Chemistry 1011 Curriculum Using
advertisement

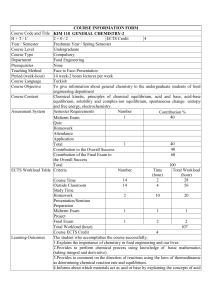
Chemistry 1011 Slot 7 - Instructor: Dr. P. G. Mezey Office: C4015 Phone: 864-8768 Detailed curriculum available from the website Chemistry 1011 Curriculum Using the Textbook: “Principles of Chemistry, A Molecular Approach” by Nivaldo J. TRO ( Prentice Hall, New Jersey 2010 ) Evaluation: Tutorials - 15% Class Tests (2) - 20% Laboratory - 10% Final Examination - 55% Chemistry 1011 Curriculum Using "Principles of Chemistry" by Tro Curriculum in PDF format Chemistry 1010 (a prerequisite for this course) covers chapters 1-9 in Tro. You should refresh yourself on those chapters, particularly if you took Chem 1010 a while ago. Take a look at the Review page for details. 18.2 Balancing Oxidation-Reduction Equations (Note: This section is usually covered with a very brief review of oxidation numbers and oxidizing/reducing agents, as covered in Chemistry 1010, which is found in section 4.9 of the Tro text). Chapter 10: Chemical Bonding II: Molecular Shapes, Valence Bond Theory 10.1 Artificial Sweeteners: Fooled by Molecular Shape (Suggested Reading) 10.2 VSEPR Theory: The Five Basic Shapes 10.3 VSEPR Theory: The Effect of Lone Pairs 10.4 VSEPR Theory: Predicting Molecular Geometries 10.5 Molecular Shape and Polarity 10.6 Valence Bond Theory: Orbital Overlap as a Chemical Bond 10.7 Valence Bond Theory: Hybridization of Atomic Orbitals (sp, sp2, sp3 hybridization only) Chapter 11: Liquids, Solids, and Intermolecular Forces 11.1 Climbing Geckos and Intermolecular Forces (Suggested Reading) 11.2 Solids, Liquids and Gases: A Molecular Comparison 11.3 Intermolecular Forces: The Forces that Hold Condensed Phases Together 11.5 Vaporization and Vapor Pressure (Excluding the Clausius-Clapeyron Equation) 11.6 Sublimation and Fusion 11.7 Heating Curve for Water 11.11 Crystalline Solids: The Fundamental Types Chapter 12: Solutions 12.1 Thirsty Solutions: Why You Should Not Drink Seawater 12.2 Types of Solutions and Solubility 12.5 Expressing Solution Concentration (Selected Topics) 12.6 Colligative Properties: Vapor Presssure Lowering, Freezing Point Depression, Boiling Point Elevation and Osmotic Pressure (Selected Topics) Chapter 13: Chemical Kinetics 13.1 Catching Lizards (Suggested Reading) 13.2 The Rate of a Chemical Reaction 13.3 The Rate Law: The Effect of Concentration on Reaction Rate 13.5 The Effect of Temperature on Reaction Rate 13.6 Reaction Mechanisms (Excluding Mechanisms with a Fast Initial Step) 13.7 Catalysis Chapter 14: Chemical Equilibrium 14.1 Fetal Hemoglobin and Equilibrium (Suggested Reading) 14.2 The Concept of Dynamic Equilibrium 14.3 The Equilibrium Constant (K) 14.4 Expressing the Equilibrium Constant in Terms of Pressure 14.5 Heterogeneous Equilibria: Reactions Involving Solids and Liquids 14.6 Calculating the Equilibrium Constant from Measured Equilibrium Concentrations 14.7 The Reaction Quotient: Predicting the Direction of Change 14.8 Finding Equilibrium Concentrations 14.9 Le Chatelier’s Principle: How a System at Equilibrium Responds to Disturbances Chapter 15: Acids and Bases 15.1 Heartburn (Suggested Reading) 15.2 The Nature of Acids and Bases 15.3 Definitions of Acids and Bases 15.4 Acid Strength an the Acid Ionization Constant (Ka) 15.5 Autoionization of Water and pH 15.6 Finding the [H3O+] and pH of Strong and Weak Acid Solutions (Excluding Polyprotic Acids) 15.7 Base Solutions 15.8 The Acid-Base Properties of Ions and Salts (Excluding Metal Ion Acidity) 15.10 Lewis Acids and Bases Chapter 16: Aqueous Ionic Equilibrium 16.1 The Danger of Antifreeze (Suggested Reading) 16.2 Buffers: Solutions that Resist pH Change 16.5 Solubility Equilibria and the Solubility Product Constant 16.6 Precipitation Chapter 18: Electrochemistry 18.1 Pulling the Plug on the Power Grid (Suggested Reading) 18.3 Voltaic (or Galvanic) Cells: Generating Electricity from Spontaneous Chemical Reactions 18.4 Standard Electrode Potentials 18.7 Batteries: Using Chemistry to Generate Electricity 18.8 Electrolysis: Driving Nonspontaneous Chemical Reactions with Electricity 18.9 Corrosion: Undesirable Redox Reactions Tutor Audiovisual csupport@mun.ca regi munlevelcim sun(2)f(22)sun(2) pmezey p..munclass