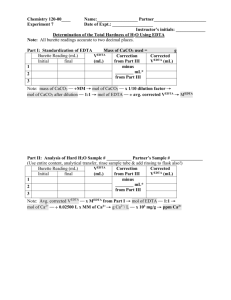
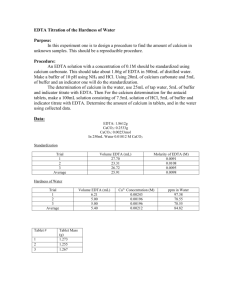
Name
advertisement

Name Date Chapter 15 • Water and Aqueous Systems Class SMALL-SCALE EXPERIMENT HARD AND SOFT WATER Small-Scale Experiment for text Section 15.3 OBJECTIVES Measure the total hardness of water in various water samples. Apply the ion exchange process to “soften” hard water. INTRODUCTION Hard water contains dissolved salts, especially those of calcium, magnesium, and iron. Most of the dissolved calcium ions in the waters of rivers, lakes, and streams originate from limestone, which is principally calcium carbonate (CaCO3). Limestone is dissolved by rainwater or by water contaminated by mining and industrial wastes containing hydrochloric and sulfuric acids. CaCO3(s) + H+(aq) Ca2+(aq) + HCO3–(aq) The reaction of water with atmospheric carbon dioxide makes precipitation slightly acidic. Acidic precipitation can also result from the reaction of atmospheric water with industrial pollutants such as sulfur dioxide and nitrogen dioxide. H2O(l) + CO2(g) H2CO3(aq) H2O(l) + SO2(g) H2SO3(aq) H2O(l) + 2NO2(g) HNO2(aq) + HNO3(aq) The location of limestone deposits varies throughout the country, as do the amounts of industrial acids contaminating rivers and contributing to the acidity of precipitation. Thus, the amount of dissolved calcium ion varies from place to place and so does the resultant hardness of water. Calcium ions, while nontoxic, present a number of household and industrial problems. For example, calcium ion reacts with the ingredients of many soaps and shampoos to form an insoluble scum that you can observe as a “bathtub ring.” The formation of this precipitate causes soap to lose some of its effectiveness. Ca2+(aq) + 2CH3(CH2)16COONa(aq) 2Na+(aq) + (CH3(CH2)16COO)2Ca(s) sodium stearate a calcium stearate an typical soap insoluble scum Hard water also accounts for the buildup of boiler scale on the insides of teapots, water heaters, and industrial boilers. The scale consists mainly of calcium carbonate that has precipitated from hard water upon heating: Ca(HCO3)2(aq) + heat CaCO3(s) + CO2(g) + H2O Calcium carbonate scale must be removed periodically because it reduces heating efficiency by acting as an insulator. Calcium carbonate also promotes corrosion and blocks pipes. Experiment 25 Hard and Soft Water 177 Name Date Class PURPOSE In this experiment, you will measure the total amount of calcium and magnesium ions in various water samples from around the country and/or from your area. You will add a particular kind of solution, called a buffer, to each water sample, which will maintain the pH at about 10.5. You will then titrate each sample with ethylenediamine tetraacetic acid, EDTA, until the indicator Ero-T turns from wine red to sky blue at the end point. You will report the results as parts per million (ppm) of calcium carbonate, CaCO 3, even though some of the hardness may be due to the presence of magnesium ions. A part per million is a milligram of calcium carbonate per liter of water sample. The higher the parts per million of CaCO3, the more calcium ions are in the water, and the harder the water is. SAFETY Wear safety goggles, an apron, and gloves when working with corrosive chemicals. Use full small-scale pipets only for the controlled delivery of liquids. Don’t chew gum, drink, or eat in the laboratory. Never taste a chemical in the laboratory. Avoid inhaling substances that can irritate your respiratory system. MATERIALS Small-scale pipets of the following solutions: various water samples (labeled) deionized water ammonia/ammonium chloride (buffer) Solids: eriochrome black T indicator (Ero-T) ethylenediamine tetraacetic acid (EDTA) EQUIPMENT small-scale balance with extra pan, or other balance EXPERIMENTAL PROCEDURE Determine the hardness of each water sample by the following method: 1. Obtain the mass of about 2500 mg of a water sample in a plastic cup. a. Place a 2500-mg weight in your small-scale balance pan, and adjust the balance beam and counterweight until the pointer indicates the zero point. b. Remove the weight and drop in enough water sample to return the pointer to the zero point. 2. Add 15 drops of buffer. 