Supplementary Materials and Methods (doc 48K)
advertisement

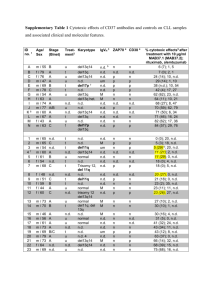
Supplementary materials and methods Specimens After obtaining informed written consent (ethics committee approval 96/08, http://www.uni-ulm.de/ethikkommission/), peripheral blood cells were collected from CLL patients and healthy donors. Patients were selected to have either a normal karyotype or 13q deletion as sole abnormality, and patients with a karyotype associated with more aggressive disease were excluded. No patient in the present study had received any prior therapy except for one patient who had received treatment 6 years before sampling. Median tumor load as measured by CD19+/CD5+ flow cytometry was 91.2% (range 83.0% to 91.8%). In the case of samples from healthy donors, buffy coats were collected from age-matched donors after oral informed consent. Cell purification and in-vitro culture PBMCs were isolated from whole blood by density-gradient centrifugation over FicollHypaque according to the manufacturer’s protocol (Biochrom AG, Berlin Germany). Cells were used either fresh or viably frozen in Dulbecco's Modified Eagle Medium (DMEM; Invitrogen, Karlsruhe Germany) containing 40% fetal calf serum (FCS) and 10% dimethyl sulfoxide (DMSO) for storage in liquid nitrogen. At the time of initiation of culture, the viability of cells was always above 90% as determined by 7-Aminoactinomycin D staining (7-AAD; Sigma, Steinheim Germany) using flow cytometry (FACSCalibur, BD Biosciences, San Jose USA). All cells were cultured at 37 °C with 10% CO2 in Dulbecco’s modified eagle medium (DMEM; Invitrogen, Carlsbad, CA, USA) supplemented with 10 % fetal calf serum and penicillin/streptomycin. CD19+ cells from CLL patients and healthy donors were isolated by magnetic beadactivated cell sorting (MACS) using CD19-microBeads following the instructions of the manufacturer (Miltenyi Biotec, Bergisch Gladbach, Germany). The purity of the CD19+ fraction was 95% ± 3% after isolation from PBMCs of healthy donors and 97% ± 2% for isolation from PBMCs of CLL patients as measured by flow cytometry. Similarly, CD14 + cells were isolated by MACS from PBMCs of healthy donors using CD14-microbeads 1 following the instructions of the manufacturer (Miltenyi Biotec, Bergisch Gladbach, Germany). The purity of the CD14+ fraction was 94% ± 3% as assessed by flow cytometry. CD14+ cells were cocultured with purified CLL B-cells or non-malignant B-cells from healthy donors in 2ml culture medium in 6 well plates at cell densities of 2.5x106/mL (CD14+ cells) and 5x106/mL (CLL/B cells). After 18 days of culture, plates were thoroughly rinsed thrice with 0.5% Trypsin-EDTA (GIBCO, Invitrogen,Germany) to remove non-adhering cells. The washing procedure was controlled using CD19+ staining of lymphocytes using flow cytometry (residual B/CLL cells sticking to the cell culture dish after rinsing were <2% of all harvested adherent cells) and phase contrast microscopy. Adherent cells were then removed by careful scraping for further analyses. Immunohistochemistry Paraffin-embedded sections from lymph nodes of CLL patients (n=4) and from a healthy donor were dewaxed with xylol and passed through decreasing concentrations of alcohol (100%, 70%, 40%). For antigen retrieval, sections were incubated in a steamer using TRS buffer pH 6.1, and then incubated with 1:100 dilution of anti-CD68 primary antibody (Dako Deutschland, Hamburg, Germany). Cryo-sections from lymph nodes of CLL patients (n=4) and from a tonsil of a healthy donor were dehydrated, fixed for 10min with acetone and incubated for 1h at RT with 1:800 dilution of anti-CD14 primary antibody (clone HCD14, Biolegend, San Diego, USA). Staining was performed using Dako REAL Detection System, Alkaline Phosphatase/RED Kit (Dako) according to the manufacturer’s instructions. Samples were analyzed using a Zeiss Axiophot microscope (Carl Zeiss, Oberkochen, Germany). Images were processed using the Diskus software (Hidgers, Koenigswinter, Germany). Flow cytometry Monoclonal antibodies specific for CD68, CD1a and relevant isotype control monoclonal antibodies were purchased from BD Pharmingen, Heidelberg, Germany. Monoclonal antibodies specific for CD14 and the respective isotype control antibody was purchased 2 from Beckman Coulter Inc., Fullerton, CA, USA. Monoclonal antibody for CD19 and its isotype control antibody was purchased from Dako Deutschland GmbH, Hamburg, Germany. Monoclonal CD15 antibody was purchased from Miltenyi Biotec, Bergisch Gladbach, Germany. To detect surface antigens, cells were stained with saturating amounts of antibodies for 20 minutes at 4°C in PBS supplemented with 1% bovine serum albumin, washed 2 