Materials and Methods. (doc 48K)
advertisement

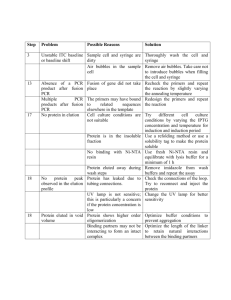
De Barros Hypoxia rejuvenates ASC properties from old donors SUPPLEMENTARY MATERIALS AND METHODS Adipose tissue cell isolation and culture of hASC Subcutaneous adipose tissue was obtained from donors aged 20 to 35 years (mean ± standard deviation: 29 ± 5 years old) and over 50 years (61 ± 7 years old) undergoing elective abdominal dermolipectomy in no obese patients (BMI < 26). No objection certificates were obtained according to the bioethic law n° 2004-800 of august 6, 2004. Cells were isolated as previously described [44]. Briefly, adipose tissue (AT) was digested in DMEM-F12 medium (Invitrogen, Carlsbad, USA) supplemented with 2 mg/ml collagenase A (Roche Diagnostic, Indianapolis, IN) and bovine serum albumin (BSA) (2%) for 45 min at 37°C, filtered through 100 μm and 25 μM nylon membrane and centrifuged at 600 g for 10 min to separate floating mature adipocytes from the stromal-vascular fraction (SVF). SVF was incubated in erythrocyte lysis buffer (ammonium chloride solution, StemCell Technologies, Vancouver, Canada) for 5 min at 4°C and washed in PBS (phosphate-buffered saline). SVF cells were counted and either used for flow cytometry analysis or seeded in vitro at 4 000 cell/cm2 in hASC expansion medium in 75 cm2 flasks The hASC culture medium was Good Manufacturing Practice (GMP): -MEM medium (Invitrogen) supplemented with 2% human plasma (EFS Midi-Pyrénées, France; enriched with human platelet factors), 100 µg/ml streptomycin, 100 U/ml penicillin, 25 µg/mL amphotericin (Invitrogen) and 10 U/mL heparin Choay (Sigma, St Louis, USA). Adherent cells were cultured for 10-14 days at 37°C in a humidified atmosphere of 5% CO2 until they reached confluence. The medium was renewed every 2 to 3 days. Colony Forming Unit-Fibroblasts Assay Freshly prepared SVF cells were seeded in 25 cm2 flasks at a final concentration of 8 cells/cm2 De Barros Hypoxia rejuvenates ASC properties from old donors in ASC expansion medium. The medium was renewed every 2 or 3 days. After 12 days, cells were washed with PBS and fixed with methanol for 15 min. The colony forming unit-fibroblasts (CFU-f) were stained with Giemsa (6%) for 30 min and scored under an optical microscope. Colonies were considered as clusters of more than 50 cells. In vivo vascular function analysis Vascular function was assessed 14 days after the onset of ischemia for control (0,9% NaCl) and treated (hASC) animal. Ischemic hindlimb conservation was first evaluated as percent of control. To provide evidence for ischemia-induced changes in vascularisation of the rescued limb, laser Doppler perfusion imaging experiments were performed in mice limbs, as previously described [13]. Mice were placed on a heating plate at 37°C to minimize temperature variation for imaging and limb perfusion was expressed as a ratio of right ischemic (I) to left non-ischemic (NI) leg. For capillary density mice were anesthetized by an intra peritoneal injection of ketaminexylazine before retro-orbital injection of Lectin GSL TRITC (Eurobio-Abcys) at 150 µl per mouse. After 30 min, mice received an intracardiac infusion of 3.6% formaldehyde before muscles (gastrocnemius, gracilis, and quadriceps) were removed, post-fixed at 4°C overnight, sunk in PBS, and frozen at -50°C in isopentane. Muscles were cut into 20 µm serial sections on a cryostat (Leica). Capillary density was evaluated using a Leica DM-RB microscope and NIS-AR image analysis software (Nikon). In vivo GFP-hASC tracking Replication defective, self-inactivating lentiviral vectors were provided by BiviC core vector production (IFR 150, Toulouse, France). Vectors were