CHEM 1152 - First Year Chemistry II (TAG OSC009) (OTM 14990)
advertisement

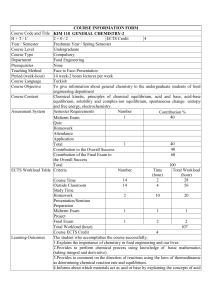
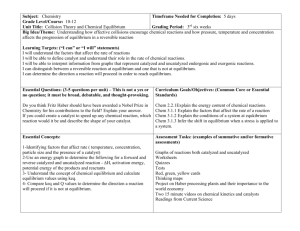
Southern State Community College Curriculum Committee – February 2012 CHEM 1152: First Year Chemistry II Page 1 of 6 I. COURSE TITLE: First Year Chemistry II COURSE NUMBER: 1152 II. TAG: OSC009 OTM: 14990 CATALOGUE PREFIX: CHEM PREREQUISITE: CHEM 1151 COREQUISITE: CHEM 1162 III. CREDIT HOURS: 4 LABORATORY HOURS: 0 IV. COURSE DESCRIPTION LECTURE HOURS: 4 OBSERVATION HOURS: 0 A continuation of the study of college chemistry covering gases, intermolecular forces of attraction and phase changes, solutions and colligative properties, chemical kinetics, chemical equilibrium, acid-base equilibria, thermodynamics, electrochemistry, and descriptive chemistry. V. ADOPTED TEXTS: Chemistry & Chemical Reactivity, 8th Edition By: Kotz, Treichel, and Townsend Cengage Learning, 2012 ISBN: 1-111-57498-7 VI. COURSE OBJECTIVES: Upon completion of this course the student should be able to: 1. 2. 3. 4. 5. 6. 7. 8. solve problems pertaining to the gas laws, use the gas laws to solve problems pertaining to reaction stoichiometry, list the principle tenets of kinetic molecular theory and use the theory to explain the gas laws, list and discuss the intermolecular forces of attraction and explain their role in determining melting points, boiling points and vapor pressures, discuss the properties of solids including the bonding in ionic, network, molecular, and metallic solids, discuss and solve problems pertaining to solution concentration, Henry'sLaw and colligative properties, discuss the solution process using appropriate terminology and chemical equations, perform calculations involving reaction rates and determine reaction orders, Southern State Community College Curriculum Committee – February 2012 CHEM 1152: First Year Chemistry II Page 2 of 6 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. VII. TAG: OSC009 OTM: 14990 define and calculate activation energy, determine rate equations from reaction mechanisms, calculate equilibrium constants from the free energy changes of reactions, discuss the nature of the equilibrium state and predict the affects of external factors on equilibrium systems, write and use equilibrium constant and equilibrium quotient expressions, explain and write chemical equations pertaining to the Arrhenius definition of acids and bases, discuss and write equations pertaining to the autoionization of water, write the Bronsted definition of acids and bases, given a Bronsted acid-base pair, write a chemical equation for the corresponding proton-transfer reaction, identify the conjugate acid-base pairs in the reaction, and predict the direction of the reaction, define and distinguish between weak and strong acids and bases and provide examples of each, solve problems pertaining to the water ionization constant, the pH scale and acid and base equilibrium constants, discuss the common ion effect, describe buffer solutions and use appropriate chemical equations and equilibrium constant expressions to show how they resist changes in pH, discuss and apply thermodynamic principles to calculations involving free energy, entropy, and equilibrium, solve problems pertaining to the Nernst equation, explain the role of electrochemistry in the operation of batteries and corrosion, and demonstrate a knowledge of the descriptive chemistry of selected elements. COURSE METHODOLOGY: This course will use lecture, Power Point, discussion, home work assignments, computer assignments, and written exams and quizzes as deemed appropriate by instructor. VIII. GRADING: The following grading scale will be used: A = 90 - 100 B = 80 – 89 C = 70 - 79 D = 60 - 69 F = < 60 Southern State Community College Curriculum Committee – February 2012 CHEM 1152: First Year Chemistry II Page 3 of 6 IX. TAG: OSC009 OTM: 14990 COURSE OUTLINE: 1. Gases a. b. c. d. Properties of gases Kinetic molecular theory of gases The gas laws Gas laws and chemical stoichiometry 2. Intermolecular Forces, Liquids, and Solids a. Physical states of matter and kinetic molecular theory b. Intermolecular forces of attraction c. Properties of liquids d. Solids 3. Solutions and their behavior a. Definitions b. Units of concentration c. The solution process