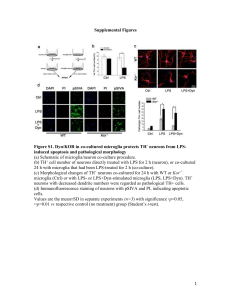
Assssing Function and Expression Differences Between Young and
advertisement

Contact Jennie Sims and Connie Wang jennie.sims.r@gmail.com, kwwang@caltech.edu Assessing Differences in Function and Expression Between Young and Aged Microglia SENS Foundation Academic Initiative Grant Proposal OVERVIEW Microglia are bone marrow-derived glial cells that serve as the phagocytic immune cells of the central nervous system (CNS). Microglial dysfunction has been hypothesized to play a role in many diseases. In particular, age-related loss of function may have a significant role in Alzheimer’s disease (AD), the most frequent cause of dementia in the elderly. In this proposal, we request a $3,000 grant to characterize loss of microglial function with age, using a variety of functional assays and gene expression assays. The results of these assays may provide insights into the mechanisms behind Alzheimer’s disease. They may also form a baseline for the behavior of “normal” young and old microglia to aid in the assessment of potential future therapies. 1 Contact Jennie Sims and Connie Wang jennie.sims.r@gmail.com, kwwang@caltech.edu TABLE OF CONTENTS OVERVIEW……………………………………………………………………………………………… 1 BACKGROUND……………………………………………………………………………………….. 3 Microglia: Function and Senescence……………………………………………….. 3 Relationship to Alzheimer’s disease………………………………………………… 3 PREVIOUS RESEARCH……………………………………………………………………………… 5 RESEARCH PLAN………………………………………………………………………………..…… 6 SPECIFIC AIMS………………………………………………………………………………………… 8 TEAM AND FACILITY….…………………………………………………………………………… 9 BUDGET….…………………………………………………………………………………………..… 11 SCIENTIFIC REFERENCES………………………………………………………………………… 12 2 Contact Jennie Sims and Connie Wang jennie.sims.r@gmail.com, kwwang@caltech.edu BACKGROUND Microglia: Function and Senescence Microglia make up about 5-12% of total brain cells1, 2 and act as the main immune cells of the CNS3. In healthy brains, microglia perform various neuroprotective functions by phagocytosing protein plaques, sick neurons, and other pathogens4, as well as by secreting cytokines and neurotrophic factors5. They typically take on a ramified, inactive form with long processes14, changing to an amoeboid morphology and becoming phagocytic upon detection of injury1. As one of the few neural cell types that undergo significant cell division 5, microglia are hypothesized to be affected by replicative senescence. Senescent rat microglia have been found to have shorter telomeres5 and larger, more deramified morphologies6 with fragmented cytoplasms7. Older microglia have also been observed in ex vivo studies to secrete higher amounts of inflammatory cytokines than younger controls8, 16. This increase in inflammatory cytokine production leads to an increase in acetylcholinesterase activity, which has been shown to negatively affect cognitive function 17. These observed differences between young and old microglia are especially significant because many microglial functions have the potential to be both neuroprotective and neurotoxic 9, which has confounded the results of previous microglia studies. For example, oxidative burst, a spike in ROS levels and production of lysosomal proteases, is an important microglial function that protects the brain from pathogens1. However, this same mechanism has also been shown to induce neuronal death in studies of old and dysfunctional microglia7, 10, 11. Relationship to Alzheimer’s disease Both the neuroprotective and neurotoxic roles of microglia have implicated them as important factors in Alzheimer’s disease (AD). AD is an age-related disease characterized by memory loss, extracellular amyloid-β (AB) deposition12, formation of intracellular neurofibrillary tau tangles10, loss of neuronal synapses, and significant neuronal death5. AD is the most common cause of dementia in the elderly, afflicting one in ten individuals over the age of 65 and nearly half of those over the age of 8513. In particular, amyloid-β plaques appear to play a key pathogenic role in AD15. Microglia have been shown not only to be recruited to AB plaques5, but also to phagocytose and break down AB in