Homework - Fort Thomas Independent Schools
advertisement

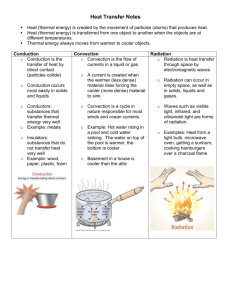
Homework P 424 1-5, 11-16 1. 2. 3. 4. 5. 6. 7. 8. 11. 12. 13. 14. 15. 16. Temperature is directly related to average KE of the particles in a substance. Celsius (0-100 C) Fahrenheit (32-212 F) Temperature is not based upon how much substance is present. It is based upon the average KE (vibrational speed) of the particles that make up the substance. Matter contains internal energy (thermal energy is the energy of the particles that compose matter). Heat is the quantity of thermal energy that flows from a hotter to a colder object.\ Heat flows from the hotter to the cooler object. Skip Thermal equilibrium is a condition where all objects are at the same temperature. This occurs after heat has been transferred from the hotter to the colder substance, so the hotter substance becomes colder and the colder substance becomes warmer until both are the same temperature. Internal energy is the kinetic and potential energy of the molecules that make up a substance. It means that it has a high or low ability to store internal energy. Substances that heat up quickly are conductors and have low specific heat capacities. The specific heat capacity of water is high. The ocean gives energy to the air above it in the winter, which is then carried over land, warming it. In the summer, the water absorbs the energy from the air above it, and the cooler air moves over the land and absorbs energy from it. The more conductive metal expands faster and contracts faster, which causes the strip to curl. Gases! Pg 463 2. Change of phase from liquid to gases absorbs thermal energy from the surroundings, leaving the surroundings cooler. 4. Change of phase from gas to liquid releases thermal energy into the surroundings, causing the surrounding to become warmer. 10.Evaporation only occurs at the surface, whereas boiling occurs throughout a liquid. 11.Atmospheric pressure inhibits boiling (does not allow it to cool), which means the water can increase in temperature beyond the normal boiling point.