הודעה על החמרה ( מידע בטיחות) בעלון לצרכן
advertisement

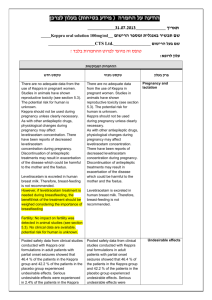
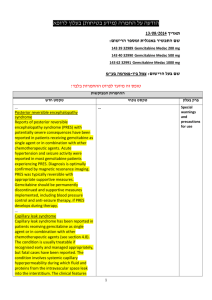
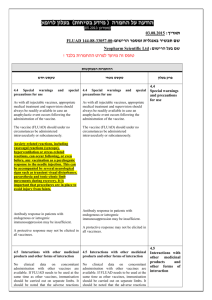
)בטיחות )מידע בטיחות החמרה (( מידע על החמרה הודעה על הודעה 12/07/2012:תאריך ARTHROTEC 50, ARTHROTEC 75 TABLETS :שם תכשיר באנגלית ARTHROTEC 50: 139 73 28553 00 :מספר רישום ARTHROTEC 75: 130 98 29104 00 פייזר פרמצבטיקה ישראל בע"מ:שם בעל הרישום השינויים בעלון מסומנים על רקע צהוב רופא בעלון ללרופא בעלון ים/ים המבוקש/פרטים על השינוי טקסט חדש טקסט נוכחי פרק בעלון Fertility Based on the mechanism of action, the use of NSAIDs, including diclofenac/misoprostol may delay or prevent rupture of ovarian follicles, which has been associated with reversible infertility in some women. In women who have difficulties conceiving or who are undergoing investigation of infertility, withdrawal of NSAIDs, including diclofenac/misoprostal should be considered. Pregnancy Diclofenac/misoprostol is contraindicated in women who are pregnant because misoprostol induces uterine contractions and is associated with abortion, premature birth, and fetal death. Use of misoprostol has been associated with birth defects (See sections 4.3 CONTRAINDICATIONS and 4.8 UNDESIRABLE EFFECTS). Diclofenac may cause premature closure of the ductus arteriosus. Women of childbearing potential should not be started on diclofenac/misoprostol until pregnancy is excluded, and should be fully counseled on the importance of adequate contraception while undergoing treatment. If pregnancy is suspected, use of the product should be discontinued. 4.6. FERTILITY, PREGNANCY AND LACTATION Pregnancy Diclofenac/misoprostol is contraindicated in women who are pregnant because misoprostol induces uterine contractions and is associated with abortion, premature birth, and fetal death. Use of misoprostol has been associated with birth defects (See sections 4.3 CONTRAINDICATIONS and 4.8 UNDESIRABLE EFFECTS). Diclofenac may cause premature closure of the ductus arteriosus. ... Inhibition of prostaglandin synthesis might adversely affect pregnancy. Data from epidemiological studies suggest an increased risk of spontaneous abortion after use of prostaglandin synthesis inhibitors in early pregnancy. In animals, administration of prostaglandin synthesis inhibitors has been shown to result in increased pre- and post-implantation loss. Women of childbearing potential should not be started on diclofenac/misoprostol until pregnancy is excluded, and should be fully counseled on the importance of adequate contraception while undergoing treatment. If pregnancy is suspected, use of the product should be discontinued. ... Postmarketing Surveillance: Blood and lymphatic system disorders: thrombocytopenia , platelet aggregation inhibition Postmarketing Surveillance: Blood and lymphatic system disorders: thrombocytopenia Immune system disorders: allergic reactions (including anaphylaxis and angioedema) Immune system disorders: allergic reactions (including anaphylaxis and angioedema) Metabolism and nutrition disorders: fluid retention Psychiatric disorders: changes in mood, nightmares Psychiatric disorders: changes in mood, nightmares Nervous system disorders: aseptic meningitis , 4.8. Undesirable effects stroke Nervous system disorders: aseptic meningitis , stroke Eye disorders: blurred vision Eye disorders: blurred vision Cardiac disorders: myocardial infarction Cardiac disorders: myocardial infarction Vascular disorders: vasculitis, hypertension Respiratory, thoracic and mediastinal disorders: dyspnea Gastrointestinal disorders: pancreatitis, stomatitis, gastrointestinal inflammation, gastrointestinal bleeding, gastrointestinal ulceration, gastrointestinal perforation Hepatobiliary disorders: hepatic failure, hepatitis, jaundice Skin and subcutaneous tissue disorders: cutaneous reactions (including rash and bullous eruption), dermatitis exfoliative, erythema multiforme, urticaria, mucocutaneous reactions, Stevens-Johnson syndrome, toxic epidermal necrolysis Renal and urinary disorders: interstitial nephritis, renal failure interstitial nephritis, glomerulonephritis, renal papillary necrosis, nephrotic syndrome, renal impairment Pregnancy, puerperium and perinatal conditions: abnormal uterine contractions, uterine rupture/perforation, retained placenta , amniotic fluid embolism,incomplete abortion, premature birth, fetal death Reproductive system and breast disorders: uterine hemorrhage, female fertility decreased Congenital, familial and genetic disorders: birth defects General disorders and administration site conditions: chills, fever , edema Respiratory, thoracic and mediastinal disorders: dyspnea Gastrointestinal disorders: pancreatitis, stomatitis, Hepatobiliary disorders: hepatic failure, hepatitis, jaundice Skin and subcutaneous tissue disorders: cutaneous reactions (including rash and bullous eruption), dermatitis exfoliative, erythema multiforme, urticaria, mucocutaneous reactions, Renal and urinary disorders: interstitial nephritis, renal failure Pregnancy, puerperium and perinatal conditions: abnormal uterine contractions, uterine rupture/perforation, retained placenta , amniotic fluid embolism,incomplete abortion, premature birth, fetal death Reproductive system and breast disorders: uterine hemorrhage Congenital, familial and genetic disorders: birth defects General disorders and administration site conditions: chills, fever