3. Add Ero-T indicator until the color is medium to dark wine red. 178 Small-Scale Chemistry Laboratory Manual Name Date Class 4. Mass or tare a full pipet of EDTA. a. Place a full pipet in a second small-scale balance pan. b. Adjust the small-scale balance to read zero. 5. Titrate the water sample with EDTA until the last tinges of red turn blue. 6. Find the mass of EDTA that was lost. a. Return the used EDTA pipet to the small-scale balance. b. Add weights until the pointer again reads zero. 7. Repeat the above steps for each of the other water samples including tap water and distilled water. Compare the hardness of water in the various other samples available in your lab. 8. Calculate the parts per million (ppm) of CaCO3 in the water sample. (1 ppm = 1 mg CaCO3/L of water.) Record your results in Table 25.1. To do the calculation, you will need the following pieces of information: a. The mass loss of EDTA used in the titration in mg. b. The mass of the water sample in mg. c. The density of EDTA solution = 1000 mg/1 mL. d. The density of water = 1000 mg/1 mL. e. 1000 mL = 1 L or 1000 mL/1 L. f. The concentration of EDTA solution = 0.002 mmol/mL. g. The overall stoichiometry of the reaction = 1 mmol CaCO3/1 mmol EDTA. h. The molar mass of CaCO3 = 100 mg/mmol. ppm CaCO3 mg CaCO3 /L H 2O a d ef gh b c To be sure you understand the calculation, substitute each quantity (number and units) into the equation and cancel the units. For subsequent calculations, simplify the equation by combining terms that are always constant. The indicator is red in the presence of Mg2+. EDTA reacts with both Ca2+ and Mg2+. The indicator turns blue when all of the Mg2+ and Ca2+ are combined with EDTA. Equations: Mg2+ + 2In– = MgIn2 blue red EDTA2– + Ca2+ → CaEDTA EDTA2– + Mg2+ → MgEDTA Experiment 25 Hard and Soft Water 179 Name Date Class EXPERIMENTAL DATA Record your results in Table 25.1 or in a copy of the table in your notebook. Table 25.1 Titration Data for Water Samples Mass of Sample (mg) Mass Loss of EDTA (mg) ppm CaCO3 Water Sample ______________ ______________ ______________ ______________ ______________ CLEANING UP Avoid contamination by cleaning up in a way that protects you and your environment. Carefully clean the plastic balance cup by rinsing the contents down the drain. Rinse the cup and dry it with a paper towel. Dispose of the paper towels in the waste bin. Wash your hands thoroughly with soap and water. QUESTIONS FOR ANALYSES Use what you learned in this experiment to answer the following questions. 1. Which sample is the hardest water you titrated? Which is the softest? What does it mean for water to be “hard” or “soft”? 2. What is a part per million? 3. In the titration, why did you not need to know the mass of the full EDTA pipet? 4. Write a balanced equation for the titration of calcium ion with EDTA. 180 Small-Scale Chemistry Laboratory Manual Name Date Class 5. EDTA is commonly used as an additive in foods. The function of the EDTA is to tie up metal cations that may cause clouding of the product. Read some labels at home and find some foods that contain EDTA in some form. List them here. NOW IT’S YOUR TURN! 1. When water is passed through an ion exchange buret, it becomes acidic because Ca+ ions are exchanged for H+ ions. The equation for the ion exchange reaction follows: Ca2+ + +H R H+ Ca2+ R + 2 H+ R = ion exchange “resin” This principle is used in a practical everyday setting in a home water softener. a. Recharge an acid resin ion exchanger with a small amount of HCl. b. Drain it thoroughly and rinse it twice with deionized water. Expel as much of the rinse water as possible. c. Draw a few drops of hard water into the exchanger and shake gently for 15 seconds. Add five drops of this water to one drop of BTB and record the color. What color does each water sample that was treated in the ion exchanger turn BTB? What ions are present in these samples? d. Expel five more drops from the exchanger, shake in a few grains of Ero-T indicator, and add a few drops of buffer. Record the color. e. Repeat for the other water samples. What color does each water sample that was treated with the ion exchanger turn Ero-T? What does this tell you about the presence of calcium ion? Explain. Experiment 25 Hard and Soft Water 181