Supplementary Table 1 (doc 102K)
advertisement

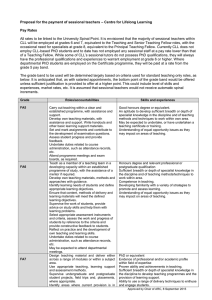
Supplementary Table 1 Cytotoxic effects of CD37 antibodies and controls on CLL samples and associated clinical and molecular features. Age/ Sex Stage (Binet) Treat- Karyotype ment2 IgVH 3 ZAP70 4 CD38 4 A B C D E F G H I J K8 L M N m / 55 f / 79 f / 76 m / 47 m / 69 m / 78 m / 54 f / 63 m / 74 m / 77 m / 60 m / 67 f / 43 m / 63 B A A A B C A A A A/B A A A C u t u u t t u del13q14 del13q del13q14 n.d. del17p 7 n.d. del13q del13q het. n.d. n.d. del13q14 del13q n.d. del17p del13q n.d. 6 n.d. n.d. um n.d. n.d. M M n.d. n.d. n.d. n.d. n.d. n.d. n n.d. p p p p n n n.d. p n.d. n.d. n p n n.d. n p p p n n n.d. p n.d. n n p % cytotoxic effects5 after treatment with 10 µg/ml MAB37.1 (MAB37.2); rituximab, alemtuzumab 6 (7); 1, 5 7 (0); 2, 1 24 (14); 10, n.d. 29 (14); 1, 10 38 (n.d.); 10, 54 42 (4); 17, 27 62 (62); 23, n.d. 64 (11); 10, 21 68 (27); 8, 47 70 (59); 62, 79 71 (50); 6, 34 71 (46); 16, 24 82 (62); 17, 36 84 (57); 29, 76 1 2 3 47 5 6 7 m / 65 m / 65 m / 54 m / 60 f / 61 f / 54 m / 68 n.d. C n.d. A B n.d. C t t t n.d. u t t n.d. M um n.d. n.d. n.d. n.d. n p n n.d. n n.d. p n p p n.d. n n.d. p 0 (0); 20, n.d. 5 (3); 18, n.d. 5 (29) 9; 20, n.d. 11 (21); 2, n.d. 11 (29); 0, n.d. 18 (0); 4, n.d. 18 (0); 5, n.d. 8 9 10 11 12 f / 48 m / 51 f / 59 f / 44 f / 60 n.d. C B A C n.d. t t u n.d. n.d. n.d. n.d. M n.d. n.d. p n n p n.d. n n n p 20 (27); 0, n.d. 21 (18); 0, n.d. 23 (2); 35, n.d. 23 (11); 11, n.d. 23 (26); 27, n.d. 13 14 m / 73 A m / 70 B u t M M n n n n 27 (10); 2, n.d. 30 (10); 1, n.d. 15 16 17 18 19 20 21 22 23 m / 46 m / 56 m / 61 m / 73 m / 69 m / 79 m / 73 f / 64 m / 69 n.d. u u n.d. t u u n.d. u n.d. n.d. del11q del13q14 normal n.d. Trisomy 12, del 11q n.d. del11q n.d. normal trisomy 12 del13q14 normal del11q; del 13q n.d. normal normal n.d. n.d. n.d. 4 del13q14 del13q14 n.d. M n.d. n.d. n.d. um n.d. M n.d. n.d. n n n n p n n n n n n.d. n.d. n p n p n n 30 (16); 4, n.d. 31 (5); 0, n.d. 41 (32); 24, n.d. 43 (34); 11, n.d. 43 (12); 8, n.d. 63 (37); 0, n.d. 66 (14); 32, n.d. 68 (39); 15, n.d. 75 (68); 16, n.d. ID no. 1 A A A A B/C A A n.d. n.d. n.d. u n.d. t u t 1 Individual CLL samples were arranged according to ascending mAb 37.1-induced DCD (samples A to N) or B cell depletion (1 to 23). 2 u (untreated) / t (treated): never treated / with prior treatment (more than three months ago); n.d.: not determined. 3 um (unmutated) / M (mutated): <2 % / >2 % sequence divergence with the closest germline gene. 4 p (positive) / n (negative): percentage of cells with higher fluorescence after staining with specific antibody than with isotype antibody >20 % / < 20 %. 5 For the freshly isolated CLL samples A to N the percentage of non-viable, i.e. annexin Vpositive cells, relative to untreated controls was determined after 24 h of treatment with the indicated antibodies. In the same manner, antibody-induced B cell depletion after an equal treatment period is indicated for the whole blood samples no. 1 to 23. In both groups, samples are arranged according to ascending mAb 37.1 effects. 6 n.d.: not determined. 7 Deletions of chromosomes 17p and 11q, on which the p53 and ATM genes are located, are printed in bold. 8 Samples K and 4 were isolated CLL lymphocytes and whole blood, respectively, from the same patient. 9 Results printed in italics and highlighted by yellow shading untypically show higher mAb 37.1 than mAb 37.2 effects.