SLO for 32A_29jan11

1
The following is our SLO (Student Learning Outcome) planned for this class. At the end of the semester, we will have covered these principles, and you will have gained some hands-on experience with them.
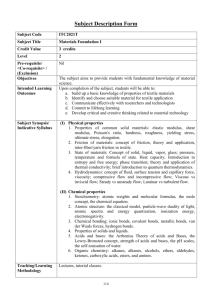
Chem 032A Fundamentals of General, Organic and Biological Chemistry
After completing the course, the student will be able to …
1. Examine matter and the chemical changes it can undergo. Be able to name and recognize symbols of common elements in the periodic table. Differentiate between the three states of matter (e.g. gas, liquid, solid). For each of the three states of matter, compare and contrast intra- and inter-molecular forces, differentiate their general physical and chemical properties and behavior, and comprehend kinetic molecular theory
2. Apply the scientific methods of testing, observing, and drawing conclusions
3. Explain atomic theory, atomic structure, isotopes and atomic weight, and electron structure (shell, subshell, orbital energy, size, shape and orientation in space). Write the electron configuration of an element or ion.
Understand periodic trends (e.g. electro-negativity, electron affinity, atomic radius, metallic character, etc.)
4. Compare and contrast ionic and covalent bonding for inorganic compounds and molecules. Write molecular formula s, formula units, and Lewis structures. Predict and draw a molecule’s shape using Valence Shell
Electron Pair Repulsion Theory and the octet rule
5. Use accurate nomenclature for inorganic and molecular compounds including acids. Students will be able to write formulas of inorganic salts, (polyatomic) ions, simple organic compounds, and acids and bases for a specified chemical. Conversely, students will be able to identify the chemical based on its formulaic description
6. Employ dimensional analysis (or the factor labeling method) as the primary problem-solving method to convert a measurement (e.g. heat, volume, mass, length, time, temperature, density, specific gravity) from one unit to another including uses of metric, exponential (or scientific) notation, significant figures, or rounding off numbers
7. Classify several common classes of chemical reactions, and be able to balance chemical equations. Write formulas for the products and reactants, and predict their phases (i.e. gas, liquid, solid, aqueous)
8. Apply the concept of the mole and Avogadro’s number. Apply procedures of stoichiometry and be able to write gram-mole-particles conversions. Comprehend and describe energy, rates, and equilibrium (Le
Chatelier’s Principle), and effects of temperature, concentration, and catalysts on such chemical reactions
9. Examine and classify different types of solutions and mixtures. Describe the following: solubility and the effect of temperature and pressure, osmotic pressure principles, solution concentration measurements (i.e. molarity), dilution methods, and solution conductivity (i.e. strong electrolytic, weak electrolytic and nonelectrolytic solutions)
10. Define, compare and contrast acids and bases. Identify common acids and bases, and explain the meaning of acidity/basicity in aqueous solution (pH). Describe buffer solutions and acid-base titrations
11. Be familiar with and practice safe laboratory procedures: a. Use of the Bunsen burner, hot plate, analytical balance (to measure mass), and analytical glassware
(e.g. graduated cylinder, buret to measure volume) b. Characterize elements in a gas discharge tube via a spectroscope and flame test c. Determine properties such as density, miscibility, and solubility d. Synthesize and purify products/substances by physical methods such as filtration, solvent extraction, and paper chromatography e. Determine percent composition of element(s) in a mixture f. Manipulate molecular models and computer-generated structures to predict molecular structure and shape g. Mix chemicals and observe, classify, and identify the resultant reaction h. Conduct chemical equilibrium and kinetic studies to predict direction and rate of reaction, respectively i. Evaluate the gas law constant experimentally j.
Measure pH using acid-base indicators, titration, and pH meter of solutions of acids, bases, and salts