Carbon Dioxide Lab
advertisement

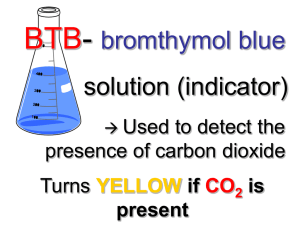
Sampling Carbon Dioxide Materials for each team 4 test tubes w/ stoppers Graduated cylinder Beaker Flask of BTB Dropper bottle of dilute ammonia Balloon Piece of string Straw Sharpie Marker Part 1: Chemical Test for Carbon Dioxide Teams need to work together in this experiment because most steps require more than one pair of hands. 1. Label the 4 test tubes A --> D. Test tube A will be your control. Rinse graduated cylinder well before use Measure and pour 15 ml of BTB into the tube. Cap test tube A and place in your test tube holder. 2. Collect a sample of pure carbon dioxide (CO2). Use the soda stream to collect a sample of pure CO2. Be careful to not break the plastic nozzle. Measure the circumference of the balloon in cm using string. All samples collected should be the same volume. Circumference will be what you use to standardized volume. 3. Secure and measure (pure CO2). Twist the rubber neck of the balloon so that no gas can leak out. Measure 15 ml of BTB into test tube B. Insert the end of the straw into the neck of the balloon. Wrap the balloon tight around the straw so it makes a tight seal. Insert the other end of the straw into the BTB. SLOWLY untwist the balloon so the gas escapes very slow through the straw and bubbles through the BTB solution. Allow all the gas to bubble through. 4. Observe the changes. What shades of colors does it pass through before reaching a final color when all the gas has been bubbled through it? 5. Measure CO2. Slowly add dilute ammonia, uniform drop by drop (titration). Add a drop. Agitate the tube. If it turns the original blue color, wait a moment to see if it remains blue. Count the number of drops needed to return the solution to its original color. Compare to tube A. You may need to add a drop or two after a few minutes if you notice the color has changed compared to the control. Record number of drops used in the data table. 6. Collect remaining samples. Collect a similar volume of air (using the air pump), human breath, and 2 different samples from car exhaust. *marks the cars you should sample. Ms. Murphy will give you instruction for collecting the car exhaust samples. Bubble samples through the BTB solution and use titration to determine the drops of ammonia needed to return the sample to its original color. Part 2: Analysis 1. Determine the percentage of CO2 in each sample. To calculate % for each sample it is based on the carbon dioxide sample having 100% and the control having 0%. Use the following formula to help calculate the percentage for each of the other samples. # ammonia drops for sample x 100 # ammonia drops for pure CO2 Pure CO2 Air Breath Car 1 Car 2 Car 3 Control Drops my group only % Table 1 Table 2 Table 3 Table 4 100 0 * * * * Table 5 Table 6 Table 7 Table 8 * * * * * * * * * * * * 2. Construct a graph – that compares the concentration of CO2 between the different samples from all the groups in class. Reference the graph scoring rubric (graph type and graph mechanics) 3. Respond to analysis questions. 1. Based on the whole class data do you feel that your CO2 results for the various samples are reliable? Explain 2. What might explain why concentrations varied among the lab groups with similar sources? 3. If a sample is suspected to contain CO2 but you don’t detect any using the BTB test, what might explain the results? 4. What does this investigation tell you about the concentration of CO2 emitted in animal breath compared to vehicle exhaust? 5. What additional information would you need to judge how much CO2 animals and vehicles each contribute to the total concentration of CO2 released into the atmosphere? 6. Using the evidence from the lab, describe how CO2 compares in the various samples. 7. Using evidence from this lab, write a scientific explanation that explains how CO2 concentration varies in different samples. 8. Currently there are approx.. 290,000,000 gasoline-burning on the road in the USA alone. By the year 2018, it is predicted there will be over 1,000,000,000 gas burning vehicles in the world. How do you think this will affect the concentration of carbon dioxide gas in the atmosphere? Explain. Figure 1 9. Consider figure 1 above. What kind of impact does fuel efficiency have on carbon dioxide released into the air? 10. Using evidence from this lab, make a claim about the impact that burning fossil fuels play in the carbon cycle. Describe how you might collect evidence to support your claim 11. State a position and defend with evidence and reasoning, whether new cars built and sold in the USA should be required to have high fuel efficiency of 40 mpg or more. Final Report Due 3/25 (white) and 3/26 (red) MUST be digital, all elements in 1 document, and turned in via eBackpack Scoring – 50 points Data – quality and completeness Graph(s) Analysis 15 pts 15 pts 20 pts