ChemistryQ
advertisement

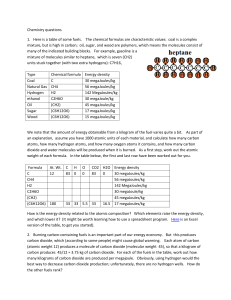
Chemistry questions 1. Here is a table of some fuels. The chemical formulas are characteristic values: coal is a complex mixture, but is high in carbon; oil, sugar, and wood are polymers, which means the molecules consist of many of the indicated building blocks. For example, gasoline is a mixture of molecules similar to heptane, which is seven (CH2) units stuck together (with two extra hydrogens): C7H16. Sticking more units together changes the freezing and boiling temperatures for the compound (and its tendency to evaporate), but so far as the energy content of a gallon or kilogram is concerned, they are all pretty much the same: “oil.” Type Coal Natural Gas Hydrogen ethanol Oil Sugar Wood Chemical formula C CH4 H2 C2H6O (CH2) (C6H12O6) (C6H12O6) Energy density 30 MegaJoules/kg 56 MegaJoules/kg 142 MegaJoules/kg 30 MegaJoule/kg 45 MegaJoules/kg 17 MegaJoules/kg 15 MegaJoules/kg Burning carbon-containing fuels is an important part of our energy economy. But this produces carbon dioxide, which (according to some people) might cause global warming. Each atom of carbon (atomic weight 12) produces a molecule of carbon dioxide (molecular weight 44), so that a kilogram of carbon produces 44/12 = 3.66 kg of carbon dioxide. For each of the fuels in the table, work out how many kilograms of carbon dioxide are produced per megajoule. Obviously, using hydrogen would the best way to decrease carbon dioxide production; unfortunately, there are no hydrogen wells. How do the other fuels rank? To answer this, you first have to work out the reaction of the fuel with oxygen. For example, burning wood looks like this C6H12O6 + 12O = 6CO2 + 6 H2O (The number of C’s, H’s, and O’s are the same on each side) The molecular weight of C6H12O6 is 6*12 + 12*1 + 6*16 = 180 grams/mole, and burning it produces 6 moles of CO2 = 6*44 g/(mole of wood) = 264 g/mole, so we get 264/180 = 1.47 kg of CO2 per kg of wood. Since the kg of wood is worth 15 MJ, we get 1.47/15 MJ = 0.098 kg of CO2 per MJ. Your turn! Fill in the table below. ©J. P. Straley ChemistryQdoc 1 Formula At. Wt. Number of C atoms C 12 1 g/mole CH4 H2 C2H6O (CH2) (C6H12O6) 180 6 g/mole Grams of CO2 CO2 per kg Energy density (per kilogram) CO2 per MJ 44 3.66 30 megaJoules/kg 0.122 kg/MJ 1.47 56 megaJoules/kg 142 MegaJoules/kg 30 megaJoule/kg 45 megaJoules/kg 17 megaJoules/kg 0.098 kg/MJ 264 2. The amount of energy obtainable from a kilogram of the fuel varies quite a bit. As part of an explanation, assume you have a mole of each material, and calculate how many carbon atoms, how many hydrogen atoms, and how many oxygen atoms it contains (measured in moles. A mole of hydrogen atoms is one gram, a mole of carbon atoms is 12 grams, and a mole of oxygen atoms is 16 grams), and how many moles of carbon dioxide and water molecules will be produced when it is burned. Multiplying the energy density (MJ/kg) by the molecular weight (kg/mole) tells you the energy per mole, which is proportional to the energy per molecule. Formula At. Wt. C g/mole 12 1 C CH4 H2 C2H6O (CH2) (C6H12O6) 180 6 H O 0 0 12 6 CO2 H2O Energy density (per kg) 1 0 30 megaJoules/kg 56 megaJoules/kg 142 MegaJoules/kg 30 megaJoule/kg 45 megaJoules/kg 6 6 17 megaJoules/kg Energy density (per mole) 0.36 MJ/mole 3.06 MJ/mole Now the puzzle is to figure out how is the energy density is related to the atomic composition. Which elements raise the energy per molecule, and which lower it? Can we write a formula Energy density per mole = A*(# of C atoms) + B*(# of H atoms) + C*(# of O atoms) ? ©J. P. Straley ChemistryQdoc 2 3. There is an active movement to ban the plastic bags that stores give out for free. There are many motivations for this. One of them is the claim that use of plastic bags increases our use of fossil fuels. Given that a plastic bag has mass 6 grams and that it is a polymer of CH2, how much hot water could we make by burning one? You will need to know that it takes 4186 J to raise the temperature of 1 kg of water by 1 Celsius degree; hot water is 50 C, some 40 C warmer than water from the water company. 4. Hydrogen and methane are gases, which makes it hard to store them in a small space. Gasoline and other forms of oil have high energy density, and this goes a long way to explaining why they are the fuels for transportation. But a car on the moon would have to carry along its oxygen, too. How many kg of oxygen do we need per kg of gasoline? What is the energy density (MJ per kg) when we include the mass of the oxygen? 5. Plants store energy from sunlight in the form of sugars (C6H12O6), perhaps polymerized into cellulose (wood) or other carbohydrates. After being buried for a long time under high pressure and temperature, they turn into coal, oil, and gas. The oxygen atoms end up either as CO2 or H2O. Write a reaction that will turn sugar into pure carbon, and a reaction that will turn sugar into methane. Show that in the process, the fuel value decreases (this has to be true, or energy would be created out of nothing). ©J. P. Straley ChemistryQdoc 3