generated in a BSL-3 facility De Barros Hypoxia rejuvenates ASC properties from old donors concentrated by ultracentrifugation for viral titration, expressed in transduction unit per ml (TU/ml). hASC 3,300 cell/cm2 were transduced overnight in 4 ml of transduction medium containing serum-free medium, 15% BIT9500 (Stem cells Technologies, Grenoble, France) and 4 μg/ml Protamine Choay (Sanofi Aventis France, Paris, France) in the presence of purified lentiviral vectors at MOI 8. Cells were collected at least 72 h after transduction for EGFPpositive cells quantification by flow cytometry analysis on a fluorescent-activated cell sorter (FACS Canto II, Becton Dickinson, Mountain View, CA). For GFP-hASC injected mice, 200 µm muscle sections were done with vibroslice on fixed tissue and fluorescence analysis was performed utilizing a Zeiss LSM510 NLO confocal microscope. Quantification of hASC in mice muscle tissue 14 days after hASC injection, DNA from muscle was extracted using DNA kit extraction (Qiagen, Courtaboeuf, France) according to the manufacturer’s recommendations for tissue, and was used for PCR. To determine how many human cells were present in mouse muscle, DNA samples were amplified with the human alu-specific primers and mouse actineB primers and were compared to a standard curve performed with a mix of increasing amount of hASC (106105-104-103-102) to a muscle sample before DNA extraction. 20 ng of gDNA was analyzed by real time PCR in a final volume of 10 µl using Sso Advanced SYBR Green supermix (BIORAD) with the primers for alu, alu for: CAT GGT GAA ACC CCG TCT CTA, alu rev: GCC TCA GCC TCC CGA GTA G and moACTB, moACTB for: GAT GCA CAG TAG GTC TAA GTG GAG, moACTB rev: CAC TCA GGG CAG GTG AAA CT (0.3 µM final). qPCR conditions were as follows: 2 minutes at 95°C, followed by 40 cycles of 5 seconds at 95°C, 30 seconds at 60°C. This was followed by a dissociation stage for 5 seconds at 95 °C to De Barros Hypoxia rejuvenates ASC properties from old donors ensure the presence of a single amplicon. Real-time PCR assays were run on StepOne detection system instrument (Life Technologies/ Applied Biosystem). Real Time PCR Total RNAs were isolated from hASC using RNeasy Mini Kit (QIAGEN).About 1µg of total RNAs was reverse transcribed using random hexamers and Multiscribe reverse transcriptase (kit High Capacity cDNA Reverse Transcription kit, Life Technologies/ Applied Biosystem). 20 ng of cDNA were analyzed by real time PCR in a final volume of 20 µl using Power SYBRgreen master mix (Life Technologies/Applied Biosystem) with the primers for VEGF, HGF, IL6TNF and PUM (0.3 µM final) or using Taqman Universal PCR Master Mix (Life Technologies/Applied Biosystem) with primers and probe Taqman Gene Expression Assay for IGF1, TGF-2, IL8, CXCL1, p16INK4A and PUM (Life Technologies/Applied Biosystem). All primers sequences are detailed in Table S1. qPCR conditions were as follows: 10 minutes at 95°C, followed by 40 cycles of 15 seconds at 95°C, 1 minute at 60°C. This was followed by a dissociation stage for 15 seconds at 95 °C to ensure the presence of a single amplicon. Real-time PCR assays were run on StepOne detection system instrument (Life Technologies/ Applied Biosystem). Relative gene expression was calculated by the dCT method and normalized to PUM. Primers used for qPCR are detailed in Table S1. Primers were designed using Primer Express software (Applied Biosystems,) and validated by testing PCR efficiency using standard curves (85%≤ efficiency ≤115%), allowing us to use the ΔΔCt method for absolute quantification. PCR product specificity was evaluated by generating a dissociation curve following the manufacturer's recommendation.