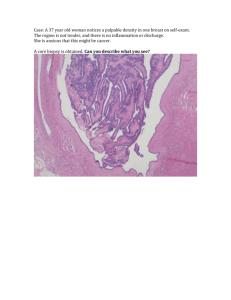
cancersubP
advertisement

NCBI PubMed A service of the U.S. National Library of Medicine and the National Institutes of Health My NCBI [Sign In] [Register] All Databases PubMed Nucleotide Protein Genome Structure OMIM PMC Journals Books PubMed Search Go Clear Limits Preview/Index History Clipboard Details Display 20 for AbstractPlus Show Sort By All: 1 Review: 0 Click to change filter selection through MyNCBI. 1: Ann Surg. 2007 Mar;245(3):408-14. Links Substance P regulates migration in rat intestinal epithelial cells. Turner DJ, Martin PC, Rao JN, Greenspon J, Zou T, Bass BL, Wang JY, Strauch ED. Cell Biology Group, Department of Surgery, University of Maryland School of Medicine, Baltimore, MD, USA. dturner@smail.umaryland.edu OBJECTIVE: The current study examined the effect of substance P (SP) upon intestinal epithelial cells and the mechanistic details of this interaction. SUMMARY BACKGROUND DATA: Intestinal epithelial cells must be capable of migration to reseal mucosal wounds for several vital intestinal functions. This process is incompletely understood; however, recent evidence implicates the neurotransmitter SP in this process. METHODS: Normal rat intestinal epithelial cells (IEC-6 cells) were studied to identify the presence of the SP receptor (NK-1 subtype) and then exposed to physiologic doses of SP and antagonists to assess for increased migration. RESULTS: Examination IEC-6 cells revealed the presence of the SP receptor. Wounding of these cells followed by subsequent exposure to SP (10 mol/L) resulted in increased migration. Similarly, SP-induced increases in intracellular calcium concentration and actomyosin stress fiber formation. These effects were all blocked through specific NK-1 receptor antagonists. CONCLUSIONS: These results indicate that SP stimulates intestinal epithelial migration and increases in calcium concentration. These data support a beneficial role for SP in the maintenance of intestinal mucosal homeostasis. PMID: 17435548 [PubMed - indexed for MEDLINE] PMCID: PMC1877018 Related Links Display 20 Substance P induces intestinal wound healing via fibroblasts--evidence for a TGF-beta-dependent effect. [Int J Colorectal Dis. 2007] Differentiated intestinal epithelial cells exhibit increased migration through polyamines and myosin II. [Am J Physiol. 1999] NF-kappaB regulates intestinal epithelial cell and bile salt-induced migration after injury. [Ann Surg. 2003] Activation of K(+) channels and increased migration of differentiated intestinal epithelial cells after wounding. [Am J Physiol Cell Physiol. 2002] Neurokinin-1 receptor (NK-1R) expression is induced in human colonic epithelial cells by proinflammatory cytokines and mediates proliferation in response to substance P. [J Cell Physiol. 2003] » See all Related Articles... AbstractPlus Sort By Show Write to the Help Desk NCBI | NLM | NIH Department of Health & Human Services Privacy Statement | Freedom of Information Act | Disclaimer J Cell Physiol. 2003 Oct;197(1):30-41. Links Neurokinin-1 receptor (NK-1R) expression is induced in human colonic epithelial cells by proinflammatory cytokines and mediates proliferation in response to substance P. Goode T, O'Connor T, Hopkins A, Moriarty D, O'Sullivan GC, Collins JK, O'Donoghue D, Baird AW, O'Connell J, Shanahan F. Department of Medicine, National University of Ireland, Cork, Ireland. We have previously shown that the receptor for substance P (SP), neurokinin-1 receptor (NK-1R), is a marker of human mucosal but not peripheral mononuclear cells. In the present study, we investigate NK-1R expression in the human colonic mucosa in vivo, particularly in the epithelial cells. We investigate the influence of proinflammatory Th1 cytokines and SP on expression and function of NK-1R in colonic epithelial cells in vitro. Using in situ hybridization to detect NK-1R mRNA, and immunohistochemistry to detect NK-1R protein, colonic epithelial cells were found to express NK-1R in vivo. In contrast, colon epithelial cell lines (Caco-2, HT29, SW620, T84) were negative for NK-1R mRNA and protein. However, stimulation with a proinflammatory cytokine cocktail containing IFNgamma, TNF-alpha, and IL-1beta, caused induction of NK-1R expression. Expression of NK-1R in human colonic epithelial cells in vivo may therefore reflect cytokine conditioning by the mucosal microenvironment. SP did not alter ion transport in monolayers of cytokine-treated T84 cells. While SP stimulated epithelial ion transport in colonic mucosae ex vivo, this was not a direct effect of SP on the epithelial cells, and appeared to be neurally mediated. However, SP (10(-10)-10(-8) M) elicited a dose-dependent proliferative effect on cytokinestimulated, but not unstimulated, SW620 cells. Proliferation of the epithelial cells in response to SP was mediated specifically via cytokine-induced NK-1R, since an NK-1R-specific antagonist (Spantide 1) completely blocked SP-mediated proliferation in the cytokine-treated cells. Our results therefore demonstrate that proinflammatory cytokines induce expression of NK-1R in human colonic epithelial cell lines, and that SP induces proliferation of the epithelial cells via cytokine-induced NK-1R. Copyright 2003 Wiley-Liss, Inc. PMID: 12942538 [PubMed - indexed for MEDLINE Lab Invest. 