vitro12. Additionally, microglia isolated from mouse models of AD express lower levels of 3 Contact Jennie Sims and Connie Wang jennie.sims.r@gmail.com, kwwang@caltech.edu AB-degrading enzymes, as well as lower levels of AB receptor proteins (scavenger receptors) 16, implying that AB degradation by microglia may play a major role in Alzheimer’s disease’s pathology. Tau protein tangles, another hallmark of Alzheimer’s disease, have also been associated with microglia in various ways. AB buildup has been seen to immediately precede tau tangle formation in AD models10, and neuroinflammation from microglia can exacerbate phosphorylation and aggregation of tau protein18. In addition, microglial accumulation has been observed to match the progression rate of AD16, and activated microglial expansion appears to precede massive neural cell death19. All of these studies suggest that microglia play a key role in the control of AD, its pathogenesis, or both. Furthermore, the age-related nature of Alzheimer’s disease and the types of microglia found around AD-associated plaques seem to indicate that aging microglia play a key role in the pathology of AD16. 4 Contact Jennie Sims and Connie Wang jennie.sims.r@gmail.com, kwwang@caltech.edu PREVIOUS RESEARCH Both morphological and secretory distinctions between young and old microglia have been investigated in previous studies, suggesting that there are differences in functional efficiency and expression profiles that should be characterized further5, 6, 7,8,16. In particular, lysosomal acidification has been shown to be important in the breakdown of amyloid-β (AB), and the lysosomal pH of active microglia has been shown to be lower than that of inactive microglia3, 4. It is also significant that microglia from adult mice are less effective in phagocytosing fibrillar AB than microglia from postnatal mice7. These studies are the motivation behind our investigation of lysosomal acidification genes Ostm1 and Clc-7. Studies have not yet compared the lysosomal acidities of young and old mouse populations. Importantly, previous studies have shown that marker expression and actual functional capacity do not always correlate1, indicating the need for functional assays as well as an analysis of gene expression profiles. 5 Contact Jennie Sims and Connie Wang jennie.sims.r@gmail.com, kwwang@caltech.edu RESEARCH PLAN In order to characterize microglia isolated from mice, it is necessary to determine if they are able to perform phagocytosis, maintain adequate lysosomal acidity, and express an oxidative burst. All of these features are key components of microglial function. A comparison will be done with isolated mouse lymphocytes to assess similarity to non-neuronal phagocytic immune cells. Phase 1: Isolation of mouse microglia and lymphocytes. Phase 2: Phagocytosis assays will be performed to examine differences in the functionality of young and old microglia, and to confirm that microglia are still viable in accordance with current theories. Microglia will be assessed for the ability to produce an oxidative burst, phagocytose E. coli, and maintain lysosomal acidity. Phase 3: Isolation of mRNA and qRT-PCR analysis. Once the functionality of the microglia has been ascertained, mRNA will be isolated. Real time PCR analysis may show differences in gene expression of housekeeping genes, lysosomal acidification genes such as Clc-7 and OSTM-1, scavenger receptor genes such as SRA and CD36, protein degradation genes such as IDE and neprilysin, and inflammatory cytokines such as IL-1B and IL-6. Savenger receptor genes that remain constant with age, RAGE and MARCO, will be used as negative controls. Insulysin (IDE), Neprilysin and MMP9 will be used as positive controls for genes that decrease in expression with age. TNFα and IL-1β will be used as positive controls for genes that increase in expression with age Research Methods Collection and Purification of Microglia Prior to primary cell isolation, animals will be euthanized and brain tissue collected by the supplier. Brain tissue will be shipped in media at 4°C. Neural tissue will be collected from dissected mice and dissociated using a neural