times, and then analyzed on a FACSCalibur (BD Biosciences, San Jose, USA). For staining of cytoplasmic epitopes, cells were fixed and permeabilized using FIX&PERM (AN DER GRUB Bio Research GMBH, Austria). Subsequently, cells were incubated with specific or isotype control antibodies, washed and signal detected by flow cytometry. Data were analyzed using FlowJo software (Tree Star, San Carlos, CA). Real time quantitative RT-PCR Total RNA was isolated from NLCs (n=4), CD14CLL-cells (n=6) and CD14B-cells (n=5) using the RNeasy Mini Kit (QIAGEN, Valencia, CA). First-strand cDNA synthesis was performed using AffinityScript QPCR cDNA Synthesis Kit (Agilent Technologies, Böblingen, Germany). Real-time PCR was performed using Absolute QPCR SYBR Green ROX Mix from Thermo Scientific, Surrey (UK) with a 7300 Real-Time PCR System (Applied Biosystems, Foster City (USA). Forward and reverse primers were used at final concentrations of 70nM in 20µl reactions. Primers used for amplification were as follows: HMBS, 5´-ACCAAGGA-GCTTGAACATGC-3´ and 5´- GAAAGACAACAGCATCATGAG-3´; TBP, 5´-GCCCGAAACGCCGAATAT-3´ and 5´-CCGTGGTTCGTG-GCTCTCT-3´. APRIL, 5´-AAGGGTATCCCTGGCAGAGT-3´ and 5´-CTGTTTTTGGGTGA-GCACTG-3´; BAFF, 5´-ACCGCGGGACTGAAAATCT3´ and 5´-CACGCTTATT-TCTGCTGTTCTGA-3´; PECAM1, 5´- TTCAGAAGGACAAGGCGATT-3´and 5´GTAGTTGCCACTGT-GCTCCA-3´ Gene expression profiling RNA Isolation and Analysis Total RNA from NLCs (n=4), CD14CLL-cells (n=4), CD14B-cells (n=4) were isolated using RNeasy mini kit from QIAGEN (Hilden, Germany) according to the manufacturer's 3 instructions. The quality of total RNA was checked on an Agilent 2100 Bioanalyzer (Agilent Technologies GmbH, Berlin, Germany). Probe Labeling and Illumina Sentrix BeadChip array Hybridization Biotin-labeled cRNA samples for hybridization on Illumina Human Sentrix-12 BeadChip arrays (Illumina, Inc.) were prepared according to Illumina's recommended sample labeling procedure based on the modified Eberwine protocol (1) . Briefly, 250 ng total RNA was used for complementary DNA (cDNA) synthesis, followed by an amplification/labeling step (in vitro transcription) to synthesize biotin-labeled cRNA according to the MessageAmp II aRNA Amplification kit (Ambion, Inc., Austin, TX). Biotin-16-UTP was purchased from Roche Applied Science, Penzberg, Germany. The cRNA was column purified according to TotalPrep RNA Amplification Kit, and eluted in 60 µl of water. Quality of cRNA was controlled using the RNA Nano Chip Assay on an Agilent 2100 Bioanalyzer and spectrophotometrically quantified (NanoDrop). Hybridization was performed at 58°C, in GEX-HCB buffer (Illumina Inc.) at a concentration of 100 ng cRNA/µl, unsealed in a wet chamber for 20h. Spike-in controls for low, medium and highly abundant RNAs were added, as well as mismatch control and biotinylation control oligonucleotides. Microarrays were washed twice in E1BC buffer (Illumina Inc.) at room temperature for 5 minutes. After blocking for 5 min in 4 ml of 1% (wt/vol) Blocker Casein in phosphate buffered saline Hammarsten grade (Pierce Biotechnology, Inc., Rockford, IL), array signals are developed by a 10-min incubation in 2 ml of 1 µg/ml Cy3-streptavidin (Amersham Biosciences, Buckinghamshire, UK) solution and 1% blocking solution. After a final wash in E1BC, the arrays are dried and scanned. Scanning and data analysis Microarray scanning was done using a Beadstation array scanner, setting adjusted to a scaling factor of 1 and PMT settings at 430. Data extraction was done for all beads individually, and outliers were removed when > 2.5 MAD (median absolute deviation). All remaining data points were used for the calculation of the mean average signal for a given probe, and standard deviation for each probe was calculated. Data analysis was 4 done by normalization of the signals using the quantile normalization algorithm without background subtraction. Gene expression from quantile normalized data was visualized by heat maps using the tree-view tool (http://rana.lbl.gov/EisenSoftware.htm) (2). Gene expression pofiling data is available at GEO (Acc No GSE21838) Class comparison analysis Genes differentially expressed between groups of arrays representing CD14B-cells and CD14CLL-cells/NLCs were identified by class comparison analyses using Biometric Research Branch (BRB) ArrayTools software; (BRB, National Cancer Institute, Bethesda, MD, USA). 5 References 1. Eberwine J, Yeh H, Miyashiro K, Cao Y, Nair S, Finnell R, et al. Analysis of gene expression in single live neurons. Proc Natl Acad Sci USA 1992; 89: 3010-3014. 2. Eisen MB, Spellman PT, Brown PO, Botstein D. Cluster analysis and display of genome-wide expression patterns. Proc Natl Acad Sci USA 1998; 95: 1486314868. 6