2003 May;83(5):731-42.Links Neurokinin-1 receptor expression and its potential effects on tumor growth in human pancreatic cancer. Friess H, Zhu Z, Liard V, Shi X, Shrikhande SV, Wang L, Lieb K, Korc M, Palma C, Zimmermann A, Reubi JC, Büchler MW. Department of General Surgery, University of Heidelberg, Germany. helmut_friess@med.uni-heidelberg.de The neurokinin-1 receptor (NK-1R) and its ligand substance P (SP) are involved in the pathogenesis of certain neural tumors. Because nerves are significantly altered in pancreatic cancer, evidence for alteration of this pathway in human pancreatic cancer was sought. Expression of NK-1R was analyzed by real-time quantitative RT-PCR, in situ hybridization, immunohistochemistry, and Western blot analysis in normal human pancreatic and pancreatic cancer tissue samples and in pancreatic cancer cell lines. Furthermore, the influence of SP analogs and of the NK-1R antagonist MEN 11467 on pancreatic cancer cell growth was analyzed by sulforhodamine B (SRB) assay. By real-time quantitative RT-PCR, NK-1R mRNA was increased 36.7-fold (p < 0.001) in human pancreatic cancer samples compared with normal controls. Enhanced NK-1R expression levels were not related to tumor grade but were associated with advanced tumor stage and poorer prognosis. By in situ hybridization and immunohistochemistry, NK-1R mRNA and immunoreactivity were only occasionally weakly present in acinar and ductal cells in the normal pancreas. In contrast, moderate to strong NK-1R mRNA signals and immunoreactivity were present in most cancer cells. By Western blot analysis, NK-1R was increased 26-fold (p < 0.01) in pancreatic cancer samples in comparison to normal controls. NK-1R mRNA was detected in five pancreatic cancer cell lines by real-time quantitative RT-PCR, with the highest levels in CAPAN-1 cells and the lowest in ASPC-1 cells. SP analogs stimulated pancreatic cancer cell growth, depending on the NK-1R expression level, and this effect could be blocked by a selective NK-1R antagonist. These findings illustrate that the NK-1R pathway is activated in human pancreatic cancer and has the potential to contribute to cancer cell growth, thus suggesting the existence of a neuro-cancer cell interaction in vivo. PMID: 12746482 [PubMed - indexed for MEDLINE] Clin Diagn Lab Immunol. 2000 May;7(3):371-6. Links Differential expression of neurokinin-1 receptor by human mucosal and peripheral lymphoid cells. Goode T, O'Connell J, Ho WZ, O'Sullivan GC, Collins JK, Douglas SD, Shanahan F. Department of Medicine, National University of Ireland, Cork, Ireland. Substance P (SP) has been implicated in peripheral and mucosal neuroimmunoregulation. However, confusion remains regarding immunocyte expression of the receptor for SP, neurokinin-1 receptor (NK-1R), and whether there is differential NK-1R expression in the mucosal versus the peripheral immune system. In the same assay systems, we examined the expression of NK1R in human lamina propria mononuclear cells (LPMC), peripheral blood mononuclear cells (PBMC), peripheral blood lymphocytes (PBL), monocytes, and monocyte-derived macrophages (MDM). Using standard reverse transcription (RT)-PCR, mRNA expression of both the long and the short isoforms of the NK1R was evident in LPMC but not in PBMC, PBL, monocytes, or MDM. However, by using nested RT-PCR NK-1R mRNA expression was detected in PBMC, PBL, monocytes, and MDM. This level of expression was found to represent one NK1R mRNA transcript in >1,000 cells. In contrast, by using competitive RT-PCR we demonstrate that LPMC express a more biologically significant level of eight NK-1R mRNA transcripts per cell. Flow cytometric detection of NK-1R expression at the protein level was evident in LPMC but not in PBMC. These findings illustrate the extreme sensitivity of nested RT-PCR and the advantages of competitive RT-PCR in comparative studies of receptor expression in different cell populations. This study suggests that, under normal conditions, readily detectable expression of NK-1R in human mononuclear cells occurs at the mucosal level rather than in the peripheral circulation. PMID: 10799448 [PubMed - indexed for MEDLINE] J Neurosci Res. 2006 Nov 15;84(7):1588-96. Links Morphine upregulates functional expression of neurokinin-1 receptor in neurons. Wan Q, Douglas SD, Wang X, Kolson DL, O'Donnell LA, Ho WZ. Division of Allergy and Immunology, Joseph Stokes Jr. Research Institute at The Children's Hospital of Philadelphia, Department of Pediatrics, Philadelphia, Pennsylvania. Neuronkinin-1 receptor (NK-1R), the neuropeptide substance P (SP) preferring receptor, is highly expressed in areas of the central nervous system (CNS) that are especially implicated in depression, anxiety, and stress. Repeated exposure to opioids may sensitize neuronal systems involved in stress response. We examined the effects of morphine, the principal metabolite of heroin, on the functional expression of NK-1R in the cortical neurons. NK-1R and mu-opioid receptor (MOR) are co-expressed in the cortical neurons. Morphine enhanced NK-1R expression in the cortical neurons at both the mRNA and protein levels. The upregulated NK-1R by morphine had functional activity, because morphinetreated cortical neurons had greater SP-induced Ca(2+) mobilization than untreated neurons. Blocking opioid receptors on the cortical neurons by naltrexone or CTAP (a mu-opioid receptor antagonist) abolished the morphine action. Investigation of the mechanism(s) responsible for the morphine action showed that morphine activated NK-1R promoter and induced the phosphorylation of p38 MAPK protein in the cortical neurons. These in vitro data provide a plausible cellular mechanism for opioid-mediated neurological disorders. PMID: 16983662