ANEMIAS
advertisement

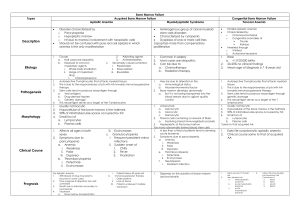
ANEMIAS Dr. Nusrum Iqbal Anemia due to marrow failure (Aplastic anemia) Aplastic anemia is defined as pancytopenia with hypocellularity (aplasia) of the bone marrow. It is an uncommon but serious condition Aplastic anemia is due to a reduction in the number of pluripotential stem cell together with a fault in those remaining or an immune reaction against them. Failure of one cell line may occur Evolution to myelodysplasia, paroxysmal nocturnal haemoglobinuria (PNH) or acute myeloblastic leukaemia occurs in some cases Causes Immune mechanisms are probably responsible for most cases of idiopathic acquired aplastic anemia Activated cytotoxic T cells in blood and bone marrow are responsible for the bone marrow failure Drugs may cause marrow aplasia, including cytotoxic drugs such as busulphan and doxorubicin Causes Some individuals develop aplasia due to sensitivity to non-cytotoxic drugs such as chloramphenicol, gold, carbimazole, chlorpromazine, phenytoin, tolbutamide, nonsteroidal anti-inflammatory Congenital aplastic anaemia are rare. Fanconi’s anemia is inherited as autosomal recessive and is associated with skeletal, renal and central nervous system abnormalities Causes of Aplastic Anaemia Primary Congenital e.g. Fanconi’s anemia Idiopathic acquired (50% of cases) Secondary Chemicals, e.g. benzene Drugs Chemotherapeutic idiosyncratic Insecticides Ionizing radiation Infections Viral, e.g. hepatitis, measles, HIV, Parvo Other, e.g. tuberculosis Pregnancy Paroxysmal nocturnal hemoglobinuria Clinical Features Anemia Bleeding Infection Ecchymoses Bleeding gums and epistaxis Mouth infections Lymphadenopathy Hepatosplenomegaly are rare Investigations The laboratory diagnosis is made on the basis of: Pancytopenia The virtual absence of reticulocytes A hypocellular or aplastic bone marrow with increased fat spaces Differential Diagnosis Causes of pancytopenia Aplastic anaemia Drugs Megaloblastic anaemia Bone marrrow inflitration or replacement Hodgkin’s and non-Hodgkin’s lymphoma Acute leukoaemia Myeloma Secondary carcinoma Myelofibrosis Hypersplenism Systemic lupus erythematosus Disseminated tuberculosis Paroxysmal nocturnal hemoglobinuria Overwhelming sepsis Treatment and Prognosis The main danger is infection and stringent measures should be undertaken to avoid this in a severely neutropenic patient immediate institution of broad-spectrum parenteral antibiotics Supportive care including transfusions of red cells platelets Leucocyte-depleted red cell platelets should be given to prevent HLA alloimmunization to minimize the risk of rejection of a bone marrow transplant to prevent febrile transfusion reactions Cause of the aplastic anemia must be eliminated if possible Course of aplastic anemia can be variable Treatment and Prognosis A bad prognosis (i.e. severe aplastic anaemia) is associated with the presence of two of the following three features: Neutrophil count <0.5x109/L Platelet count of <20x109/L Reticulocyte count of <40x109/L In severe aplastic anaemia, there is a very poor outcome without treatment. Bone marrow transplantation is the treatment of choice for patietns under 20 years of age Treatment and Prognosis Immunosuppression produces similar long-term survival. Transplantation versus immuno-suppression is a contentious issue in the 20-45 age range Patients over the age of 45 are not eligible for bone marrow transplantation because of the high risk of graft-versus-host disease With matched transplants, the 3 year survival rate is up to 90% Treatment and Prognosis Immunosuppressive therapy is used for those pateints over the age of 45 years; Antilymphocyte globulin (ALG) and cyclosporin are used alone or in combination. ALG alone produces a hematological recovery in 50-60%, 80% in patietns also receiving cyclosporin Levels of haemopoietic growth factors are normal or increased in most patients with aplastic anemia, and are ineffective as primary treatment Treatment and Prognosis Androgens (e.g. oxymethalone) are sometimes useful in patients not responding to immunosuppression in patients with moderately severe aplastic anaemia Steroids have little activity in severe aplastic anaemia are used for serum sickness due to ALG. Also used to treat children with congenital pure red cell aplasia (diamondbalckfan syndrome) Adult pure red cell aplasia is associated with a thymoma in 30% of cases and thymectomy may induce a remission HEMOLYTIC ANEMIAS Haemolytic anemias are caused by increased destruction of red cells Breakdown of normal red cells occurs in the macrophages of the bone marrow, liver and spleen Consequences of Haemolysis The bone marrow can increase its output by six to eight times by increasing the proportion of cells committed to erythropoiesis (erythroid hyperplasia) by expanding the volume of active marrow Immature red cells (reticulocytes) are released prematurely. These cells are larger than mature cells stain light blue on a peripheral blood film Sites ofHaemolysis Extravascular