d. Factors affecting solubility e. Colligative properties 4. Chemical kinetics a. Rates of chemical reactions b. . Effect of concentration on reaction rate c. Integrated rate laws d. Collision theory and activation energy e. Reaction mechanisms 5. Thermodynamics a. Entropy and the second and third laws of thermodynamics b. Entropy changes c. Gibbs free energy d. ∆G°and the equilibrium consta 6. Chemical equilibria a. The equilibrium state b. The equilibrium constant c. The reaction quotient d. Equilibrium calculations e. External factors affecting equilibria 7. Acids and Bases Southern State Community College Curriculum Committee – February 2012 CHEM 1152: First Year Chemistry II Page 4 of 6 a. b. c. d. e. f. g. h. i. j. TAG: OSC009 OTM: 14990 The Arrhenius definition Auto-ionization of water The Bronsted definition The pH scale Strong acids and bases Weak acids and bases Acid-base reactions The common ion effect and buffer solutions Acid-base titration curves Acid-base indicators 8. Electrochemistry a. Chemical change leading to electric current b. Electrochemical cells and potentials c. Voltaic cells at nonstandard conditions d. Common batteries and storage cells e. Chemical change from electrical energy 9. Descriptive chemistry of selected elements COURSE CALENDAR Week # 1 Chapter 11 2 11 3 12 ,13 4 14 5 15 Topics/Assignments 11.1 Gas Pessure 11.2 Gas Laws 11.3 Ideal Gas Law 11.4 Gas Laws and Chemical Reactions 11.5 Gas Mixtures and Partial Pressures 11.6 Kinetic Molecular Theory of Gases Applications – Deep Sea Diving, Isotope Separation 12.1 States of Matter 12.2-12.3 Intermolecular Forces and Molecules 12.4 Properties of Liquids 13.4 Properties of Solids and phase changes 14.1 Units of Concentration 14.2-14.3 Solution process and factors affecting solubility 14.4–14.5 Colligative Properties and Colloids 15.1 Rates of Chemical Reactions 15.2 Reaction Conditions and Rate Southern State Community College Curriculum Committee – February 2012 CHEM 1152: First Year Chemistry II Page 5 of 6 6 15 7 16 8 17 9 17 10 18 11 18 12 19 13 20 14 20 15 21,22 16 X. 15.3 Effect of Concentration on Reaction Rate 15.4 Integrated Rate Laws 15.5 Arrhenius Equation 15.6 Reaction Mechanisms 16.1-16.2 Chemical Equilibrium Constants and Reaction Quotient 16.3 -16.5 Equilibrium calculations 16.6 Disturbing a Chemical Equilibrium 17.1-17.2 Acids, Bases, and the Bronsted-Lowery Concept 17.3 Water and pH 17.4/17.7 Equilibrium acid/base calculations 17.5 Prediction of the direction of acid/base reactions 17.6 Types of Acid-Base Reactions 18.1-18.2 Common Ion Effect and Buffers 18.3 Titrations 18.4-18.5 Ksp 18.6-18.7 Complex Ions 19.1 Energy Transfer 19.2-19.5 Entropy 19.6-19.7 Gibbs Free Energy 20.1 Oxidation-Reduction 20.2-20.3 Voltaic Cells 20.4-20.5 Electrochemical Cells and the Nernst Equation 20.6 Electrochemistry and Thermodynamics 20.7 Electrolysis Chemistry of Main Group and Transition Elements Final OTHER REQUIRED BOOKS, SOFTWARE, AND MATERIALS: A calculator is required. XI. EVALUATION: Discretion of instructor XII. TAG: OSC009 OTM: 14990 SPECIFIC MANAGEMENT REQUIREMENTS: Southern State Community College Curriculum Committee – February 2012 CHEM 1152: First Year Chemistry II Page 6 of 6 TAG: OSC009 OTM: 14990 You may not use programmable calculators or cell phone calculators for tests. Please understand that your work may be seen by others. Others may see your work when being distributed, during group project work, or if it is chosen for demonstration purposes. There is also a possibility that your papers may be submitted electronically to other entities, for reasons such as for plagiarism checks. ACADEMIC MISCONDUCT: Any student who commits any type of academic misconduct as stated in the current college catalogue will receive an "F" for the exam, quiz, or evaluated project. XIII. OTHER INFORMATION FERPA: Students need to understand that your work may be seen by others. Others may see your work when being distributed, during group project work, or if it is chosen for demonstration purposes. Students also need to know that there is a strong possibility that your work may be submitted to other entities for the purpose of plagiarism checks. DISABILITIES: Students with disabilities may contact the Disabilities Service Office, Central Campus, at 800-628-7722 or 937-393-3431.