dissociation kit (130-092-628, Miltenyi Biotec, CA). The procedure will be altered to use the same enzyme mixes with longer incubation times and less harsh mechanical dissociation (using a 3mL plastic transfer pipette) to reduce shearing. After tissue dissociation into single cell suspension, 6 Contact Jennie Sims and Connie Wang jennie.sims.r@gmail.com, kwwang@caltech.edu magnetic separation will be performed using magnetic CD11b microbeads and magnetic separation columns (130-042-201, Miltenyi Biotec, CA). Phagocytosis Assay Microglia will be incubated for two hours at 37°C, followed by two hours in the presence of E. coli that have been transformed with a GFP-containing plasmid. The SENS Foundation already holds stocks of E. coli that have been confirmed by fluorescence microscopy to express GFP. The presence of the GFP will be detected by flow cytometry in three conditions: E. coli alone, microglia alone, and combined microglia/E. coli, for both young and old microglia. Fluorescence detected in the microglia population of the experimental microglia/E. coli sample will indicate that the microglia has phagocytosed GFP-expressing E. coli. Mononuclear effector cells from peripheral blood will be enriched by lymphocyte separation medium according to the manufacturer’s protocol, and used as a positive control. Acidification Assay The lysosome acidification assay (Invitrogen, P35361, NY) will be performed according to the manufacturer’s instructions, and the results will be analyzed using the Becton Dickinson FACScan flow cytometer. Reactive Oxygen Species (ROS) - Oxidative Spike Assay Cells will be incubated with dihydrorhodamine 123 (D-23806, Invitrogen, NY), which will be taken up by the cell and oxidized to cationic rhodamine 123, causing green florescence that will be measured using the Becton Dickinson FACScan flow cytometer. 7 Contact Jennie Sims and Connie Wang jennie.sims.r@gmail.com, kwwang@caltech.edu SPECIFIC AIMS Characterize isolated microglia on the basis of gene expression using RT-PCR. Confirm that isolated microglia retain the functional characteristics described in previous studies. Correlate the functional assays with gene expression data. 8 Contact Jennie Sims and Connie Wang jennie.sims.r@gmail.com, kwwang@caltech.edu TEAM AND FACILITY Team Kelsey Moody Kelsey currently serves as a research scientist at SENS Foundation, and heads the Research Center’s OncoSENS program. He has extensive experience in the methods described, having worked as Chief Technology Officer at ImmunePath, Inc, a biotechnology start-up specializing in cell-based therapies. He will serve as Principal Investigator on the project, and will directly supervise the work of all members of the team. Dave Halvorsen After completing his BSc, David spent several months as an intern at ImmunePath, Inc. culturing stem cells. He then interned at the SENS Foundation Research Center, producing lentivirus and working with the flow cytometer. He currently works as a researcher on the OncoSENS project. Jennie Sims Jennie is a senior at the University of Colorado, Boulder, where she is pursuing a triple major in molecular, cellular, and developmental biology, integrative physiology, and neuroscience, with a minor in leadership. She has two years of molecular and cellular biology laboratory experience. Connie Wang Connie is a junior at the California Institute of Technology, where she is pursuing a double major in bioengineering and business, economics, and management. Connie has laboratory experience in molecular biology and bioimaging. Thomas Hunt Thomas Hunt is an aspiring scientist who is interning at the SENS Foundation Research Center. He has a broad understanding of lab procedures, including aseptic technique and cell and tissue culturing. 9 Contact Jennie Sims and Connie Wang jennie.sims.r@gmail.com, kwwang@caltech.edu Facility All work on this project will be conducted at the SENS Foundation Research Center in Mountain View, California. The Research Center has a fluorescence microscope and Becton Dickinson FACScan flow cytometer for fluorescence-based assays, a real-time PCR machine (ABI Prism 7700) for qRT-PCR, and mammalian cell culture equipment. 