haemolysis The red cells are removed from the circulation by macrophages in the reticuloendothelial system, particularly the spleen Sites of Haemolysis Intravascular Haemolysis When red cells are rapidly destroyed within the circulation, haemoglobin is liberated Excess free plasma Hb is filtered by the renal glomerulus and enters the urine In the renal tubular cell, Hb is broken down and becomes deposited in the cells as haemosiderin Some of the free plasma Hb is oxidized to methaemoglobin, which dissociates into ferrihaem and globin. Plasma haemopexin binds ferrihaem The liver plays an important role in removing Hb bound to hepatoglobin and haemopexin and any remaining free Hb Evidence for Haemolysis Increased red cell breakdown leads to: Elevated serum bilirubin (unconjugated) Excess urinary urobilinogen (resulting from bilirubin breakdown in the intestine Reduced plasma haptoglobin Raised serum lactic dehydrogenase (LDH) Evidence for Haemolysis Increased red cell production leads to: Reticulocytosis Erythroid hyperplasia of the bone marrow Evidence of abnormal red cell in some haemolytic anaemias: Spherocytes Sickle cells Red cell fragments Demonstration of shortened red cell life-span Red cell survival studes using 51Cr-labbled red cells are useful in complicated cases, and for quantitation of the severity of haemolysis Causes of Hemolytic Anaemia Inherited Red cell membrane defect Hereditary spherocytosis Hereditary elliptocytosis Haemoglobin abnormalities Thalasaemia Sickle cell disease Metabolic defects Glucose-6-phosphate Dehydrogenase deficiency Pyruvate kinase deficiency Causes of Hemolytic Anemia Acquired Immune Autoimmune Warm cold Alloimune Hemolytic transfusion Reactions Hemolytic disease of the new born After allogeneic bone marrow or organ transplantation Drug-induced Causes of Haemolytic Anaemia Acquired Non-immune Acquired membrane defects Paroxysmal nocturnal haemoglobinuria Mechanical Microangiopathic haemolytic anaemia Valve prosthesis March haemoglobinuria Secondary to systemic disease: Renal and liver failure Causes of Hemolytic Anaemia Miscellaneous Infections, e.g. malaria, Clostridium welchii Drugs and chemicals causing damage to the red cell membrane or oxidative hemolysis Hypersplenism Burns Red cell membrane defects The normal red cell membrane consists of a lipid bilayer crossed by integral proteins with an underlying lattice of proteins (or cytoskeleton), spectrin, actin, ankyrin and protein 4.1, attached to integral proteins Hereditary Spherocytosis (HS) Surface-to-volume ratio decrease, the cells become spherocytic. Spherocytes are more rigid and less deformable than normal red cells. They are unable to pass through the splenic microcirculation and they die Deficiency in the structural protein spectrin quantitative defects in other membrane proteins have been identified Abnormal red cell membrane associated with increased permeability to sodium Hereditary Spherocytosis (HS) HS is the most common inherited hemolytic anemia in northern Europeans, affecting 1 in 5000 25% of patients autosomal dominant HS is due to a defect in the red cell membrane, resulting in the cells losing part of the cell membrane as they pass through the spleen Clinical Features Jaundice at birth Some patients may go through life with no symptoms and are detected only during family studies Anemia, splenomegaly ulcers on the leg course of the disease may be interrupted by aplastic, hemolytic and megaloblastic Aplastic anemia usually occurs after infections, parvovirus megaloblastic anemia is the result of folate depletion Chronic hemolysis leads to the formation of pigment gallstones Investigations Anaemia: mild, but occasionally can be severe Blood film: this shows spherocytes and reticulocytes Hemolysis: is evident (e.g. the serum bilirubin and urinary urobilinogen will be raised Osmotic fragility: when red cells are placed in solutions of increasing hypotonicity, they take in water, swell, and eventually useful to confirm a suspicion of spherocytosis on a blood film Direct antiglobulin (Coombs’) test: negative in spherocytosis Treatment The spleen, which is the site of cell destruction, should be removed in all but the mildest cases Raised bilirubin and especially the presence of gallstones should encourage splenectomy Best to postpone splenectomy until after child-hood Splenectomy should be preceeded by pneumococcal and Hib immunization and followed by lifelong penicillin prophylaxis Folate levels should be monitored, or folic acid can be given prophylactically Hereditary Elliptocytosis This disorder of the red cell membrane is inherited in an autosomal dominant manner and has a prevalence of 1 in 2500 in Caucasians Red cells are elipiptical owing to spectrin and other protein abnormaliteis Hereditary Stomatocytosis Stomatocytes are red cells in which the pale central area appears slit-like. There presence in large numbers may occur in a hereditary haemolytic anaemia associated with a membrane defect, but excess alcohol intake is also a common cause