy (FEGTEM; FEI E.O Tecnai F20 G2 Field-Emission TEM, 200 keV). The assessment of in vitro bioactivity was carried out in a SBF solution (pH 7.4) at 37 °C. The SBF solution has a composition and concentration similar to that of human plasma. After soaking for 10 h, the sample was removed from the SBF, washed with deionized water, and dried at room Fig. 1. TGA curve of the as-synthesized SiO2-CaO-P2O5 produced with a triblock copolymer as the template. The N2 adsorption-desorption isotherm plot of the prepared mesoporous bioactive glass was measured and is shown in Fig. 2, together with the corresponding pore size distribution. The curve can be identified as a type IV isotherm with a well-defined P/P0 step between 0.4 and 0.8, which indicates that the mesoporous bioactive glass has a mesoporous structure. The sample shows type H1 hysteresis loop between adsorption and desorption modes, according to BDDT classification, which is indicative of mesoporous materials with the characteristic of cylindrical pores open at both ends. Due to the mesoporous texture, the material exhibits a high specific surface area, which enhances ion release from the surface and therefore the bioactivity of the sample [6]. The pore size distribution is shown in inset of Fig. 2. The sample presents a single-modal pore size distribution centered around 5.28 nm. The ordering of mesoporous channel structures may greatly influence hydroxyapatite forming ability, the protein adsorption 1225 World Journal of Engineering and nutrient-delivery behavior for better in vivo bioactivity [7], together with presenting the option of filling the pores with drugs or biological molecules [8]. nm. The electron diffraction pattern (inset in Fig. 4) indicates clearly visible diffraction rings, whose interplanar spacings are in good agreement with the characteristic spacings of an apatite structure. This supports the result of the XRD result. Fig. 2. N2 adsorption-desorption isotherm and BJH pore size distribution curves for mesoporous SiO2-CaO-P2O5 calcined at 600 °C for 3 h. Fig. 4. TEM images of mesoporous SiO2-CaO-P2O5 after soaking in SBF for 10 h. Conclusion Fig. 3(a) illustrates the wide angle XRD diffraction pattern of the mesoporous SiO2-CaO-P2O5 bioactive glass. A broad band between 20 and 30o can be clearly noted in XRD pattern of mesoporous bioactive glass. It can be seen that after being calcined at 600 °C for 3 h the sample almost has an amorphous state, which is indicative of the internal disorder and glassy nature of this material. Highly ordered mesoporous SiO2-CaO-P2O5 bioactive glass has been obtained by the sol-gel method, using a nonionic triblock copolymer as the organic template. The mesostructured SiO2-CaO-P2O5 calcined at 600 °C has an amorphous form, a specific surface area and pore volume of 317.24 m2/g and 0.474 cm3/g, respectively, and with a narrow pore size distribution. In vitro bioactivity test of the mesoporous SiO2-CaO-P2O5 shows that it could develop an apatite layer on the surface after soaking in SBF for 10 h. This material is an excellent candidate for use in grafts for bone tissue regeneration. References 1. Bagshaw, S. A., Prouzet, E., and Pinnavaia, T. J. Templating of mesoporous molecular sieves by nonionic polyethylene oxide surfactants. Science, 269 (1995) 1242-1244. 2. Xiu, T. P., Liu, Q., and Wang, J. C. Alkali-free borosilicate glasses with wormhole-like mesopores. J. Mater. Chem., 16 (2006) 4022-4024. 3. Yan, X. X., Yu, C. Z., Zhou, X. F., Tang, J. W., and Zhao, D. Y. Highly ordered mesoporous bioactive glasses with superior in vitro bone-forming bioactivities. Angew. Chem. Int. Ed., 43 (2004) 5980-5984. 4. Tanev, P. T., and Pinnavaia, T. J. A neutral templating route to mesoporous molecular sieves. Science, 267 (1995) 865-867. 5. Vallet-Regi, M., Ragel, C. V., and Salinas, A. J. Glasses with medical applications. Eur. J. Inorg. Chem., 6 (2003) 1029-1042. 6. Hench, L. L., Splinter, R. J., Allen, W. C., and Greenlee, T. K. Bonding mechanism at the interface of ceramics prosthetic materials. J. Biomed. Mater. Res. Symp., 2 (1971) 117-141. 7. Fan, J., Yu, C. Z., Gao, T., Lei, J., Tian, B. Z., Wang, L. M., Luo, Q., Tu, B., Zhou W. Z., and Zhao, D. Y. Cubic mesoporous silica with large controllable entrance sizes and advanced adsorption properties. Angew. Chem., Int. Ed., 42 (2003) 3146-3150. 8. Slowing, I. I., Trewyn, B. G., Giri, S., and Lin, V. S. Y. Mesoporous silica nanoparticles for drug delivery and biosensing applications. Adv. Funct. Mater., 17 (2007) 1225-1236. Fig. 3. XRD patterns of mesoporous SiO2-CaO-P2O5 before (a) and after being soaked in SBF for 10 h (b). The bone-forming activity of mesoporous SiO2-CaO-P2O5 in vitro was then tested in SBF to monitor the formation of apatite on the surface of the mesoporous SiO2-CaO-P2O5. XRD pattern of the bioactive glass after soaking in SBF for 10 h is shown in Fig. 3(b), a diffraction peak at 31.7° emerge, corresponding to the (211) reflection of hydroxide apatite. The result indicates that the bioactive glass can induce the formation of an apatite layer on their surface in SBF even for short soaking period. This is consistent with the known superiority of bioactive glass in their in vitro bone-forming bioactivity. The bright field (Fig. 4) TEM image reveals that the surface of the mesoporous SiO2-CaO-P2O5 undergoes important changes during the reaction with SBF. The growth of apatite nanocrystals on the surface of the mesoporous SiO2-CaO-P2O5 can be observed after soaking for 10 h, and the crystallite size is about 6-8 1226