10 Contact Jennie Sims and Connie Wang jennie.sims.r@gmail.com, kwwang@caltech.edu BUDGET Kits/Materials Cost ea. Qty Total cost Magnetic mRNA Isolation Kit 268.00 1 268.00 SYBR Master Mix kit 109.00 1 109.00 ROS Assay kit (Dihydrohodamine 123) 181.92 1 181.92 RT-PCR Primers 12.53 15 187.95 MultiScribe Reverse Transcriptase 74.00 2 148.00 pHrodo Phagocytosis Kit 873.99 1 873.99 Lymphocyte separation medium 31.78 1 31.78 Water Bath Sonicator 250.00 1 250.00 SUBTOTAL 2050.64 Husbandry Cost ea. Qty Total cost 30 mo. C57/BL6 mice 168.00 5 840.00 1 mo. C57/BL6 mice 28.00 5 140.00 Food, bedding, etc. 50.00 1 50.00 SUBTOTAL 1030.00 11 Contact Jennie Sims and Connie Wang jennie.sims.r@gmail.com, kwwang@caltech.edu SCIENTIFIC REFERENCES TOTAL 1. 3080.64 Hinze, A. & Stolzing, A. Differentiation of mouse bone marrow derived stem cells toward microglia-like cells. BMC Cell Biology 12, 35 (2011). 2. Kettenmann, H., Hanisch, U.-K., Noda, M. & Verkhratsky, A. Physiology of microglia. Physiol. Rev. 91, 461–553 (2011). 3. Majumdar, A. et al. Activation of microglia acidifies lysosomes and leads to degradation of Alzheimer amyloid fibrils. Mol. Biol. Cell 18, 1490–1496 (2007). 4. Majumdar, A., Capetillo-Zarate, E., Cruz, D., Gouras, G. K. & Maxfield, F. R. Degradation of Alzheimer’s amyloid fibrils by microglia requires delivery of ClC-7 to lysosomes. Mol. Biol. Cell 22, 1664–1676 (2011). 5. Flanary, B. The role of microglial cellular senescence in the aging and Alzheimer diseased brain. Rejuvenation Res 8, 82–85 (2005). 6. Streit, W. J., Sammons, N. W., Kuhns, A. J. & Sparks, D. L. Dystrophic microglia in the aging human brain. Glia 45, 208–212 (2004). 7. Floden, A. M. & Combs, C. K. β-Amyloid Stimulates Murine Postnatal and Adult Microglia Cultures in a Unique Manner. J. Neurosci. 26, 4644–4648 (2006). 8. Njie, E. G. et al. Ex vivo cultures of microglia from young and aged rodent brain reveal age-related changes in microglial function. Neurobiol. Aging 33, 195.e1–12 (2012). 9. Luo, X.-G., Ding, J.-Q. & Chen, S.-D. Microglia in the aging brain: relevance to neurodegeneration. Molecular Neurodegeneration 5, 12 (2010). 10. Blasko, I. et al. How chronic inflammation can affect the brain and support the development of Alzheimer’s disease in old age: the role of microglia and astrocytes. Aging Cell 3, 169–176 (2004). 12 Contact Jennie Sims and Connie Wang jennie.sims.r@gmail.com, kwwang@caltech.edu 11. Parvathenani, L. K. et al. P2X7 Mediates Superoxide Production in Primary Microglia and Is Up-regulated in a Transgenic Mouse Model of Alzheimer’s Disease. J. Biol. Chem. 278, 13309–13317 (2003). 12. Simard, A. R., Soulet, D., Gowing, G., Julien, J.-P. & Rivest, S. Bone marrow-derived microglia play a critical role in restricting senile plaque formation in Alzheimer’s disease. Neuron 49, 489–502 (2006). 13. Kawas, C., Gray, S., Brookmeyer, R., Fozard, J. & Zonderman, A. Age-specific incidence rates of Alzheimer’s disease: the Baltimore Longitudinal Study of Aging. Neurology 54, 2072–2077 (2000). 14. Nimmerjahn, A., Kirchhoff, F. & Helmchen, F. Resting microglial cells are highly dynamic surveillants of brain parenchyma in vivo. Science 308, 1314–1318 (2005). 15. Lee, J. K. et al. Intracerebral transplantation of bone marrow-derived mesenchymal stem cells reduces amyloid-beta deposition and rescues memory deficits in Alzheimer’s disease mice by modulation of immune responses. Stem Cells 28, 329–343 (2010). 16. Hickman, S. E., Allison, E. K. & El Khoury, J. Microglial dysfunction and defective betaamyloid clearance pathways in aging Alzheimer’s disease mice. J. Neurosci. 28, 8354– 8360 (2008). 17. Li, Y. et al. Neuronal–Glial Interactions Mediated by Interleukin-1 Enhance Neuronal Acetylcholinesterase Activity and mRNA Expression. J. Neurosci. 20, 149–155 (2000). 18. Bhaskar, K. et al. Regulation of tau pathology by the microglial fractalkine receptor. Neuron 68, 19–31 (2010). 19. Wada, R., Tifft, C. J. & Proia, R. L. Microglial activation precedes acute neurodegeneration in Sandhoff disease and is suppressed by bone marrow transplantation. Proc. Natl. Acad. Sci. U.S.A. 97, 10954–10959 (2000). 13